Advanced Manufacturing of Benzofuran-2(3H)-one: A Green Catalytic Breakthrough for Global Supply Chains
The global demand for high-purity heterocyclic intermediates continues to surge, driven by the expanding pharmaceutical and agrochemical sectors. Among these critical building blocks, benzofuran-2(3H)-one stands out as a versatile scaffold utilized in the synthesis of complex bioactive molecules, including novel antioxidants and agricultural agents like ICIA 5504. However, traditional manufacturing routes have long been plagued by environmental inefficiencies and cumbersome purification protocols. A pivotal shift in this landscape is documented in patent CN102746260A, which introduces a remarkably economical and environmentally benign methodology for preparing this valuable lactone. By leveraging silica sulfonic acid as a heterogeneous catalyst, this innovation addresses the chronic pain points of waste generation and catalyst recovery that have historically burdened producers. The technical breakthrough lies not merely in the substitution of reagents but in a fundamental re-engineering of the reaction workflow to prioritize atom economy and operational simplicity.
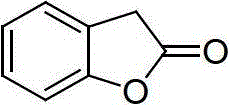
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of benzofuran-2(3H)-one has relied heavily on homogeneous acid catalysis or aggressive acylating agents, both of which present severe logistical and environmental drawbacks. One prevalent method utilizes dilute sulfuric acid to catalyze the dehydration condensation of o-hydroxyphenylacetic acid; however, this approach necessitates a subsequent neutralization step using alkali to quench the acid, resulting in the production of substantial quantities of saline wastewater that requires costly treatment. Another common route involves the use of acetic anhydride or excess acetyl chloride to induce cyclization, a strategy that generates stoichiometric amounts of acetic acid by-products which must be separated, thereby inflating raw material costs and complicating downstream purification. Furthermore, alternative pathways involving the hydrolysis of o-chlorophenylacetic acid derivatives suffer from similar issues, producing large volumes of sodium chloride waste and acidic effluents. These legacy processes are characterized by poor atom economy, high E-factors, and significant safety risks associated with handling corrosive liquid acids and volatile acyl chlorides on a commercial scale.
The Novel Approach
In stark contrast to these archaic techniques, the methodology disclosed in the patent employs silica sulfonic acid, a solid superacid catalyst, to drive the intramolecular cyclization of o-hydroxyphenylacetic acid with exceptional efficiency. This heterogeneous catalytic system operates under reflux conditions in the presence of a water-carrying agent, such as toluene or xylene, facilitating the continuous removal of reaction-generated water via azeotropic distillation. The use of a solid catalyst fundamentally transforms the workup procedure; instead of complex neutralization and extraction sequences, the catalyst is simply recovered by filtration, allowing for immediate reuse in subsequent batches. This streamlined process not only drastically reduces the consumption of auxiliary chemicals but also ensures that the only by-product of the reaction is water, aligning perfectly with green chemistry principles. The reaction equation illustrates this elegant transformation, where the starting material is cleanly converted into the target lactone with minimal structural degradation or side reactions.

Mechanistic Insights into Silica Sulfonic Acid-Catalyzed Cyclization
The efficacy of this synthesis hinges on the unique surface properties of silica sulfonic acid, which acts as a robust proton donor to activate the carboxylic acid moiety of the substrate. Mechanistically, the sulfonic acid groups anchored on the silica surface protonate the carbonyl oxygen of the acetic acid side chain, increasing its electrophilicity and rendering it more susceptible to nucleophilic attack. The ortho-positioned phenolic hydroxyl group then performs an intramolecular nucleophilic attack on the activated carbonyl carbon, forming a tetrahedral intermediate. Subsequent elimination of a water molecule, driven thermodynamically by the continuous azeotropic removal of water from the reaction vessel, leads to the formation of the stable five-membered lactone ring. This solid-state acid catalysis avoids the solvation effects often seen with liquid acids, potentially offering a distinct transition state environment that favors the desired cyclization over polymerization or other degradation pathways.
From an impurity control perspective, the heterogeneous nature of the catalyst plays a crucial role in maintaining high product purity. Because the active sites are confined to the solid surface, there is a reduced likelihood of promoting uncontrolled side reactions that often occur in bulk liquid acid solutions, such as sulfonation of the aromatic ring or ether formation. The patent data indicates that conversion rates consistently exceed 97%, with HPLC analysis confirming product purity levels ranging from 96.9% to 99.0% depending on the specific solvent system employed. The ability to terminate the reaction precisely when the residual starting material drops below 1% prevents over-reaction or thermal decomposition of the sensitive lactone product. Furthermore, the physical separation of the catalyst via filtration ensures that no metal or acidic residues contaminate the final organic phase, simplifying the crystallization or distillation steps required to meet stringent pharmaceutical specifications.
How to Synthesize Benzofuran-2(3H)-one Efficiently
The operational protocol derived from this patent offers a straightforward pathway for scaling this reaction from laboratory benchtop to pilot plant production. The process begins with the precise charging of o-hydroxyphenylacetic acid, a selected water-carrying solvent, and a catalytic amount of silica sulfonic acid into a reactor equipped with a Dean-Stark trap for water separation. Heating the mixture to reflux initiates the cyclization, with the progress of the reaction monitored by tracking the volume of water collected or by sampling for residual starting material. Once the reaction endpoint is reached, defined by the cessation of water evolution or HPLC confirmation of low starting material content, the mixture is cooled to allow for the safe recovery of the solid catalyst.
- Mix o-hydroxyphenylacetic acid with a water-carrying agent (such as toluene) and silica sulfonic acid catalyst in a reaction vessel.
- Heat the mixture to reflux while utilizing azeotropic distillation to continuously remove generated water from the system.
- Upon completion, cool the reaction, filter to recover the solid catalyst, and separate the solvent via distillation to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this silica sulfonic acid-catalyzed route presents a compelling value proposition centered on cost stability and operational resilience. The elimination of corrosive liquid acids and expensive acylating agents directly translates to a reduction in raw material expenditure and lowers the barrier for safe storage and handling within the facility. Moreover, the recyclability of the catalyst represents a significant long-term saving, as the same batch of silica sulfonic acid can be deployed across multiple production cycles without the need for frequent replenishment, effectively decoupling catalyst consumption from production volume. This efficiency gain is further amplified by the simplified post-reaction processing, which removes the need for neutralization tanks, salt filtration units, and extensive wastewater treatment infrastructure, thereby reducing both capital expenditure and ongoing utility costs.
- Cost Reduction in Manufacturing: The transition to a heterogeneous catalytic system eliminates the substantial costs associated with purchasing, storing, and disposing of large quantities of liquid sulfuric acid and neutralizing bases. By removing the neutralization step entirely, the process avoids the generation of tons of saline waste, which typically incurs high disposal fees and regulatory compliance costs. Additionally, the high atom economy of the reaction ensures that the majority of the input mass is converted into valuable product rather than waste by-products, maximizing the yield per kilogram of raw material purchased and optimizing the overall cost of goods sold.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply chain continuity by reducing dependency on specialized reagents like acetyl chloride, which can be subject to market volatility and strict transportation regulations. The use of common industrial solvents like toluene or xylene ensures that raw materials are readily available from multiple global suppliers, mitigating the risk of single-source bottlenecks. Furthermore, the simplicity of the workup procedure shortens the overall production cycle time, allowing manufacturers to respond more agilely to fluctuations in market demand and maintain consistent inventory levels for downstream customers.
- Scalability and Environmental Compliance: From a sustainability standpoint, this method offers a clear path to compliant large-scale manufacturing by minimizing the environmental footprint of the production process. The absence of heavy metal catalysts and the generation of water as the sole by-product simplify the permitting process and reduce the liability associated with hazardous waste management. The ease of scaling is evident in the straightforward equipment requirements—a standard reflux setup with a water separator—which can be easily replicated in larger reactors without the need for complex pressure vessels or cryogenic cooling systems often required by alternative synthetic routes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the underlying patent documentation, providing a factual basis for evaluating the feasibility of this route for your specific application needs. Understanding these nuances is critical for R&D teams assessing process transfer and for procurement specialists evaluating supplier capabilities.
Q: What are the primary advantages of using silica sulfonic acid over traditional sulfuric acid for this synthesis?
A: Silica sulfonic acid acts as a heterogeneous solid acid catalyst, which eliminates the need for neutralization steps required by liquid sulfuric acid. This significantly reduces salt waste generation and simplifies the post-reaction workup to a simple filtration process.
Q: Can the catalyst be reused in the production of benzofuran-2(3H)-one?
A: Yes, the patent explicitly demonstrates that the silica sulfonic acid catalyst can be recovered via filtration after the reaction and reused in subsequent batches without significant loss of catalytic activity, thereby lowering material costs.
Q: What is the typical conversion rate achieved with this green synthesis method?
A: According to the technical data in patent CN102746260A, the conversion rate of o-hydroxyphenylacetic acid can reach above 97%, with isolated yields consistently exceeding 97% across various solvent systems like toluene and chlorobenzene.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzofuran-2(3H)-one Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient synthetic routes is essential for maintaining competitiveness in the global fine chemicals market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits observed in laboratory patents are fully realized in industrial manufacturing. We are committed to delivering high-purity benzofuran-2(3H)-one that meets stringent purity specifications, supported by our rigorous QC labs which utilize advanced analytical techniques to verify identity and assay. Our capability to implement solid-acid catalyzed processes allows us to offer a product with a superior impurity profile, free from the salt contaminants often associated with traditional acid-catalyzed methods.
We invite you to engage with our technical procurement team to discuss how this optimized manufacturing route can benefit your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages specific to your volume requirements. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, ensuring that our benzofuran-2(3H)-one seamlessly integrates into your downstream synthesis of pharmaceuticals or agrochemicals.
