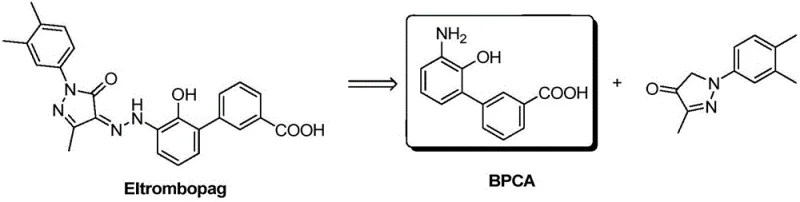
Advanced Aqueous Hydrogenation Strategy for High-Purity Eltrombopag Intermediate BPCA Manufacturing
The pharmaceutical industry continuously seeks robust synthetic routes for critical thrombopoietin receptor agonists, and patent CN111087315A represents a significant technological leap in the manufacturing of Eltrombopag intermediates. This specific intellectual property discloses a highly efficient method for synthesizing 3'-amino-2'-hydroxybiphenyl-3-carboxylic acid, commonly referred to as BPCA, which serves as a pivotal building block in the final assembly of the active pharmaceutical ingredient. The core innovation lies in the strategic substitution of traditional organic solvents with water, coupled with a precise catalytic hydrogenation protocol that operates under alkaline conditions. By fundamentally altering the reaction medium from alcohol to water, the inventors have successfully addressed long-standing issues regarding impurity profiles and operational complexity. This report analyzes the technical merits of this aqueous hydrogenation strategy, demonstrating how it aligns with modern green chemistry principles while delivering the high-purity standards required by global regulatory bodies. For R&D directors and procurement specialists, understanding this shift is crucial for evaluating potential suppliers who can leverage such cost-effective and environmentally benign processes to secure the supply chain for this vital medication.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the Eltrombopag intermediate BPCA has been plagued by significant inefficiencies inherent to the use of alcoholic solvents such as methanol or ethanol. In these conventional protocols, the hydrogenation of the nitro-biphenyl precursor typically results in a reaction mixture where the product remains dissolved, necessitating a subsequent concentration step to remove the solvent before isolation can occur. This concentration process is not only energy-intensive but also extends the duration during which the sensitive intermediate is exposed to elevated temperatures and potential oxidative environments. Furthermore, the amino and hydroxyl groups present on the biphenyl ring of the BPCA molecule are highly susceptible to oxidation, particularly under alkaline conditions or upon prolonged exposure to air. In alcohol-based systems, the removal of solvent often leads to the formation of oxidized dimers and other complex impurities that are notoriously difficult to separate from the desired product. These impurities directly compromise the purity of the final Eltrombopag API, forcing manufacturers to implement rigorous and costly purification steps that reduce overall yield and increase the environmental footprint of the production facility.
The Novel Approach
The methodology outlined in patent CN111087315A introduces a paradigm shift by utilizing water as the primary reaction solvent, thereby circumventing the drawbacks associated with volatile organic compounds. In this novel approach, the nitro-biphenyl starting material is mixed with an alkali, a catalyst, and water, followed by the introduction of hydrogen to drive the reduction. A key advantage of this aqueous system is that the product, upon completion of the reaction and subsequent acidification, precipitates directly from the solution without the need for solvent evaporation or concentration. This elimination of the concentration step drastically simplifies the post-treatment workflow and significantly reduces the time the intermediate is vulnerable to oxidative degradation. By maintaining the reaction in an aqueous phase and swiftly adjusting the pH to the isoelectric point (approximately pH 5) under controlled conditions, the process minimizes the generation of oxidation impurities. The result is a streamlined operation that not only enhances the chemical purity of the BPCA intermediate but also aligns with sustainable manufacturing goals by reducing waste generation and solvent consumption.
Mechanistic Insights into Catalytic Hydrogenation in Alkaline Aqueous Media
The success of this synthetic route relies heavily on the precise control of the catalytic hydrogenation mechanism within an alkaline aqueous environment. The reaction utilizes transition metal catalysts, specifically palladium substances like palladium on carbon (Pd/C) or palladium hydroxide, although Raney nickel is also noted as a viable alternative. The presence of the alkali, such as sodium hydroxide or potassium carbonate, plays a dual role: it facilitates the dissolution of the carboxylic acid starting material by forming a soluble salt, and it creates a basic environment that influences the electronic state of the nitro group during reduction. The hydrogen gas, introduced at pressures ranging from 0.3 to 5 MPa, adsorbs onto the catalyst surface and reacts with the nitro group to form the corresponding amine. Crucially, the reaction is exothermic, and the patent specifies maintaining temperatures between 20°C and 80°C depending on the specific substrate (chloro-substituted vs. non-chloro). This thermal control is vital to prevent over-reduction or side reactions that could compromise the integrity of the biphenyl scaffold. The rapid absorption of hydrogen indicates the efficiency of the catalyst in this medium, ensuring that the conversion to the amino intermediate is both rapid and complete.
Impurity control is mechanistically achieved through the management of the intermediate's redox stability. The BPCA molecule contains a phenolic hydroxyl group ortho to the newly formed amino group, a structural motif that is prone to autoxidation. In the patented process, the reaction mixture is kept under an inert atmosphere or reduced air contact immediately after hydrogenation. The workup involves filtering off the catalyst and then carefully adjusting the pH of the filtrate to approximately 5 using an acidic substance like hydrochloric acid. At this pH, the zwitterionic nature of the amino acid causes it to precipitate out of the aqueous solution. This immediate precipitation effectively removes the product from the reactive alkaline solution where oxidation is most favorable. By avoiding the concentration of alcohol solutions which would leave a thin film of reactive material exposed to air for extended periods, this aqueous precipitation method kinetically traps the product in its reduced, high-purity state, thereby suppressing the formation of the problematic oxidative dimers that plague conventional syntheses.
How to Synthesize Eltrombopag Intermediate BPCA Efficiently
The operational procedure for implementing this technology is designed for scalability and ease of execution in standard pharmaceutical manufacturing equipment. The process begins with the preparation of an aqueous alkaline solution, into which the nitro-biphenyl precursor and the heterogeneous catalyst are dispersed. Once the reactor is sealed and purged with nitrogen to remove oxygen, hydrogen is introduced to initiate the reduction. The reaction progress is monitored by hydrogen uptake, and once absorption ceases, a brief holding period ensures complete conversion. The subsequent workup is notably simple: the catalyst is removed via filtration, and the clear filtrate is treated with acid to induce crystallization. This straightforward sequence eliminates the need for complex extraction or distillation units, making it highly attractive for large-scale production. For detailed operational parameters and specific stoichiometric ratios, please refer to the standardized synthesis guide below.
- Mix the nitro-biphenyl precursor with alkali (e.g., NaOH), catalyst (Pd/C), and water in a hydrogenation reactor.
- Introduce hydrogen gas at 0.3-5 MPa pressure and maintain temperature between 20-80°C until hydrogen absorption ceases.
- Filter off the catalyst, adjust the filtrate pH to approximately 5 using acid to precipitate the pure BPCA solid, then dry.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this aqueous hydrogenation technology offers substantial benefits that resonate deeply with procurement managers and supply chain leaders focused on cost efficiency and reliability. The primary driver for cost reduction is the replacement of expensive and flammable organic solvents with water, which is universally available and virtually free. Furthermore, the elimination of the solvent concentration step translates directly into reduced energy consumption, as there is no need to power large-scale evaporators or distillation columns to recover alcohol. This simplification of the unit operations also means that batch cycles can be completed more rapidly, increasing the throughput of existing manufacturing assets without the need for capital expansion. The reduction in waste generation, particularly the avoidance of contaminated alcoholic waste streams, further lowers the environmental compliance costs associated with waste disposal and treatment. These factors combine to create a manufacturing profile that is not only economically superior but also more resilient to fluctuations in raw material markets.
- Cost Reduction in Manufacturing: The transition to a water-based solvent system fundamentally alters the cost structure of Eltrombopag intermediate production. By removing the requirement for large volumes of methanol or ethanol, manufacturers eliminate the recurring expense of purchasing, storing, and recovering these solvents. Additionally, the energy savings derived from skipping the concentration step are significant, as thermal separation processes are among the most energy-intensive operations in fine chemical synthesis. The simplified workflow also reduces labor hours and equipment maintenance costs associated with complex solvent recovery systems. Consequently, the overall cost of goods sold (COGS) for the BPCA intermediate is drastically lowered, allowing for more competitive pricing strategies in the global marketplace while maintaining healthy margins.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the complexity of synthesis and the availability of specialized reagents. This patented method enhances reliability by utilizing commodity chemicals such as water, sodium hydroxide, and hydrogen, which are readily available in almost any industrial region. The robustness of the reaction conditions, which tolerate a range of temperatures and pressures, ensures consistent production outcomes even with minor variations in utility supplies. Moreover, the high purity achieved directly from the crystallization step reduces the risk of batch failures due to specification deviations. This consistency allows supply chain planners to forecast inventory levels with greater confidence and reduces the need for safety stock buffers that tie up working capital.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to commercial production often introduces unforeseen challenges, particularly regarding heat transfer and mass transfer in viscous or hazardous solvent systems. The aqueous nature of this reaction mitigates many of these risks, as water has excellent heat capacity and poses minimal fire hazard compared to alcohols. The process is described as suitable for continuous operation, which is the gold standard for high-volume manufacturing, enabling seamless scale-up from pilot plants to multi-ton production lines. From an environmental standpoint, the reduction in volatile organic compound (VOC) emissions and hazardous waste generation simplifies the permitting process and ensures compliance with increasingly stringent global environmental regulations, safeguarding the long-term viability of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of Eltrombopag intermediates, based on the specific innovations disclosed in the patent literature. These insights are intended to clarify the operational advantages and quality assurances provided by this advanced manufacturing route. Understanding these details helps stakeholders make informed decisions about supplier qualification and process validation.
Q: Why is water preferred over alcohol solvents for BPCA synthesis?
A: Water eliminates the need for energy-intensive concentration steps required with alcohol solvents. It also reduces the exposure time of the sensitive amino-phenol intermediate to air, significantly lowering the formation of oxidative dimer impurities.
Q: What catalysts are effective for this hydrogenation process?
A: The patent specifies palladium-based catalysts such as Palladium on Carbon (Pd/C) or Palladium Hydroxide, as well as Raney Nickel. Pd/C is highlighted as the most preferred option for optimal activity and selectivity.
Q: How does the alkaline condition improve product quality?
A: Maintaining an alkaline environment during hydrogenation stabilizes the intermediate. Crucially, the post-reaction workup involves rapid acidification to pH 5 under reduced air contact, which weakens the reducibility of the BPCA and prevents oxidation into difficult-to-remove dimers.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Eltrombopag Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceuticals like Eltrombopag depends on a supply chain built on technical excellence and unwavering quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the demanding volume requirements of global drug developers. We have integrated advanced green chemistry principles, such as the aqueous hydrogenation techniques described in recent patents, into our own manufacturing platforms to deliver high-purity intermediates with superior impurity profiles. Our rigorous QC labs and stringent purity specifications guarantee that every batch of BPCA we produce meets the exacting standards necessary for downstream API synthesis, providing our partners with the confidence they need to advance their clinical and commercial programs.
We invite you to engage with our technical procurement team to discuss how our capabilities can support your specific project needs. By leveraging our expertise in process optimization, we can provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how our efficient manufacturing methods can reduce your overall procurement spend. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to evaluate the tangible benefits of partnering with a supplier who prioritizes both innovation and reliability in the production of critical pharmaceutical intermediates.
