Optimizing Flunixin Meglumine Production: A Deep Dive into High-Efficiency Catalytic Synthesis for Global Supply Chains
Optimizing Flunixin Meglumine Production: A Deep Dive into High-Efficiency Catalytic Synthesis for Global Supply Chains
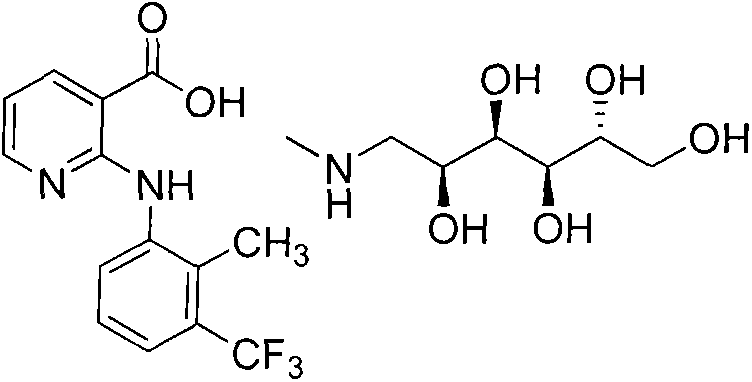
The veterinary pharmaceutical landscape demands increasingly efficient and cost-effective manufacturing routes for critical active ingredients, particularly for widely used anti-inflammatory agents like Flunixin Meglumine. Patent CN103694167A introduces a transformative synthetic methodology that addresses long-standing inefficiencies in the production of this vital compound. By leveraging a novel dual-catalyst system comprising cupric oxide and p-toluenesulfonic acid in an aqueous medium, this technology achieves a remarkable total yield of approximately 90%, significantly outperforming historical benchmarks. The process not only streamlines the reaction kinetics but also simplifies downstream processing through intelligent pH-controlled precipitation strategies. For R&D directors and supply chain managers, this represents a pivotal shift towards greener, more economical manufacturing that does not compromise on the stringent purity standards required for veterinary drug registration, such as EP 7.5 compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
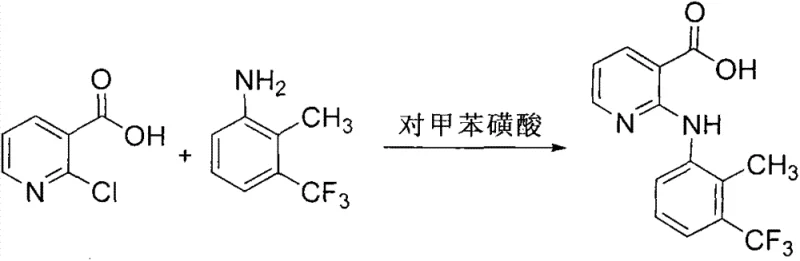
Historically, the synthesis of Flunixin has been plagued by suboptimal yields, harsh reaction conditions, and cumbersome purification protocols that inflate operational expenditures. As illustrated in prior art such as US5484931, traditional methods often rely solely on p-toluenesulfonic acid as a catalyst, necessitating a large excess of 2-methyl-3-trifluoromethylaniline to drive the reaction, which results in yields stagnating around 83% and generates significant waste. Other approaches, depicted in literature like Heterocycles (1994), utilize high-boiling solvents like ethylene glycol at temperatures reaching 165°C, creating severe energy burdens and complicating solvent recovery due to the difficulty of removing high-boiling components. These legacy processes often involve multi-step sequences with intermediate esterifications and hydrolyses, each step introducing potential yield losses and increasing the complexity of impurity profiles. The reliance on organic solvents and extreme thermal conditions not only escalates safety risks but also imposes heavy loads on wastewater treatment facilities, making these routes increasingly untenable in a modern regulatory environment focused on sustainability and cost containment.
The Novel Approach
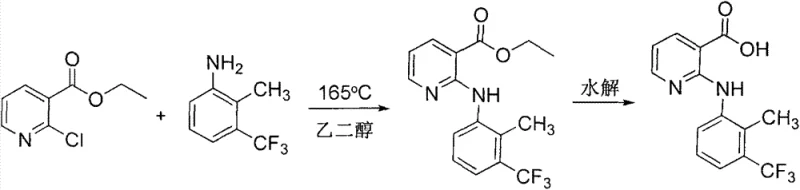
The innovative strategy detailed in CN103694167A fundamentally re-engineers the reaction landscape by introducing cupric oxide as a co-catalyst alongside p-toluenesulfonic acid, creating a synergistic effect that dramatically accelerates reaction rates in water. This dual-catalyst system allows the condensation of 2-chloronicotinic acid and 2-methyl-3-trifluoromethylaniline to proceed efficiently under mild reflux conditions, eliminating the need for the excessive reagent ratios and extreme temperatures characteristic of older methods. The process is elegantly simple: after the reaction reaches completion, typically within 2 to 4 hours, the mixture is cooled and treated with an alkali solution to adjust the pH to 10.0–11.0, which effectively precipitates unreacted aniline for immediate recovery and reuse. Subsequent acidification of the filtrate to pH 5.0–6.0 induces the crystallization of high-purity Flunixin, bypassing the need for chromatographic purification or complex extractions. This streamlined workflow not only boosts the isolated yield of the intermediate to over 95% but also ensures that the final salification with meglumine in acetonitrile produces a product meeting >99.9% HPLC purity specifications with minimal effort.
Mechanistic Insights into CuO/p-TsOH Dual-Catalyzed Condensation
The efficacy of this synthesis lies in the cooperative interaction between the Brønsted acid (p-toluenesulfonic acid) and the Lewis acidic properties of cupric oxide (CuO) within the aqueous phase. While p-toluenesulfonic acid protonates the pyridine nitrogen or the carboxylic acid group to enhance electrophilicity, the copper species likely coordinates with the chlorine leaving group or the carbonyl oxygen, facilitating the nucleophilic aromatic substitution by the aniline derivative. This coordination lowers the activation energy barrier for the displacement of the chloride ion, allowing the reaction to proceed rapidly at the boiling point of water rather than requiring the superheated conditions of solvent-free protocols. The presence of water as a solvent further stabilizes the transition states through hydrogen bonding networks, while the insolubility of the copper species may create a heterogeneous catalytic surface that promotes localized high concentrations of reactants. This mechanistic synergy explains the observed reduction in reaction time and the ability to use stoichiometric or near-stoichiometric amounts of the expensive aniline precursor, as the equilibrium is driven forward more effectively than in single-catalyst systems.
From an impurity control perspective, the pH-swing purification strategy is a masterclass in process chemistry design. By exploiting the basicity of the unreacted 2-methyl-3-trifluoromethylaniline, the process converts it into a free base that precipitates out of the acidic aqueous solution upon basification, allowing for its physical removal via simple filtration. This step is critical for preventing the carryover of genotoxic impurities or colored byproducts into the final API. The subsequent acidification to the isoelectric point of Flunixin ensures that the product crashes out of solution with high selectivity, leaving soluble inorganic salts and polar byproducts in the mother liquor. This precise control over solubility parameters minimizes the formation of inclusion complexes or solvates, which are common pitfalls in crystallization processes, thereby ensuring a consistent crystal form and particle size distribution that is essential for downstream formulation into veterinary injectables or boluses.
How to Synthesize Flunixin Efficiently
The operational protocol derived from this patent offers a robust framework for laboratory and pilot-scale production, emphasizing simplicity and reproducibility. The process begins with the suspension of 2-chloronicotinic acid and 2-methyl-3-trifluoromethylaniline in water, followed by the addition of the catalytic system (CuO and p-TsOH) and heating to reflux. Monitoring via HPLC ensures the reaction is halted once the starting acid content drops below 5%, typically after 2 to 4 hours. The workup involves a sequential pH adjustment: first to alkaline conditions to recover excess aniline, and then to acidic conditions to isolate the Flunixin intermediate. Finally, the dried intermediate is reacted with meglumine in acetonitrile at 70–80°C, followed by hot filtration to remove insolubles and controlled cooling to crystallize the final salt. For a comprehensive, step-by-step standard operating procedure including exact gram-scale quantities and safety precautions, please refer to the detailed guide below.
- Condense 2-chloronicotinic acid and 2-methyl-3-trifluoromethylaniline in water using cupric oxide and p-toluenesulfonic acid as catalysts under reflux for 2-4 hours.
- Adjust pH to 10-11 with alkali to remove unreacted aniline, then acidify filtrate to pH 5-6 to precipitate pure Flunixin.
- React purified Flunixin with meglumine in acetonitrile at 70-80°C, followed by hot filtration and cooling crystallization to obtain the final salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route translates directly into enhanced margin protection and supply security. The shift from expensive, high-boiling organic solvents to water as the primary reaction medium represents a drastic reduction in raw material costs and solvent disposal fees. Furthermore, the ability to recover and recycle the excess 2-methyl-3-trifluoromethylaniline—a relatively costly fluorinated building block—significantly lowers the effective material cost per kilogram of API produced. The simplified workup, which relies on filtration and crystallization rather than energy-intensive distillation or column chromatography, reduces utility consumption and shortens the overall cycle time per batch. These factors combine to create a manufacturing process that is not only economically superior but also more resilient to fluctuations in solvent markets and waste treatment capacity.
- Cost Reduction in Manufacturing: The elimination of high-boiling solvents like ethylene glycol removes the need for complex vacuum distillation equipment and the associated energy costs, leading to substantial operational savings. Additionally, the high catalytic efficiency allows for reduced catalyst loading and shorter reaction times, which increases reactor throughput and asset utilization without requiring capital investment in new hardware. The recovery of unreacted aniline further optimizes the bill of materials, ensuring that expensive fluorinated precursors are not wasted in the mother liquor.
- Enhanced Supply Chain Reliability: By utilizing water as the solvent and readily available inorganic catalysts like cupric oxide, the process reduces dependency on specialized or regulated organic solvents that may face supply chain disruptions. The robustness of the reaction conditions (atmospheric pressure, moderate temperature) minimizes the risk of batch failures due to equipment malfunction or thermal runaway, ensuring consistent delivery schedules. The high purity achieved (>99.9%) reduces the likelihood of regulatory rejection or customer complaints, securing long-term contracts with major veterinary pharmaceutical companies.
- Scalability and Environmental Compliance: The aqueous nature of the reaction aligns perfectly with green chemistry principles, significantly reducing the volatile organic compound (VOC) emissions associated with traditional organic synthesis. This makes the process easier to permit and operate in regions with strict environmental regulations. The simplicity of the isolation steps (filtration and crystallization) scales linearly from kilogram to multi-ton production, avoiding the engineering challenges often encountered when scaling up complex extraction or distillation processes, thus guaranteeing a stable supply of high-quality Flunixin Meglumine.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and beneficial effects reported in CN103694167A, providing a transparent view of the technology's capabilities. Understanding these details is crucial for technical teams evaluating the feasibility of technology transfer or scale-up activities.
Q: How does the CuO/p-TsOH dual catalyst system improve yield compared to traditional methods?
A: The addition of cupric oxide (CuO) alongside p-toluenesulfonic acid significantly enhances catalytic efficiency in aqueous media. While traditional methods using only p-TsOH achieve yields around 83% with excess reagents, this dual-catalyst system drives the reaction to completion more efficiently, achieving isolated yields of up to 95% for the intermediate and a total yield of approximately 90% for the final salt, while allowing for the recovery of unreacted starting materials.
Q: What are the environmental and cost benefits of using water as the primary solvent?
A: Utilizing water as the reaction solvent eliminates the need for expensive, high-boiling organic solvents like ethylene glycol or dimethylbenzene found in older protocols. This drastically reduces solvent procurement costs and simplifies waste treatment, as water is non-toxic and easy to handle. Furthermore, the workup involves simple pH adjustments and filtration, avoiding complex distillation steps required to remove high-boiling solvents.
Q: Is this synthesis method scalable for industrial production of veterinary APIs?
A: Yes, the method is specifically designed for industrial scalability. It operates at atmospheric pressure with standard reflux conditions (approx. 100°C) rather than the extreme temperatures (165°C+) required by solvent-free or high-boiling solvent methods. The equipment requirements are low, and the purification relies on straightforward crystallization and filtration, making it highly suitable for multi-ton commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Flunixin Meglumine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and rigorous quality control. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the CuO/p-TsOH catalytic system are fully realized in our manufacturing facilities. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art HPLC and GC-MS instrumentation to verify that every batch of Flunixin Meglumine meets or exceeds EP and USP standards. Our commitment to process optimization allows us to offer a product with superior impurity profiles, critical for the safety and efficacy of veterinary formulations.
We invite global partners to leverage our technical capabilities to secure a competitive advantage in the veterinary API market. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data from recent pilot batches and detailed route feasibility assessments to demonstrate how our optimized synthesis can enhance your supply chain efficiency and reduce your total cost of ownership.
