Optimizing Cinepazide Maleate Production: A Technical Analysis of Patent CN101602751A
Optimizing Cinepazide Maleate Production: A Technical Analysis of Patent CN101602751A
The pharmaceutical landscape for cardiovascular therapeutics continues to evolve, driven by the demand for safer, more efficient manufacturing processes for active ingredients. Patent CN101602751A, published in December 2009, introduces a transformative methodology for the preparation of Cinepazide Maleate, a potent piperazine derivative widely utilized for improving cerebral metabolism and treating cerebrovascular diseases. This patent addresses critical bottlenecks in the traditional synthesis of the key intermediate, 1-piperazine acetyl pyrrolidine, by shifting from hazardous organic solvents to a benign aqueous system. The structural complexity of the final API, as depicted below, requires precise control over the acylation steps to ensure high purity and minimize toxic impurities.
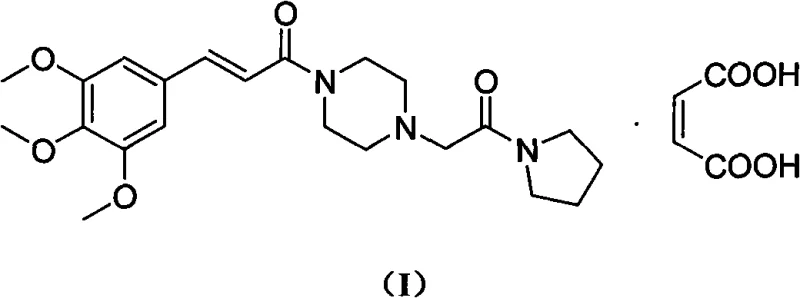
The core innovation lies in the strategic utilization of piperazine dihydrochloride, often a low-value byproduct in the synthesis of other piperazine-based drugs like Flunarizine Hydrochloride. By converting this salt into a reactive mono-hydrochloride species in situ, the process achieves a remarkable balance between reactivity and selectivity. This approach not only valorizes waste streams from upstream pharmaceutical manufacturing but also establishes a robust foundation for cost reduction in pharmaceutical intermediates manufacturing. For R&D directors and process chemists, this represents a significant opportunity to streamline supply chains and enhance the sustainability profile of their production portfolios without compromising on the stringent quality standards required for cardiovascular medications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Cinepazide Maleate and its precursors has been plagued by significant safety and efficiency challenges. Early methodologies, such as those disclosed in US Patent 3634411, relied heavily on dry benzene as a solvent for the acylation reaction. Benzene is a known carcinogen with high toxicity, posing severe occupational health risks and creating complex regulatory hurdles for waste disposal in modern pharmaceutical facilities. Furthermore, alternative routes described in literature, such as the synthesis of chloroacetyl pyrrolidine using diethyl ether (Farmaco, 1963), introduce extreme safety hazards due to the low boiling point and high flammability of ether, which can lead to explosive conditions during scale-up. These legacy processes often require rigorous anhydrous conditions and expensive solvent recovery systems, driving up operational expenditures.
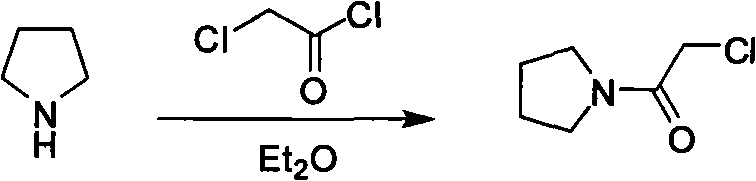
Beyond safety concerns, the atom economy and raw material utilization in conventional routes are suboptimal. For instance, methods disclosed in CN1631877A utilize a large excess of free piperazine to drive the reaction towards the mono-substituted product. This results in a significant amount of unreacted piperazine remaining in the mixture, which is difficult to separate from the product without resorting to energy-intensive wet distillation or vacuum distillation techniques. The formation of di-substituted byproducts is also a persistent issue under reflux conditions, complicating the purification process and lowering the overall yield of the desired intermediate. These inefficiencies translate directly into higher production costs and a larger environmental footprint, making such processes less attractive for modern, green chemistry-compliant manufacturing.
The Novel Approach
The methodology outlined in CN101602751A offers a paradigm shift by leveraging aqueous chemistry to overcome these historical limitations. Instead of relying on toxic organic solvents like benzene or chloroform, the new process utilizes water as the primary reaction medium. This fundamental change eliminates the fire hazards associated with ether and the toxicity issues linked to benzene, creating a inherently safer working environment. The process begins by reacting piperazine dihydrochloride with a base (such as NaOH or urea) in water to generate a piperazine mono-hydrochloride system. This in situ generation ensures that the concentration of free piperazine is carefully controlled, effectively suppressing the formation of di-substituted impurities without the need for a massive excess of starting material.
Furthermore, the patent introduces a cyclic feeding strategy where chloroacetyl pyrrolidine is added in batches to the reaction system containing the piperazine salt. This controlled addition, combined with pH regulation between 10 and 13, maximizes the conversion of the valuable piperazine moiety. The result is a high-yield process that produces 1-piperazine acetyl pyrrolidine with exceptional purity, achievable through simple extraction and crystallization steps rather than complex distillation. This streamlined workflow not only reduces energy consumption but also simplifies the equipment requirements, making it highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Aqueous Acylation and Selectivity Control
From a mechanistic perspective, the success of this synthesis relies on the delicate equilibrium between the protonated and deprotonated forms of piperazine in an aqueous environment. In traditional organic solvents, the high nucleophilicity of free piperazine often leads to over-acylation, resulting in the unwanted bis-amide byproduct. By starting with piperazine dihydrochloride and adding a stoichiometric amount of base, the process generates the mono-hydrochloride salt. This species acts as a buffer, releasing free piperazine gradually as it is consumed by the electrophilic chloroacetyl pyrrolidine. This "slow-release" mechanism kinetically favors the formation of the mono-substituted product (1-piperazine acetyl pyrrolidine) over the di-substituted side product.
The pH control step is critical in this mechanism. Maintaining the reaction mixture at a pH of 10 to 13 ensures that there is sufficient free base available to neutralize the HCl generated during the acylation, driving the reaction forward according to Le Chatelier's principle. However, the aqueous medium also facilitates the solubility of the inorganic salts formed (such as NaCl), allowing them to remain in the aqueous phase during the subsequent extraction. This phase separation is a powerful purification tool; the organic product partitions into the extraction solvent (e.g., dichloromethane or ethyl acetate), while the inorganic impurities and any remaining unreacted piperazine salts stay in the water. This intrinsic selectivity reduces the burden on downstream purification, ensuring that the final intermediate meets the rigorous purity specifications required for API synthesis.
Additionally, the use of urea or sodium carbonate as alkaline agents provides a milder basic environment compared to strong hydroxides, which can sometimes lead to hydrolysis of the sensitive amide bonds or the acid chloride moiety. The patent data indicates that this温和 (mild) condition preserves the integrity of the chloroacetyl group until it reacts with the piperazine nitrogen, thereby maximizing the yield. The ability to recycle the mother liquor or reuse the aqueous phase further enhances the green chemistry credentials of this route, aligning with global trends towards sustainable pharmaceutical manufacturing.
How to Synthesize 1-Piperazine Acetyl Pyrrolidine Efficiently
The synthesis of the key intermediate 1-piperazine acetyl pyrrolidine via this patented route involves a sequence of carefully controlled unit operations designed to maximize safety and yield. The process begins with the preparation of the reactive piperazine species in water, followed by the gradual addition of the acylating agent. The reaction is monitored by pH and temperature to ensure optimal conversion. Once the reaction is complete, the product is isolated through liquid-liquid extraction, washed to remove residual salts, and finally crystallized to achieve pharmaceutical-grade purity. The detailed standardized synthesis steps are provided in the guide below.
- React piperazine dihydrochloride with an alkali in water to generate a piperazine mono-hydrochloride system in situ.
- Add chloroacetyl pyrrolidine to the reaction system and maintain pH between 10 and 13 using alkaline substances.
- Extract the product, wash with sodium carbonate solution, dry, and crystallize using toluene to obtain high-purity 1-piperazine acetyl pyrrolidine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the technology described in CN101602751A offers compelling economic and logistical benefits. The most significant advantage is the drastic simplification of the raw material supply chain. By enabling the use of piperazine dihydrochloride—a common byproduct in the manufacture of other piperazine drugs—manufacturers can source feedstock from a wider, potentially lower-cost pool of suppliers. This reduces dependency on pure piperazine, the price of which can be volatile. Furthermore, the elimination of hazardous solvents like benzene and ether removes the need for specialized explosion-proof infrastructure and costly solvent recovery units, leading to substantial capital expenditure (CAPEX) savings for new facilities or retrofitting projects.
- Cost Reduction in Manufacturing: The process achieves cost optimization through multiple mechanisms. Firstly, the high atom economy resulting from the cyclic feeding strategy means that less raw material is wasted as unreacted starting material or byproducts. Secondly, the shift to water as a solvent eliminates the purchase and disposal costs associated with large volumes of organic solvents. Thirdly, the purification process avoids energy-intensive vacuum distillation, relying instead on simpler extraction and crystallization, which significantly lowers utility costs (steam and electricity). These factors combine to create a leaner, more cost-effective production model that enhances margin potential in a competitive generic drug market.
- Enhanced Supply Chain Reliability: The robustness of the aqueous reaction conditions contributes to greater supply chain stability. Unlike moisture-sensitive reactions that require strictly anhydrous conditions and inert gas protection, this process is tolerant to the presence of water, reducing the risk of batch failures due to environmental fluctuations. The use of common, non-hazardous reagents like sodium hydroxide and sodium carbonate ensures that raw materials are readily available from multiple global suppliers, mitigating the risk of supply disruptions. Additionally, the simplified workflow shortens the overall cycle time from raw material intake to finished intermediate, allowing for faster response to market demand fluctuations.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental risks, but this technology mitigates those risks at the source. The absence of flammable ethers and carcinogenic benzenes simplifies the permitting process for new manufacturing lines and reduces insurance premiums. The aqueous waste stream is easier to treat in standard effluent treatment plants compared to mixed organic waste, ensuring compliance with increasingly stringent environmental regulations (such as REACH in Europe or EPA guidelines in the US). This environmental compatibility future-proofs the manufacturing asset, ensuring long-term operational continuity without the threat of regulatory shutdowns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and claims within patent CN101602751A, providing a reliable basis for feasibility assessments. Understanding these details is crucial for technical teams evaluating the transfer of this technology to pilot or commercial scales.
Q: What are the primary safety advantages of the new synthesis route for Cinepazide Maleate?
A: The new route replaces hazardous solvents like benzene and diethyl ether with water, significantly reducing fire risks and toxicity exposure while simplifying waste treatment.
Q: How does this method improve raw material utilization compared to conventional processes?
A: By utilizing piperazine dihydrochloride (a pharmaceutical byproduct) and employing a cyclic feeding strategy, the process maximizes piperazine usage and minimizes unreacted starting material waste.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the method operates at mild temperatures (0-30°C) in an aqueous environment, avoiding complex vacuum distillation steps, which makes it highly scalable and energy-efficient for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cinepazide Maleate Supplier
The technological advancements detailed in patent CN101602751A underscore the potential for producing high-quality cardiovascular intermediates with superior efficiency and safety. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of critical intermediates. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of Cinepazide Maleate intermediate adheres to the highest international pharmacopoeia standards. We are committed to leveraging such innovative green chemistry routes to deliver value to our global partners.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to optimize their supply chains. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can support your long-term business goals.
