Revolutionizing Cinepazide Maleate Production: A Technical Breakthrough for Global Pharmaceutical Supply Chains
The pharmaceutical industry continuously seeks robust synthetic routes that balance efficiency, safety, and cost-effectiveness, particularly for cardiovascular agents like Cinepazide Maleate. Patent CN101508684A introduces a transformative methodology for synthesizing this calcium channel blocker, addressing critical bottlenecks found in legacy manufacturing processes. By shifting from a hazardous acid chloride intermediate to a mild, in situ carboxyl activation strategy, this technology offers a compelling value proposition for reliable pharmaceutical intermediates supplier networks aiming to optimize their production lines. The core innovation lies in the direct conversion of trans-3,4,5-trimethoxycinnamic acid into the target amide structure using alkyl chloroformates, effectively bypassing the isolation of unstable acyl chlorides. This technical advancement not only streamlines the operational workflow but also aligns with modern green chemistry principles by reducing the reliance on aggressive reagents like thionyl chloride. For procurement and technical teams, understanding this shift is vital for evaluating long-term supply security and cost reduction in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Cinepazide free base relied heavily on the preparation of trans-3,4,5-trimethoxycinnamoyl chloride as a discrete intermediate. As illustrated in prior art such as US3634411 and JP2180876A, this conventional pathway necessitates the use of thionyl chloride (SOCl₂) to activate the carboxylic acid, a process fraught with significant operational hazards. The generation of corrosive hydrogen chloride and sulfur dioxide gases demands specialized scrubbing systems and corrosion-resistant reactor materials, driving up capital expenditure. Furthermore, the resulting acid chloride is highly reactive and susceptible to hydrolysis, requiring strictly anhydrous conditions and often toxic solvents like anhydrous benzene or chloroform to maintain stability during the subsequent amidation step. These factors collectively contribute to a complex waste stream and lower overall process safety, making the traditional route less desirable for modern, regulated pharmaceutical environments.
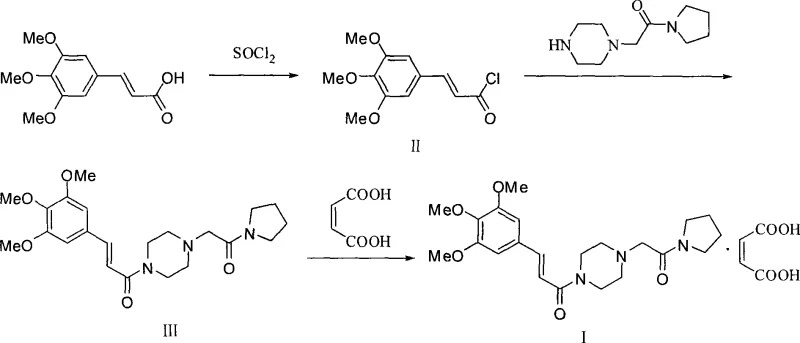
The Novel Approach
In stark contrast, the methodology disclosed in CN101508684A employs a "one-pot" activation strategy that fundamentally alters the reaction landscape. Instead of isolating the volatile acid chloride, the process utilizes alkyl chloroformates, such as ethyl chloroformate, in the presence of an organic base like triethylamine to generate a mixed anhydride intermediate in situ. This activated species then reacts directly with 1-[(1-tetrahydropyrrolecarbonyl)methyl]piperazine to form the desired amide bond. This approach eliminates the need for handling isolated acid chlorides, thereby removing a major safety hazard and simplifying the workup procedure. The reaction proceeds under mild conditions, typically between -20°C and 20°C, which significantly reduces energy consumption compared to the heating often required in older protocols. By integrating the activation and coupling steps, the new route achieves comparable or superior yields while drastically reducing the environmental footprint and equipment complexity.
Mechanistic Insights into One-Pot Carboxyl Activation
The mechanistic elegance of this synthesis lies in the controlled formation of a mixed anhydride, which serves as a highly electrophilic yet manageable acylating agent. When trans-3,4,5-trimethoxycinnamic acid is treated with ethyl chloroformate and triethylamine in dichloromethane, the carboxylate anion attacks the carbonyl carbon of the chloroformate, displacing the chloride ion. This generates a transient mixed anhydride species that is sufficiently reactive to undergo nucleophilic attack by the piperazine nitrogen but stable enough to prevent significant side reactions like self-condensation. The use of dichloromethane as a solvent is critical here, as it provides excellent solubility for both the organic acid and the amine components while facilitating efficient heat transfer during the exothermic activation phase. Maintaining the temperature at approximately -10°C during activation ensures that the mixed anhydride does not decompose before the amine is introduced, maximizing the conversion efficiency.
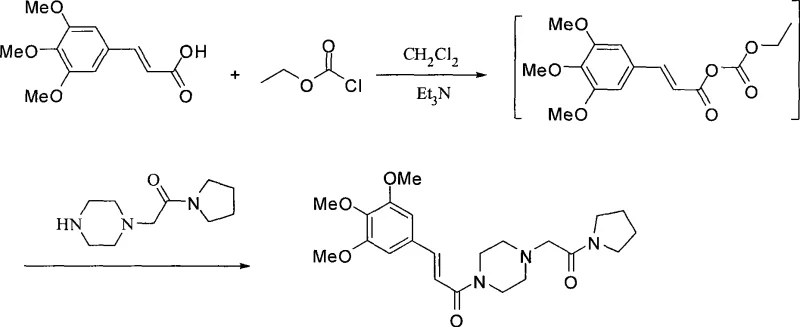
Impurity control is another critical aspect where this mechanism excels over the acid chloride route. In traditional methods, trace moisture can lead to the rapid hydrolysis of the acid chloride back to the starting acid, creating difficult-to-remove impurities that affect the final API purity. In the mixed anhydride approach, while hydrolysis is still a risk, the reaction kinetics favor the amidation when the amine is added promptly. Furthermore, the workup procedure described involves a precise pH swing—acidification to pH 1-2 followed by basification to pH 12-14—which effectively partitions the product from unreacted starting materials and urea byproducts formed from the activator. This rigorous purification protocol ensures that the resulting high-purity Cinepazide Maleate meets stringent pharmacopeial standards, minimizing the burden on downstream crystallization steps.
How to Synthesize Cinepazide Free Base Efficiently
Implementing this synthesis requires careful attention to stoichiometry and temperature control to replicate the high yields reported in the patent data. The process begins with the dissolution of the cinnamic acid derivative and the base in a dry solvent, followed by the slow addition of the activating agent to manage exotherms. Once the activated intermediate is formed, the amine component is introduced to drive the coupling reaction to completion. Detailed operational parameters, including specific molar ratios and agitation speeds, are essential for scaling this chemistry from gram-scale laboratory experiments to multi-kilogram production batches. For a comprehensive breakdown of the standardized operating procedures and quality control checkpoints required for this synthesis, please refer to the technical guide below.
- Dissolve trans-3,4,5-trimethoxycinnamic acid and triethylamine in dichloromethane at -10°C.
- Activate the carboxyl group by slowly adding ethyl chloroformate and stirring for 1 hour to form a mixed anhydride intermediate.
- Add 1-[(1-tetrahydropyrrolecarbonyl)methyl]piperazine solution dropwise, maintain temperature, and stir for 3 hours to complete amidation.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers substantial strategic advantages for procurement managers and supply chain directors focused on resilience and margin optimization. The elimination of thionyl chloride not only removes a hazardous material from the supply chain but also negates the need for expensive corrosion-resistant infrastructure, leading to significant capital expenditure savings. Moreover, the simplified "one-pot" nature of the reaction reduces the number of unit operations, which directly translates to shorter cycle times and increased throughput capacity within existing manufacturing facilities. This efficiency gain allows suppliers to respond more agilely to market demand fluctuations, ensuring a more stable supply of critical cardiovascular intermediates without the need for massive inventory buffers.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the reduction in raw material costs and waste disposal fees. By avoiding the use of thionyl chloride and toxic solvents like benzene, manufacturers can lower their expenditure on specialized hazardous waste treatment and personal protective equipment. Additionally, the higher atom economy of the mixed anhydride route compared to the acid chloride method means less raw material is wasted as byproducts, further enhancing the overall cost efficiency of the production line.
- Enhanced Supply Chain Reliability: Relying on stable, commercially available reagents like ethyl chloroformate and triethylamine mitigates the risk of supply disruptions often associated with highly regulated or hazardous chemicals. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in utility supply, such as cooling water temperature, ensuring consistent batch-to-bquality. This reliability is crucial for maintaining uninterrupted production schedules for downstream API manufacturers who depend on just-in-time delivery models.
- Scalability and Environmental Compliance: The mild reaction temperatures and standard solvent systems make this process inherently scalable, facilitating the commercial scale-up of complex pharmaceutical intermediates from pilot plants to full-scale production. The reduced generation of acidic gases and hazardous waste aligns with increasingly strict environmental regulations, minimizing the risk of regulatory shutdowns or fines. This environmental compliance not only protects the manufacturer's license to operate but also enhances the brand reputation of partners sourcing from this supply chain.
Frequently Asked Questions (FAQ)
To assist technical and procurement teams in evaluating this technology, we have compiled answers to common inquiries regarding the synthesis and quality attributes of Cinepazide Maleate produced via this method. These insights are derived directly from the experimental data and technical specifications outlined in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for assessing the feasibility of integrating this route into your existing supply network.
Q: Why is the one-pot activation method superior to the traditional acid chloride route?
A: The traditional route requires isolating unstable trans-3,4,5-trimethoxycinnamoyl chloride using toxic thionyl chloride, posing safety risks. The new method activates the acid in situ using ethyl chloroformate, eliminating the need to isolate hazardous intermediates and simplifying purification.
Q: What are the key cost drivers reduced in this new synthesis protocol?
A: Cost reductions are achieved by removing the thionyl chloride step, which requires specialized corrosion-resistant equipment and gas scrubbing systems. Additionally, the milder reaction temperatures (-10°C to 20°C) significantly lower energy consumption compared to refluxing conditions often used in older methods.
Q: Is this process scalable for industrial production of Cinepazide Maleate?
A: Yes, the patent explicitly states the method is suitable for industrial scale production. The use of common solvents like dichloromethane and standard organic bases like triethylamine ensures that the process can be easily transferred from laboratory to multi-ton manufacturing without complex engineering changes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cinepazide Maleate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving demands of the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the one-pot activation method are seamlessly translated into reliable supply solutions. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of Cinepazide Maleate delivered meets the highest international standards for safety and efficacy. Our infrastructure is designed to handle complex chemistries with precision, providing our partners with a secure and compliant source for their cardiovascular drug portfolios.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the potential economic impacts of switching to this greener, more efficient manufacturing process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume needs, ensuring a partnership built on transparency, technical excellence, and mutual growth.
