Advanced Enantioselective Synthesis of Statin Intermediates for Commercial Scale Manufacturing
Advanced Enantioselective Synthesis of Statin Intermediates for Commercial Scale Manufacturing
The pharmaceutical industry continuously seeks robust, scalable, and economically viable pathways for producing high-value Active Pharmaceutical Ingredients (APIs), particularly within the lucrative class of HMG-CoA reductase inhibitors, commonly known as statins. Patent CN1636004A introduces a groundbreaking methodology for the manufacture of mevalonic acid derivatives, specifically targeting key intermediates for drugs such as Pitavastatin and Fluvastatin. This technology represents a significant paradigm shift from traditional racemic synthesis followed by resolution, moving instead towards direct enantioselective construction of the critical 3,5-dihydroxy side chain. By leveraging advanced transition metal catalysis, specifically asymmetric transfer hydrogenation, this process addresses long-standing challenges regarding stereocontrol, material efficiency, and environmental impact. For R&D directors and procurement specialists, understanding the nuances of this patent is crucial for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity compounds with consistent batch-to-batch reproducibility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of statin side chains has relied heavily on the formation of racemic beta,delta-dihydroxy esters, which subsequently require chiral resolution to isolate the biologically active enantiomer. A prominent example found in prior art involves the synthesis of Pitavastatin precursors where a racemic erythro-beta,delta-dihydroxy ester is formed and then treated with chiral amines like alpha-methylbenzylamine to separate enantiomers. The fundamental economic and ecological flaw in this approach is the inherent maximum theoretical yield of 50% for the desired isomer, meaning half of the synthesized material must be discarded or subjected to energy-intensive recycling processes. Furthermore, traditional hydrogenation methods often employ catalysts that are highly sensitive to heteroatoms; for instance, the quinoline moiety present in Pitavastatin precursors is known to coordinate strongly with metal centers, leading to catalyst deactivation and incomplete reactions. These factors collectively drive up the cost of goods sold (COGS) and complicate the supply chain for high-purity pharmaceutical intermediates.
The Novel Approach
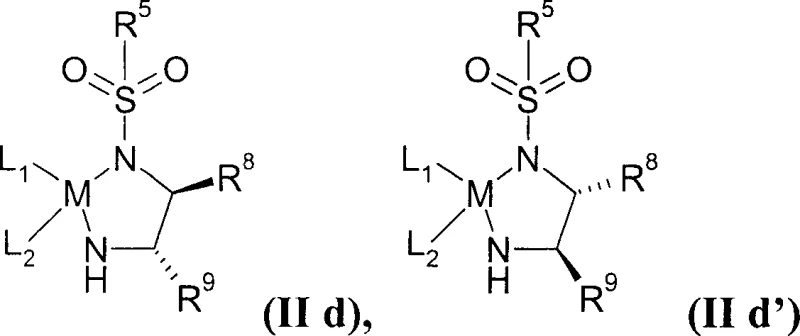
In stark contrast, the methodology disclosed in CN1636004A utilizes a sophisticated asymmetric transfer hydrogenation strategy that constructs the chiral centers with exceptional precision from the outset. The core innovation lies in the use of specific Ruthenium(II) complexes, such as those defined by Formula (IId), which facilitate the reduction of beta-keto esters directly to the desired (S)-hydroxy configuration with enantiomeric excess values exceeding 95%, and optimally reaching 99%. This eliminates the need for downstream resolution steps entirely, theoretically doubling the material efficiency compared to racemic routes. Moreover, the process demonstrates remarkable robustness; it tolerates the presence of quinoline rings without catalyst poisoning and, surprisingly, functions effectively even in the presence of water (up to 3% by Karl-Fischer analysis) and without strict inert gas atmospheres. This resilience translates to simplified reactor operations and reduced dependency on ultra-dry solvents, offering substantial cost reduction in API manufacturing.
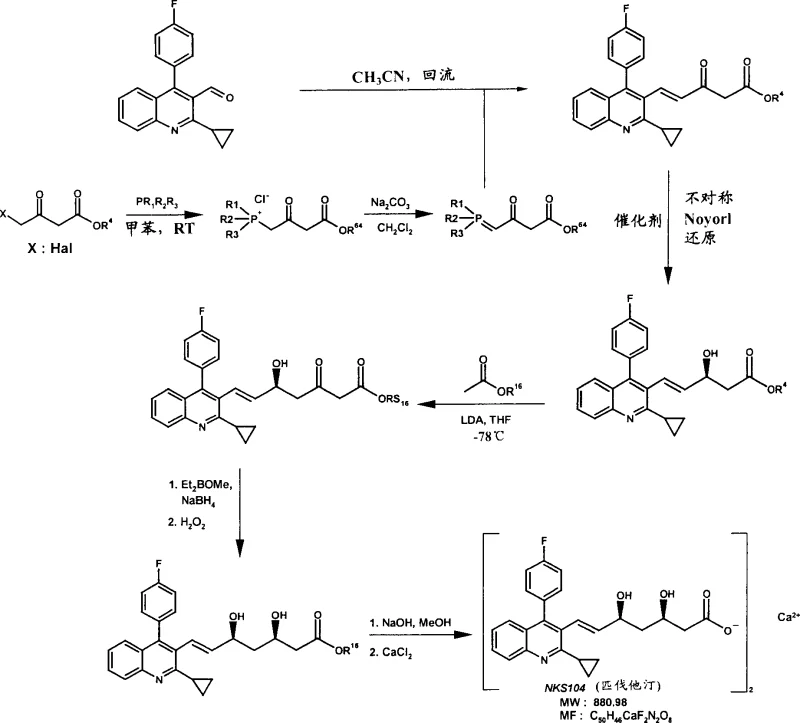
Mechanistic Insights into Ru-Catalyzed Asymmetric Transfer Hydrogenation
The heart of this technological advancement is the catalytic cycle involving chiral Ru(II) species ligated with diamines and arene groups, often referred to as Noyori-type catalysts. In the context of this patent, the catalyst operates via an outer-sphere mechanism where the hydride and proton are transferred concertedly from the metal-ligand complex to the carbonyl substrate. The specific ligands, such as p-toluenesulfonyl-1,2-diphenylethylenediamine (TsDPEN), create a chiral pocket that dictates the facial selectivity of the hydride attack on the prochiral ketone. What makes the catalysts described in Formula (IId) particularly unique is their electronic and steric tuning, which prevents the basic nitrogen atoms of the quinoline ring (in Pitavastatin synthesis) or the indole ring (in Fluvastatin synthesis) from binding irreversibly to the ruthenium center. Typically, such coordination would block the active site, but the specific ligand architecture here maintains catalytic turnover. Additionally, the mechanism accommodates hydrogen donors like formic acid/triethylamine azeotropes or 2-propanol, allowing for a clean reduction without the need for high-pressure hydrogen gas equipment, thereby enhancing safety profiles in commercial scale-up of complex pharmaceutical intermediates.
Impurity control is another critical aspect managed by this mechanistic pathway. In traditional borohydride reductions, over-reduction or non-selective reduction of other functional groups can occur. However, the transfer hydrogenation described here is highly chemoselective for the beta-keto ester functionality. The reaction conditions, typically ranging from room temperature to mild heating (e.g., 50°C in DMF), avoid the harsh thermal stresses that often generate degradation byproducts. Furthermore, because the chirality is installed catalytically rather than via stoichiometric chiral auxiliaries, there are no auxiliary-derived impurities to remove during purification. The resulting products, such as the (E)-(S)-hydroxy esters, exhibit high crystallinity, which serves as an additional purification step during isolation. This intrinsic ability to suppress side reactions and facilitate crystallization ensures that the final API intermediates meet the stringent purity specifications required by global regulatory bodies, reducing the burden on quality control labs.
How to Synthesize Pitavastatin Intermediates Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing the critical mevalonate side chain attached to various cyclic cores. The process begins with the preparation of a phosphonium ylide, which undergoes a Wittig reaction with a cyclic aldehyde (such as a quinoline-3-carbaldehyde) to establish the carbon backbone and the E-alkene geometry. Following this, the pivotal asymmetric transfer hydrogenation step installs the first chiral center at the C-5 position (relative to the ester). Subsequent steps involve an aldol condensation with tert-butyl acetate using strong bases like LDA at cryogenic temperatures (-78°C) to extend the chain, followed by a stereoselective reduction of the newly formed ketone to set the second chiral center at C-3. The detailed standardized synthesis steps see the guide below.
- Perform Wittig reaction between phosphonium salts and cyclic aldehydes in nitrile solvents at reflux to form alpha,beta-unsaturated keto-esters.
- Execute asymmetric transfer hydrogenation using chiral Ru(II) catalysts (e.g., Noyori type) with hydrogen donors like formic acid/triethylamine or 2-propanol.
- Conduct aldol condensation with tert-butyl acetate using LDA at -78°C, followed by stereoselective ketone reduction and final hydrolysis to the calcium or sodium salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the technology described in CN1636004A offers compelling strategic advantages beyond mere chemical elegance. The primary driver is the drastic simplification of the manufacturing workflow. By removing the resolution step, the number of unit operations is significantly reduced, which directly correlates to lower labor costs, reduced solvent consumption, and shorter cycle times. The elimination of the 50% material discard associated with racemic resolution means that the effective throughput of the manufacturing plant is doubled without increasing capital expenditure on new reactors. This efficiency gain allows for a more responsive supply chain capable of meeting sudden spikes in demand for statin APIs. Furthermore, the robustness of the catalyst system against moisture and heteroatom poisoning reduces the risk of batch failures due to minor deviations in raw material quality or environmental conditions, ensuring enhanced supply chain reliability.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the atom economy of the asymmetric step. Since the chiral center is created directly with high enantiomeric excess, there is no need to purchase expensive chiral resolving agents or manage the logistics of recycling the unwanted enantiomer. Additionally, the ability to use simpler hydrogen donors like formic acid or 2-propanol instead of high-pressure hydrogen gas lowers the safety infrastructure costs and insurance premiums associated with the production facility. The reduced solvent usage, stemming from fewer purification and crystallization steps, also contributes to significant operational expenditure savings, making the final intermediate more price-competitive in the global market.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of synthetic routes. This method mitigates risk by utilizing robust catalysts that are less sensitive to operational variances. The patent explicitly notes that the reaction can proceed in the presence of water and without inert gas, which relaxes the specifications for solvent drying and reactor purging. This tolerance means that production is less likely to be halted by minor equipment leaks or solvent quality fluctuations. Moreover, the starting materials, such as phosphonium salts and cyclic aldehydes, are commercially available or easily synthesized from commodity chemicals, reducing dependency on niche suppliers and minimizing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling chemical processes often reveals hidden bottlenecks, but this route is designed with scalability in mind. The exothermic nature of the reactions is manageable, and the absence of high-pressure hydrogenation steps simplifies the engineering requirements for large-scale reactors. From an environmental perspective, the process aligns with green chemistry principles by maximizing atom efficiency and minimizing waste generation. The reduction in solvent waste and the elimination of resolution byproducts lower the burden on wastewater treatment facilities. This environmental compliance is increasingly critical for maintaining operating licenses in regulated jurisdictions, ensuring long-term viability of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and claims within CN1636004A, providing clarity on the feasibility and advantages of the technology for potential partners and licensees.
Q: How does this process improve enantiomeric excess compared to traditional resolution?
A: The patent describes achieving an enantiomeric excess (ee) of ≥95%, preferably ≥98%, and most preferably ≥99% directly through asymmetric catalysis, eliminating the theoretical 50% yield loss associated with resolving racemic mixtures.
Q: Can the catalyst tolerate quinoline moieties found in Pitavastatin precursors?
A: Yes, surprisingly, the specific Ru(II) catalysts described (Formula IId) remain active and do not get deactivated by the quinoline nitrogen atoms, which typically poison hydrogenation catalysts in prior art methods.
Q: Is strict anhydrous condition required for the transfer hydrogenation step?
A: No, the invention demonstrates that the transfer hydrogenation can successfully proceed in aqueous solvent systems (e.g., containing up to 3% water) and without inert gas protection, simplifying operational requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pitavastatin Intermediate Supplier
The technological potential of the asymmetric transfer hydrogenation route for statin intermediates is immense, offering a clear path to higher purity and lower costs. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are successfully translated into industrial reality. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of Pitavastatin or Fluvastatin intermediate meets the highest international standards. We understand the critical nature of API supply chains and are committed to delivering consistency and quality.
We invite you to collaborate with us to leverage this advanced chemistry for your product portfolio. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain and reduce your overall production costs.
