Revolutionizing Chiral Amine Production: A Metal-Free Hydrogen Borrowing Strategy for Commercial Scale-Up
The landscape of chiral amine synthesis is undergoing a significant transformation driven by the urgent need for cleaner, more economically viable processes in the pharmaceutical industry. Patent CN109111380B, published in late 2020, introduces a groundbreaking methodology that challenges the long-standing dominance of transition metal catalysis in asymmetric amination reactions. This innovation leverages a metal-free hydrogen borrowing strategy, utilizing a catalytic amount of ketone and a readily available base to facilitate the asymmetric N-alkylation of chiral tert-butyl sulfinamide with 1-arylethanol substrates. For R&D directors and process chemists, this represents a paradigm shift away from complex, metal-dependent systems toward a more sustainable and operationally simple protocol. The ability to achieve high diastereoselectivity (dr up to 99:1) and respectable yields (41% to 82%) without the use of expensive Iridium, Ruthenium, or Rhodium complexes addresses two of the most persistent pain points in API manufacturing: cost control and impurity management. As we delve deeper into the technical specifics, it becomes clear that this technology offers a robust pathway for the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of optically pure chiral amines has relied heavily on transition metal-catalyzed hydrogen borrowing reactions or stoichiometric chiral resolution techniques. While effective, these conventional approaches suffer from inherent inefficiencies that complicate large-scale production. Traditional borrowing hydrogen methodologies typically require precious metal catalysts such as [Cp*IrCl2]2 or [Ru(p-cymene)Cl2]2, which not only drive up raw material costs but also introduce the risk of heavy metal contamination in the final product. Regulatory agencies impose stringent limits on residual metals in active pharmaceutical ingredients, necessitating additional downstream purification steps like metal scavenging, which adds time, waste, and expense to the manufacturing process. Furthermore, many existing protocols require harsh conditions or specialized ligands that are difficult to source reliably, creating supply chain vulnerabilities. The reliance on stoichiometric chiral resolving agents in alternative methods further exacerbates the issue, capping theoretical yields at 50% and generating substantial chemical waste, which contradicts the principles of green chemistry and atom economy that modern pharmaceutical manufacturing strives to uphold.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN109111380B offers a streamlined, metal-free alternative that fundamentally simplifies the reaction architecture. By employing a synergistic catalytic system composed of a simple ketone and an inorganic or organic base, this method activates the alcohol substrate for nucleophilic attack without the need for transition metals. The process operates effectively in common organic solvents like toluene at reflux temperatures, typically around 120°C, under an inert atmosphere. This simplicity translates directly into operational ease; the reaction is a true one-pot procedure where the chiral tert-butyl sulfinamide serves as both the nitrogen source and the chirality inducer. The elimination of transition metals means the resulting chiral amines are free from heavy metal residues, drastically reducing the burden on quality control laboratories and purification teams. Moreover, the system demonstrates remarkable versatility, accommodating a wide range of aryl and heteroaryl substituents while maintaining high stereocontrol, making it a highly attractive option for the diverse library synthesis required in early-stage drug discovery and process development.
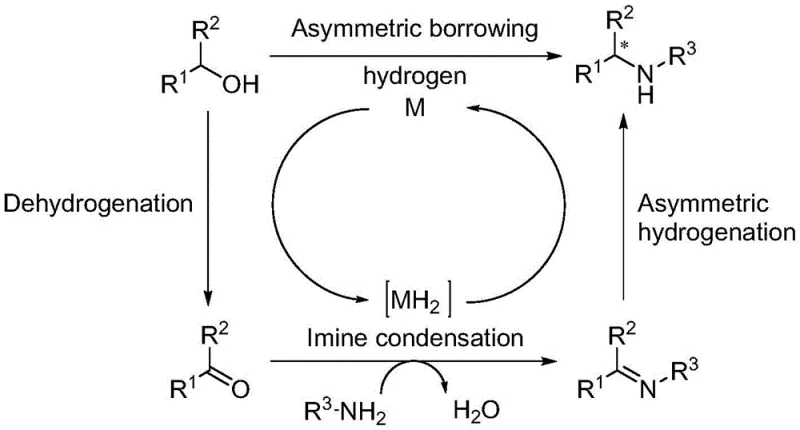
Mechanistic Insights into Metal-Free Hydrogen Borrowing Amination
The mechanistic elegance of this invention lies in its ability to mimic the efficiency of transition metal catalysis using simple organic molecules. In traditional borrowing hydrogen cycles, a metal center facilitates the dehydrogenation of the alcohol to a ketone, which then condenses with an amine to form an imine, followed by metal-mediated reduction back to the amine. In this metal-free variant, the catalytic ketone plays a pivotal role in shuttling hydrogen equivalents. The base activates the alcohol, promoting dehydrogenation to generate the reactive carbonyl intermediate in situ. This intermediate then undergoes condensation with the chiral sulfinamide to form a sulfinylimine. The stereochemical outcome is rigorously controlled by the chiral sulfur auxiliary present in the sulfinamide, which directs the subsequent reduction step. The hydrogen required for this reduction is borrowed from the initial dehydrogenation of the alcohol substrate, ensuring high atom economy where water is the primary byproduct. This closed-loop hydrogen transfer avoids the need for external reducing agents like borohydrides or silanes, which are often hazardous and generate stoichiometric waste. The result is a clean reaction profile that minimizes side products and simplifies the isolation of the target chiral amine, providing R&D teams with a predictable and robust synthetic tool.
From an impurity control perspective, this mechanism offers distinct advantages over metal-catalyzed routes. The absence of metal-ligand complexes eliminates the formation of metal-associated degradation products that can be notoriously difficult to separate from the API. Additionally, the use of tert-butyl sulfinamide as a chiral source allows for the easy removal of the auxiliary group post-reaction if a free amine is desired, or it can be retained as a protected chiral building block. The high diastereoselectivity observed (dr values frequently exceeding 98:2) indicates that the transition state is tightly organized, likely through specific hydrogen bonding or steric interactions facilitated by the base and the sulfinyl oxygen. This level of control ensures that the formation of unwanted enantiomers is minimized, reducing the need for costly recrystallization or chiral chromatography steps later in the process. For process chemists, understanding this mechanism validates the scalability of the reaction, as the kinetics are driven by stable organic species rather than sensitive metal centers that might deactivate over time or upon exposure to trace impurities.
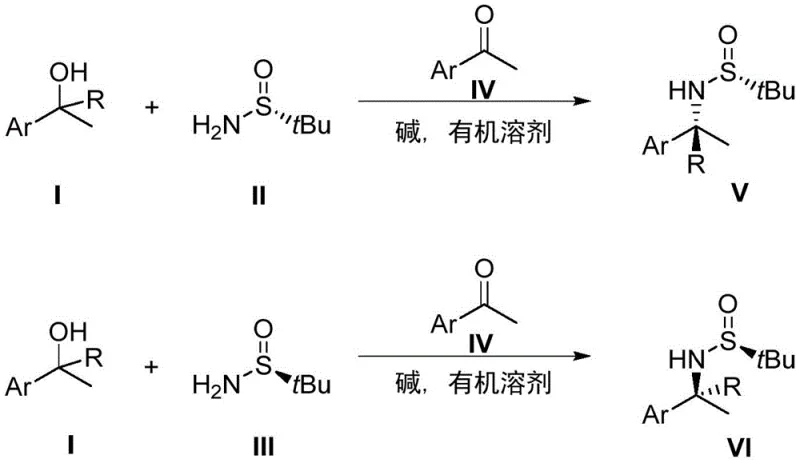
How to Synthesize Chiral Amines Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and stereoselectivity. The protocol is designed to be user-friendly, utilizing standard glassware and heating equipment without the need for specialized high-pressure reactors often associated with hydrogenation reactions. The key to success lies in the precise molar ratios of the reactants; the patent specifies that the 1-arylethanol substrate should be used in excess, typically 2 to 4 times the molar amount of the chiral sulfinamide, to drive the equilibrium towards product formation. The catalytic ketone is used in small amounts (0.1 to 0.2 equivalents), acting as a true turnover catalyst, while the base is employed in near-stoichiometric quantities relative to the sulfinamide to ensure complete activation of the alcohol. Reaction times can vary from 2 to 24 hours depending on the electronic nature of the substrate, with electron-deficient aromatics generally reacting faster. Workup is straightforward, involving simple filtration to remove the base followed by solvent evaporation, making it highly amenable to automation and parallel synthesis workflows.
- Prepare the reaction mixture by combining 1-arylethanol substrate, chiral tert-butyl sulfinamide, catalytic ketone, and base in an organic solvent under inert atmosphere.
- Heat the sealed reaction vessel to reflux temperatures (approximately 120°C) and maintain stirring for 2 to 24 hours to facilitate the asymmetric amination.
- Upon completion, filter off the base, remove the solvent under reduced pressure, and purify the crude product via column chromatography to isolate the high-purity chiral amine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free technology presents a compelling value proposition centered on cost stability and supply security. The primary economic benefit stems from the complete elimination of precious metal catalysts. Iridium and Ruthenium prices are subject to significant market volatility and geopolitical supply risks; by removing these materials from the bill of materials, manufacturers can achieve substantial cost savings and insulate their production budgets from external shocks. Furthermore, the simplified downstream processing—specifically the removal of metal scavenging steps—reduces the consumption of auxiliary materials and shortens the overall cycle time per batch. This efficiency gain translates directly into increased throughput capacity without the need for capital investment in new equipment. The use of commodity chemicals like acetophenone derivatives and sodium hydroxide as catalysts ensures that raw materials are readily available from multiple global suppliers, mitigating the risk of single-source dependency that often plagues specialized ligand supply chains.
- Cost Reduction in Manufacturing: The economic impact of switching to this organocatalytic system is profound, primarily driven by the reduction in raw material expenses and waste disposal costs. Precious metal catalysts are not only expensive to purchase but also expensive to recover or dispose of in compliance with environmental regulations. By utilizing inexpensive ketones and bases, the direct material cost per kilogram of product is drastically lowered. Additionally, the high atom economy of the borrowing hydrogen mechanism means less waste is generated per unit of product, reducing the financial burden associated with solvent recovery and hazardous waste treatment. The simplified purification process also lowers labor and utility costs, as fewer unit operations are required to bring the material to specification. These cumulative savings contribute to a significantly improved gross margin profile for the manufactured intermediate, enhancing the overall competitiveness of the final drug product in the marketplace.
- Enhanced Supply Chain Reliability: Supply chain resilience is a critical metric for pharmaceutical manufacturers, and this technology strengthens it by relying on widely available feedstocks. Unlike complex chiral ligands that may have long lead times or limited manufacturing capacity, the reagents used in this process are bulk commodities produced at scale globally. This abundance ensures consistent availability and stable pricing, allowing for more accurate long-term forecasting and inventory planning. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in reagent quality, reducing the frequency of batch failures and the need for rigorous incoming quality testing. For supply chain planners, this reliability minimizes the need for safety stock and allows for leaner inventory models, freeing up working capital and improving cash flow efficiency across the organization.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to commercial production often reveals hidden bottlenecks, particularly regarding heat transfer and safety. This metal-free amination method is inherently safer as it avoids the use of pyrophoric reagents or high-pressure hydrogen gas, which require specialized infrastructure and safety protocols. The reaction proceeds at atmospheric pressure in standard solvents, making it easily transferable to existing multipurpose reactors without modification. From an environmental standpoint, the process aligns perfectly with green chemistry initiatives by eliminating heavy metals and minimizing waste generation. This compliance simplifies the regulatory filing process and reduces the environmental footprint of the manufacturing site. The ability to scale safely and sustainably ensures that supply can meet demand as the drug candidate progresses through clinical trials to commercial launch, preventing supply disruptions that could delay patient access to critical therapies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into their existing manufacturing portfolios. The answers reflect the practical realities of running metal-free borrowing hydrogen reactions at scale.
Q: Why is eliminating transition metals critical for pharmaceutical intermediate synthesis?
A: Transition metal residues such as Iridium or Ruthenium are strictly regulated in final drug substances due to toxicity concerns. Removing them requires expensive scavenging steps. This metal-free method eliminates that burden entirely.
Q: What represents the primary cost driver in traditional chiral amine synthesis?
A: Traditional methods often rely on precious metal catalysts and stoichiometric chiral reagents. This new approach uses catalytic amounts of inexpensive ketones and cheap bases, significantly lowering raw material costs.
Q: Can this methodology be applied to deuterated drug candidates?
A: Yes, the patent explicitly demonstrates high deuterium incorporation rates (up to 96%) when using deuterated alcohol substrates, making it highly suitable for metabolic stability studies in drug development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Amine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of metal-free catalytic technologies in modern pharmaceutical synthesis. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN109111380B can be successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped to handle sensitive organocatalytic reactions with stringent purity specifications, supported by rigorous QC labs that utilize advanced analytical techniques to verify stereochemical integrity and impurity profiles. We understand that the transition to new synthetic routes requires confidence in execution, and our team of expert process chemists is dedicated to optimizing reaction parameters to maximize yield and minimize cost for our clients. Whether you require custom synthesis of chiral intermediates or full-scale commercial manufacturing, our commitment to quality and reliability remains unwavering.
We invite you to explore how this metal-free amination technology can enhance your drug development pipeline and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs, demonstrating the tangible economic benefits of adopting this greener synthetic route. We encourage you to contact us to request specific COA data for similar chiral amine structures and to discuss route feasibility assessments for your target molecules. By partnering with NINGBO INNO PHARMCHEM, you gain access to a supply chain that prioritizes innovation, sustainability, and operational excellence, ensuring that your critical pharmaceutical intermediates are delivered on time and to the highest standards of quality.
