Advanced Catalytic Transfer Hydrogenation for High-Purity Chiral Amines
Introduction to Advanced Asymmetric Synthesis Technologies
The landscape of fine chemical manufacturing is undergoing a significant transformation driven by the demand for safer, more efficient, and highly stereoselective synthetic routes. Patent CN1328230C introduces a groundbreaking catalytic transfer hydrogenation process that addresses critical challenges in the production of optically active compounds, specifically targeting the synthesis of primary and secondary amines. This technology leverages sophisticated transition metal complexes to facilitate the reduction of N-substituted imines and iminium salts without the logistical and safety burdens associated with high-pressure gaseous hydrogen. By utilizing a defined system of metal ligands and hydrogen donors, this method offers a robust pathway for generating high-purity pharmaceutical intermediates with exceptional enantiomeric control. The versatility of this approach allows for the processing of a wide array of substrates, making it a cornerstone technology for modern process chemistry.
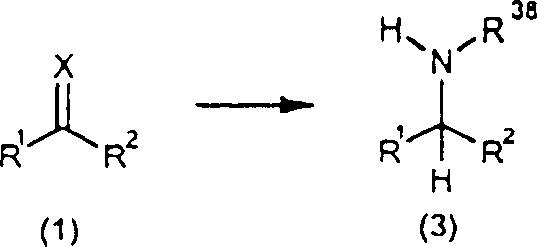
For procurement specialists and supply chain managers, the implications of adopting such a technology are profound. It represents a shift towards processes that are not only chemically elegant but also commercially viable on a large scale. The ability to produce complex chiral amines under mild conditions reduces the dependency on specialized high-pressure infrastructure, thereby lowering the barrier to entry for manufacturing these high-value ingredients. As a reliable pharmaceutical intermediates supplier, understanding the nuances of such patented methodologies is essential for ensuring a continuous and compliant supply of critical building blocks for the global healthcare industry. This report delves deep into the technical specifications and commercial advantages of this transfer hydrogenation methodology.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for the synthesis of chiral amines have long relied on direct hydrogenation using molecular hydrogen gas, a process that necessitates the use of high-pressure reactors and stringent safety protocols. These conventional approaches often require expensive noble metal catalysts that may lack sufficient stereoselectivity, leading to difficult and costly downstream purification steps to remove unwanted enantiomers. Furthermore, the handling of gaseous hydrogen poses inherent safety risks, including flammability and explosion hazards, which complicate facility design and increase insurance and compliance costs. In many cases, the reaction conditions are harsh, potentially degrading sensitive functional groups present in complex pharmaceutical intermediates. The need for specialized equipment and the potential for lower yields due to side reactions make these traditional routes less attractive for cost-sensitive commercial manufacturing environments.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a catalytic transfer hydrogenation mechanism that operates effectively under atmospheric pressure and moderate temperatures ranging from -20°C to +110°C. This method employs a liquid hydrogen donor system, typically a mixture of formic acid and triethylamine, which serves as a safe and controllable source of hydride equivalents. The use of well-defined chiral catalysts, such as rhodium or ruthenium complexes with bidentate ligands, ensures high levels of enantioselectivity, often exceeding 99% ee in optimized examples. This eliminates the need for high-pressure infrastructure and significantly mitigates safety risks associated with gaseous hydrogen. The mild reaction conditions preserve the integrity of sensitive substrates, allowing for the synthesis of complex molecules that would otherwise be unstable under traditional hydrogenation conditions, thus offering a superior alternative for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Asymmetric Transfer Hydrogenation
The core of this innovative process lies in the precise coordination chemistry of the catalyst system. The patent specifies the use of Group VIII metals, particularly Ruthenium, Rhodium, and Iridium, which are coordinated with specific bidentate ligands such as diamines and aminoalcohols containing chiral centers. These ligands, often derived from structures like N-tosyl-1,2-diphenylethylenediamine, create a chiral environment around the metal center that dictates the stereochemical outcome of the hydride transfer. The mechanism involves the formation of a metal-hydride species through the dehydrogenation of the hydrogen donor, followed by the concerted transfer of the hydride and a proton to the imine substrate. This outer-sphere mechanism avoids the direct coordination of the substrate to the metal in a way that could lead to racemization, ensuring high fidelity in the creation of the chiral center.
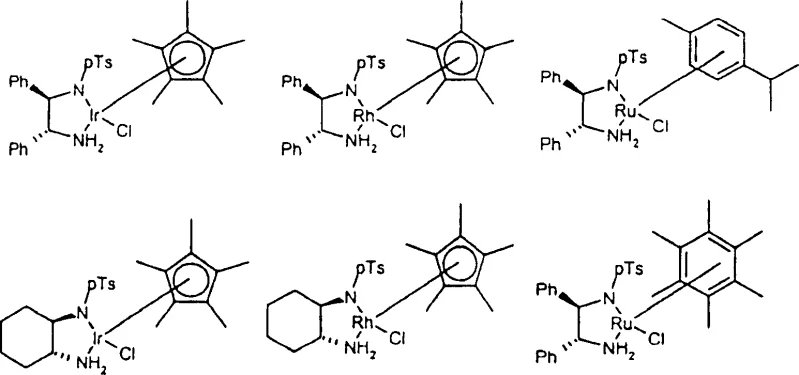
Furthermore, the impurity profile of the final product is tightly controlled by the stability of the catalyst and the specificity of the hydrogen donor. The use of N-phosphinyl or N-sulfonyl protecting groups on the imine nitrogen enhances the electrophilicity of the substrate, facilitating smoother reduction while preventing side reactions such as over-reduction or polymerization. The patent describes how the electronic properties of the ligands can be tuned by varying substituents on the aromatic rings or the phosphorus atoms, allowing chemists to optimize the reaction rate and selectivity for specific substrates. This level of mechanistic control is crucial for R&D directors who require consistent quality and reproducibility in the production of active pharmaceutical ingredients. The ability to fine-tune the catalyst system ensures that even challenging prochiral substrates can be converted into their corresponding amines with minimal formation of diastereomeric impurities.
How to Synthesize Chiral Amines Efficiently
The synthesis of these high-value chiral amines follows a logical and scalable sequence that begins with the preparation of the imine substrate from readily available carbonyl precursors. The process involves the condensation of ketones or aldehydes with protected amines, such as O-phosphinyl hydroxylamines, to form the N-substituted imine intermediate. Once the substrate is prepared, it is subjected to the transfer hydrogenation conditions using the pre-formed or in-situ generated metal catalyst. The reaction is typically conducted in polar solvents like methanol or acetonitrile, with the hydrogen donor added in molar excess to drive the equilibrium towards completion. Detailed standardized synthesis steps see the guide below.
- Preparation of N-substituted imine or iminium salt substrate from carbonyl precursors using phosphinyl or sulfonyl protecting groups.
- Assembly of the catalytic system using Group VIII metals (Rh, Ru, Ir) coordinated with chiral bidentate ligands such as N-tosyl diamines.
- Execution of the transfer hydrogenation reaction using a formic acid-triethylamine mixture as the hydrogen donor under mild thermal conditions.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this transfer hydrogenation technology offers substantial benefits that extend beyond mere chemical efficiency. For procurement managers, the elimination of high-pressure hydrogen gas simplifies the sourcing of raw materials and reduces the regulatory burden associated with storing hazardous gases. The catalysts used, while based on precious metals, are employed in low loadings due to their high turnover numbers, which helps in managing the overall cost of goods sold. The process operates under mild conditions, which translates to lower energy consumption for heating and cooling compared to traditional high-temperature and high-pressure processes. This energy efficiency contributes directly to a reduced carbon footprint, aligning with modern sustainability goals and environmental compliance standards required by major multinational corporations.
- Cost Reduction in Manufacturing: The transition to transfer hydrogenation removes the necessity for capital-intensive high-pressure reactors and the associated safety systems, leading to significant savings in facility maintenance and operation. By avoiding the use of gaseous hydrogen, companies can also reduce insurance premiums and safety training costs. The high selectivity of the catalyst minimizes waste generation and reduces the need for extensive purification steps like chromatography, which are often the most expensive part of fine chemical production. Additionally, the ability to use simple hydrogen donors like formic acid and triethylamine, which are commodity chemicals, ensures a stable and cost-effective supply chain for reagents.
- Enhanced Supply Chain Reliability: The reliance on liquid reagents rather than compressed gases enhances the reliability of the supply chain by removing dependencies on specialized gas delivery logistics. The robustness of the catalyst system allows for longer campaign runs without frequent catalyst replacement, ensuring consistent production schedules. The mild reaction conditions also mean that the process is less susceptible to disruptions caused by utility fluctuations, such as steam pressure drops, which can halt high-pressure operations. This stability is critical for meeting the just-in-time delivery requirements of downstream pharmaceutical manufacturers who depend on a steady flow of high-quality intermediates.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the absence of mass transfer limitations often encountered in gas-liquid hydrogenations. The liquid-phase nature of the reaction facilitates easier heat management and mixing in large-scale reactors. Furthermore, the byproducts of the hydrogen donor decomposition are typically benign, such as carbon dioxide and water, which simplifies waste treatment and disposal. This aligns with green chemistry principles and helps manufacturers meet increasingly stringent environmental regulations regarding volatile organic compound emissions and hazardous waste generation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this transfer hydrogenation technology. These answers are derived directly from the experimental data and claims presented in the patent documentation to provide clarity on the process capabilities. Understanding these details is vital for stakeholders evaluating the feasibility of integrating this method into their existing manufacturing workflows. The responses cover aspects ranging from catalyst selection to substrate compatibility and product isolation.
Q: What are the primary advantages of transfer hydrogenation over traditional high-pressure hydrogenation?
A: Transfer hydrogenation eliminates the need for gaseous hydrogen and high-pressure reactors, significantly enhancing operational safety and reducing capital expenditure on specialized equipment while maintaining high enantioselectivity.
Q: Which metal catalysts are most effective for this specific asymmetric reduction process?
A: The patent highlights Ruthenium(II), Rhodium(I/III), and Iridium(I/III) complexes, particularly when coordinated with chiral diamine or aminoalcohol ligands like N-tosyl-1,2-diphenylethylenediamine, as providing superior stereocontrol.
Q: How is the optical purity of the final amine product ensured during synthesis?
A: High enantiomeric excess (ee) is achieved through the use of enantiomerically purified chiral ligands on the metal center, which direct the hydride transfer to specific faces of the prochiral imine substrate, often yielding >99% ee.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Amines Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of advanced synthetic methodologies in delivering high-quality pharmaceutical intermediates to the global market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of technologies like transfer hydrogenation are fully realized in practical manufacturing settings. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of chiral amine meets the exacting standards required for drug substance synthesis. Our commitment to technical excellence allows us to navigate the complexities of asymmetric synthesis with precision and reliability.
We invite potential partners to engage with our technical procurement team to discuss how this innovative hydrogenation process can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this safer and more efficient route. We encourage you to contact us to obtain specific COA data and route feasibility assessments that demonstrate our capability to deliver superior value. Let us collaborate to optimize your supply chain and accelerate the development of your next-generation therapeutic agents.
