Advanced Synthesis of Pemetrexed Disodium Intermediates for Commercial API Production
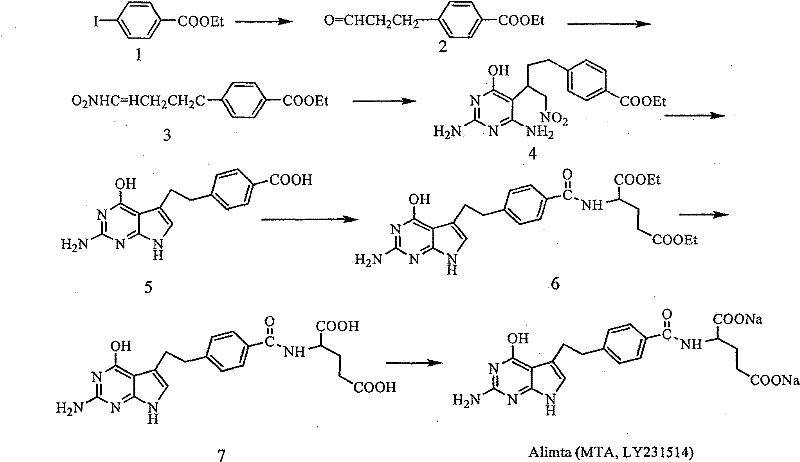
The pharmaceutical landscape for oncology treatments continues to evolve, with Pemetrexed Disodium (Alimta) remaining a cornerstone therapy for non-small cell lung cancer and mesothelioma. As demand for high-purity active pharmaceutical ingredients (APIs) intensifies, the efficiency of synthetic routes for key intermediates becomes a critical bottleneck for global supply chains. Patent CN1800169B introduces a transformative approach to synthesizing the critical intermediate 1-nitro-2-(2,6-diamino-4-(3H)oxopyrimidin-5-yl)-4-(4-carboxyphenyl)-1-butane. This innovation addresses longstanding inefficiencies in prior art by streamlining the construction of the pyrrolo[2,3-d]pyrimidine core. By shifting from complex ester-protected pathways to a direct Michael addition strategy using free acid precursors, this technology offers a robust foundation for cost reduction in pharmaceutical intermediates manufacturing. For R&D directors and procurement leaders, understanding this mechanistic shift is essential for securing a reliable pemetrexed disodium supplier capable of meeting rigorous quality and volume demands.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Pemetrexed intermediates has been plagued by multi-step sequences that introduce significant operational complexity and yield losses. Conventional routes, such as those described in US Patent 5,344,932 and improved by Taylor (US 6,066,732), rely heavily on the use of ester protecting groups, specifically ethyl or methyl esters, to mask the carboxylic acid functionality during the coupling and cyclization stages. While chemically sound, this protection-deprotection strategy necessitates additional unit operations, including esterification and subsequent hydrolysis, which consume valuable reagents and generate substantial waste streams. Furthermore, alternative routes explored by major pharmaceutical entities, such as the Heck coupling method utilizing methyl p-bromobenzoate, have demonstrated suboptimal performance with total yields to key intermediates hovering around 26.5%. These low yields, combined with the requirement for expensive palladium catalysts and rigorous purification to remove heavy metal residues, create significant barriers to efficient commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
The methodology disclosed in CN1800169B represents a paradigm shift by eliminating the need for ester protection entirely during the critical bond-forming steps. Instead of starting with esterified benzoic acid derivatives, this novel approach utilizes 1-nitro-4-(4-carboxyphenyl)-1-butene directly. This substrate undergoes a Michael addition with 2,6-diamino-4-(3H)oxopyrimidine under controlled alkaline conditions to form the key nitro-intermediate. By bypassing the esterification and hydrolysis cycle, the process drastically reduces the number of reaction vessels required and shortens the overall production timeline. The direct use of the free acid not only simplifies the workflow but also enhances the atom economy of the synthesis. This streamlined pathway allows manufacturers to achieve higher throughput with reduced solvent consumption, directly addressing the need for reducing lead time for high-purity pharmaceutical intermediates while maintaining strict environmental compliance standards.
Mechanistic Insights into Alkaline-Mediated Michael Addition
The core of this technological advancement lies in the precise control of the Michael addition reaction under homogeneous alkaline conditions. The reaction between the nitro-alkene and the diaminopyrimidine is facilitated by maintaining a pH between 8 and 14, typically using alkali metal or alkaline earth metal hydroxides. This specific pH range is critical for deprotonating the active methylene species without causing degradation of the sensitive pyrimidine ring or the nitro group. The reaction proceeds efficiently at moderate temperatures ranging from 30°C to 60°C, which minimizes thermal stress on the reactants and prevents the formation of polymeric byproducts often seen in harsher conditions. The homogeneous nature of the reaction mixture ensures uniform heat transfer and mass transfer, which is vital for maintaining consistent reaction kinetics across large batch sizes. This mechanistic precision results in a crude product with a significantly cleaner impurity profile, reducing the burden on downstream crystallization and purification units.
Following the formation of the nitro-intermediate, the subsequent cyclization to form the pyrrolo[2,3-d]pyrimidine core is achieved through a carefully orchestrated acid-base treatment sequence. The process involves cooling the reaction mixture and adjusting the pH to acidic levels (pH 0.5-6) using protonic acids such as sulfuric or hydrochloric acid, followed by a return to alkaline conditions. This pH swing facilitates the intramolecular condensation required to close the five-membered pyrrole ring. The final precipitation is induced by the addition of acetic acid, which allows for the isolation of the benzoic acid derivative in high purity. This controlled cyclization mechanism avoids the use of aggressive dehydrating agents or high-temperature reflux conditions typical of older methods, thereby preserving the structural integrity of the molecule and ensuring that the final API meets stringent pharmacopeial specifications for related substances.
How to Synthesize Pemetrexed Key Intermediate Efficiently
The implementation of this synthesis route requires careful attention to stoichiometry and pH control to maximize the yield of the nitro-intermediate. The process begins with the dissolution of the nitro-alkene and diaminopyrimidine in an aqueous or aqueous-ethanol solvent system. The standardized protocol emphasizes the gradual addition of the base to maintain the target pH window, preventing local exotherms that could degrade the product. Once the Michael addition is complete, indicated by the disappearance of starting materials, the product is isolated via acidification.
- React 1-nitro-4-(4-carboxyphenyl)-1-butene with 2,6-diamino-4-(3H)oxopyrimidine under alkaline conditions at pH 8-14.
- Maintain reaction temperature between 30°C and 60°C until raw materials are consumed.
- Precipitate the product by adjusting pH to 5-6 with acetic acid and filter to obtain the key intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers tangible strategic benefits beyond mere chemical elegance. The elimination of esterification and hydrolysis steps translates directly into a reduction in raw material costs, as expensive alcohols and hydrolysis catalysts are no longer required. Furthermore, the reduction in unit operations decreases the consumption of utilities such as steam for heating and chilled water for cooling, leading to substantial cost savings in manufacturing overhead. The use of water and ethanol as primary solvents aligns with green chemistry principles, simplifying waste treatment protocols and reducing the environmental footprint associated with volatile organic compound (VOC) emissions. These factors collectively enhance the economic viability of producing high-purity pharmaceutical intermediates at a commercial scale.
- Cost Reduction in Manufacturing: The streamlined process eliminates multiple reaction steps associated with protecting group chemistry, specifically the installation and removal of ethyl or methyl esters. This reduction in step count lowers the consumption of reagents, solvents, and labor hours per kilogram of finished product. Additionally, the higher yields reported for the key cyclization step (improving from roughly 57% in prior art to 75% in this method) mean that less starting material is wasted, directly improving the cost of goods sold (COGS) and allowing for more competitive pricing structures in the global market.
- Enhanced Supply Chain Reliability: By utilizing readily available starting materials such as 1-nitro-4-(4-carboxyphenyl)-1-butene, the supply chain becomes less vulnerable to bottlenecks associated with specialized protected intermediates. The robustness of the reaction conditions, which tolerate a broader range of operational parameters compared to sensitive palladium-catalyzed couplings, ensures consistent batch-to-batch quality. This reliability is crucial for maintaining continuous API production schedules and mitigating the risk of stockouts that can disrupt downstream drug formulation and distribution networks.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on aqueous media and moderate temperatures, which are easier to manage in large-scale reactors than anhydrous or cryogenic conditions. The avoidance of heavy metal catalysts removes the need for complex and costly metal scavenging steps, simplifying the purification train. Moreover, the reduced generation of organic waste and the use of benign solvents facilitate easier compliance with increasingly stringent environmental regulations, ensuring long-term operational sustainability for manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical advantages of this route over traditional methodologies.
Q: What is the primary advantage of the new synthesis route for Pemetrexed intermediates?
A: The new route utilizes a direct Michael addition with a free carboxylic acid precursor, eliminating the need for esterification and subsequent hydrolysis steps found in conventional methods, thereby improving overall yield and reducing processing time.
Q: How does this method improve impurity control compared to prior art?
A: By avoiding harsh esterification conditions and utilizing a homogeneous alkaline reaction system, the process minimizes side reactions and degradation, resulting in a cleaner crude product profile that simplifies downstream purification.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the process operates at mild temperatures (30-60°C) using water or water-ethanol mixtures as solvents, which are cost-effective and environmentally favorable, making it highly scalable for industrial API manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pemetrexed Disodium Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to industrial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of advanced synthesis routes like CN1800169B are fully realized in practice. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of Pemetrexed intermediate meets the highest global standards. Our commitment to quality assurance ensures that our clients receive materials that are ready for immediate API synthesis without the need for extensive rework or purification.
We invite pharmaceutical companies and contract manufacturing organizations to collaborate with us on optimizing their supply chains for oncology therapeutics. By leveraging our technical proficiency in heterocyclic chemistry and process optimization, we can help you achieve significant efficiencies in your production workflows. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your long-term strategic goals in the competitive landscape of generic and branded drug production.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
