Advanced Manufacturing of Pemetrexed Disodium: A Novel Low-Temperature Crystallization Strategy for Global Supply Chains
Advanced Manufacturing of Pemetrexed Disodium: A Novel Low-Temperature Crystallization Strategy for Global Supply Chains
The pharmaceutical landscape for oncology treatments continues to evolve, driven by the demand for higher purity active pharmaceutical ingredients (APIs) and more robust manufacturing processes. Patent CN102911176A introduces a significant technological breakthrough in the preparation of Pemetrexed Disodium, a critical thymidylate synthase inhibitor used in the treatment of non-small cell lung cancer and mesothelioma. This patent details a novel synthetic route that fundamentally alters the purification paradigm, shifting away from energy-intensive, high-temperature crystallization methods toward a sophisticated room-temperature protocol utilizing an ethanol and dimethyl sulfoxide (DMSO) mixed solvent system. For global supply chain leaders and R&D directors, this innovation represents a pivotal opportunity to enhance product stability while simultaneously addressing the persistent challenges of oxidative degradation that have plagued conventional manufacturing techniques. By integrating this advanced methodology, manufacturers can achieve superior impurity profiles and ensure the consistent delivery of high-purity pemetrexed disodium required for stringent regulatory compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Pemetrexed Disodium has been fraught with technical inefficiencies that compromise both yield and product quality. As illustrated in the prior art reaction schemes, traditional pathways often involve complex multi-step sequences culminating in purification processes that rely heavily on thermal energy. Specifically, methods disclosed in patents such as WO0114379 and CN1778797 necessitate heating crystallization steps at temperatures ranging from 45°C to 70°C using mixed solvents like ethanol/water or isopropanol/water. This thermal dependency introduces a critical vulnerability: the Pemetrexed molecule is inherently susceptible to oxidation, and exposing the crude product to elevated temperatures during the critical crystallization phase accelerates oxidative degradation, leading to the formation of undesirable impurities that are difficult to remove. Furthermore, alternative purification strategies involving acetone solvents, as seen in CN1406238, pose significant risks regarding residual solvent limits, while aqueous salting-out methods described in CN101417998 often fail to effectively remove non-polar organic impurities, resulting in a final product that may struggle to meet the rigorous purity specifications demanded by international pharmacopoeias.
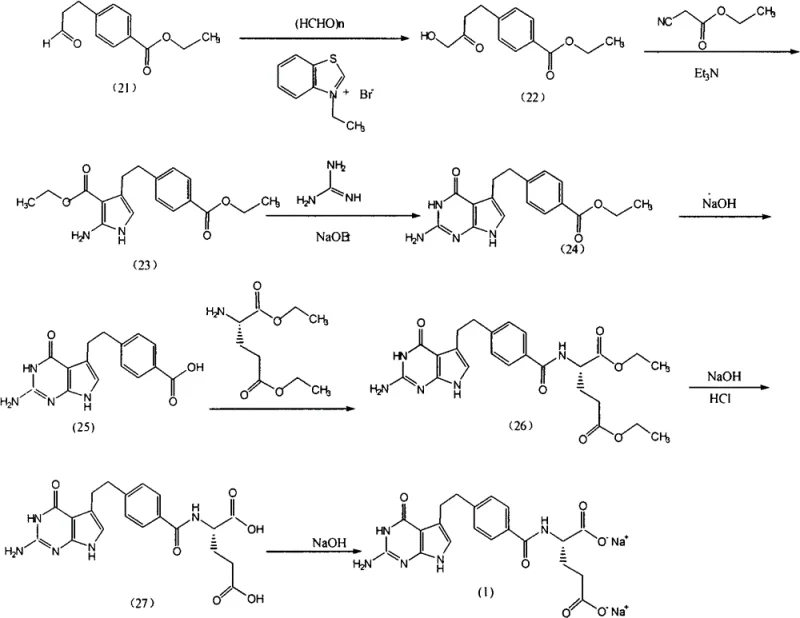
The Novel Approach
In stark contrast to these legacy methods, the technology outlined in CN102911176A presents a streamlined and chemically elegant solution that prioritizes molecular integrity throughout the manufacturing process. The core innovation lies in the strategic application of a specific ethanol and DMSO mixed solvent system, optimized at a volume ratio of approximately 10:1.5 to 10:2.5, which facilitates effective crystallization at ambient or even sub-ambient temperatures (-6°C to -8°C). This approach effectively decouples the purification process from thermal stress, thereby preserving the delicate pyrrolo-pyrimidine structure of the API and preventing the onset of oxidative side reactions. The reaction pathway begins with the hydrolysis of the methyl ester precursor to the free acid, followed by a highly efficient condensation with L-glutamic acid diethyl ester to form a stable tosylate intermediate, before final saponification yields the target disodium salt. By operating under these mild conditions, the process not only safeguards the chemical stability of the product but also simplifies the downstream processing requirements, offering a distinct advantage for the commercial scale-up of complex oncology intermediates where consistency and purity are paramount.
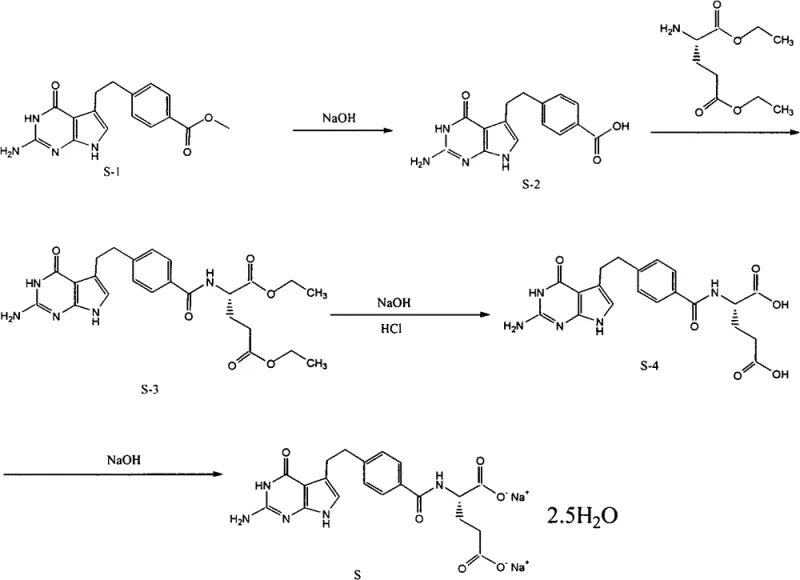
Mechanistic Insights into Peptide Coupling and Crystallization Dynamics
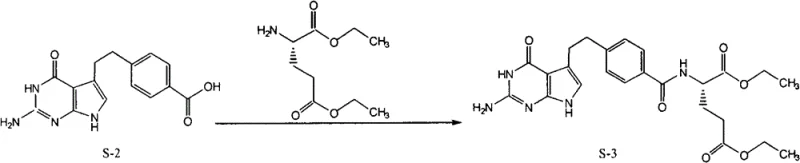
The success of this novel synthesis route hinges on the precise control of reaction kinetics during the coupling and crystallization phases, particularly in the formation of the amide bond between the benzoic acid derivative and the glutamic acid moiety. The patent specifies the use of 6-chloro-2,4-dimethoxy-1,3,5-triazine (CDMT) in conjunction with N-methylmorpholine (NMM) as the activating system for the carboxylic acid intermediate. This activation strategy generates a highly reactive O-acylisourea or triazine-mediated intermediate in situ, which subsequently undergoes nucleophilic attack by the amino group of the L-glutamic acid diethyl ester. The choice of CDMT is particularly astute for this application, as it minimizes the risk of racemization at the chiral center of the glutamic acid residue, a common pitfall in peptide synthesis that can lead to the formation of diastereomeric impurities. By maintaining the reaction temperature below 5°C during the activation phase and allowing it to warm to room temperature for the coupling, the process ensures high stereochemical fidelity, which is essential for the biological activity of the final anticancer agent. This mechanistic precision ensures that the resulting intermediate (S-3) possesses the necessary structural integrity to withstand the subsequent saponification steps without compromising the enantiomeric excess.
Beyond the coupling chemistry, the crystallization mechanism employed in the final purification step offers profound insights into impurity control and lattice formation. The utilization of DMSO as a co-solvent with ethanol modifies the solubility parameters of the reaction medium, creating a environment where the target Pemetrexed Disodium molecule has reduced solubility while many organic impurities remain solvated. Unlike traditional aqueous systems where the high polarity of water can sometimes trap organic contaminants within the crystal lattice or fail to dissolve them adequately for removal, the ethanol/DMSO mixture provides a balanced polarity that favors the exclusion of non-polar byproducts. Furthermore, the absence of heating during this phase prevents the kinetic energy that typically drives the incorporation of impurities into the growing crystal structure. The result is a crystalline product with a highly ordered lattice structure, evidenced by the reported HPLC purity levels exceeding 99.3% in the experimental examples. This level of purity is achieved not through repetitive recrystallizations, which degrade yield, but through a single, highly optimized crystallization event that leverages thermodynamic stability at low temperatures to naturally reject impurities.
How to Synthesize Pemetrexed Disodium Efficiently
The implementation of this synthesis protocol requires strict adherence to the specified reaction conditions and solvent ratios to maximize yield and purity. The process is designed to be robust, utilizing readily available reagents such as sodium hydroxide, tosic acid, and standard organic solvents, which facilitates easy sourcing and inventory management for large-scale operations. The initial hydrolysis step converts the methyl ester to the free acid under basic conditions, followed by a careful pH adjustment to precipitate the intermediate. Subsequent coupling with the glutamic acid derivative forms the key carbon-nitrogen bond, and the final saponification reveals the free acid groups necessary for salt formation. The critical final step involves dissolving the crude disodium salt in water, filtering out insolubles, and then inducing crystallization by adding the ethanol/DMSO mixture at controlled temperatures. For a detailed breakdown of the specific reagent quantities, reaction times, and workup procedures, please refer to the standardized synthesis guide below.
- Hydrolyze 4-[2-(2-amino-4,7-dihydro-4-oxo-3H-pyrrolo[2,3-d]pyrimidin-5-yl)ethyl] methyl benzoate (S-1) using sodium hydroxide to generate the free acid intermediate (S-2).
- Condense the acid (S-2) with L-glutamic acid diethyl ester using CDMT and N-methylmorpholine to form the diethyl ester tosylate salt (S-3).
- Perform saponification on (S-3) followed by salt formation and purification using an ethanol/DMSO mixed solvent system at room temperature to obtain the final disodium salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel manufacturing process translates directly into tangible operational efficiencies and risk mitigation strategies. The shift away from high-temperature processing and complex solvent systems addresses several pain points associated with the traditional production of Pemetrexed Disodium. By eliminating the need for prolonged heating cycles during crystallization, the process significantly reduces energy consumption, which is a major component of manufacturing overhead in chemical production. Furthermore, the simplified purification workflow reduces the number of unit operations required, thereby shortening the overall production cycle time and increasing the throughput capacity of existing manufacturing facilities. This efficiency gain is crucial for meeting the growing global demand for oncology treatments without necessitating massive capital expenditures on new infrastructure. Additionally, the use of common solvents like ethanol and DMSO, rather than specialized or hazardous reagents, streamlines the supply chain logistics and reduces the regulatory burden associated with solvent handling and disposal.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived primarily from the elimination of energy-intensive heating steps and the reduction in solvent usage associated with multiple recrystallizations. Traditional methods often require significant thermal input to drive crystallization, which not only increases utility costs but also places greater stress on reactor equipment, leading to higher maintenance expenses. By operating at room temperature or mild cooling conditions, this novel method drastically lowers the thermal load on the production system. Moreover, the high purity achieved in a single crystallization step minimizes the need for reprocessing off-spec batches, thereby reducing material waste and maximizing the yield of saleable product per batch. This lean manufacturing approach ensures that cost reduction in API manufacturing is achieved through process optimization rather than compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the variability inherent in complex chemical syntheses, particularly when sensitive intermediates are involved. The robustness of this new method, characterized by its tolerance to ambient conditions and use of stable reagents, significantly enhances the predictability of production schedules. The avoidance of high-temperature oxidation means that batch-to-batch variability due to degradation products is minimized, leading to a more consistent supply of high-quality material. This reliability is essential for pharmaceutical companies that must adhere to strict Just-In-Time delivery schedules to support clinical trials and commercial drug launches. By partnering with a reliable pemetrexed disodium supplier who utilizes this advanced technology, buyers can mitigate the risk of supply disruptions caused by failed batches or extended purification timelines.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this process offers distinct advantages over conventional routes. The reduction in solvent volume and the use of less hazardous solvent combinations contribute to a smaller environmental footprint, aligning with the increasing industry focus on green chemistry principles. The simplicity of the workup procedures, involving standard filtration and washing steps, makes the process highly amenable to scaling from pilot plant quantities to multi-ton commercial production without encountering the mixing or heat transfer limitations often seen in more complex protocols. This scalability ensures that the supply of high-purity pemetrexed disodium can be rapidly expanded to meet market surges. Furthermore, the improved impurity profile reduces the burden on wastewater treatment systems, as fewer toxic byproducts are generated, facilitating easier compliance with increasingly stringent environmental regulations governing pharmaceutical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this novel Pemetrexed Disodium synthesis method. These insights are derived directly from the technical specifications and experimental data provided in the patent literature, offering a transparent view into the capabilities of this manufacturing platform. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this supply source into their existing procurement networks.
Q: Why is the ethanol/DMSO solvent system preferred for Pemetrexed Disodium purification?
A: The ethanol/DMSO mixed solvent system allows for effective crystallization at room temperature or low temperatures (-6°C to -8°C), which prevents the thermal oxidation of the sensitive pyrrolo-pyrimidine core that occurs in traditional high-temperature aqueous or acetone-based processes.
Q: How does this method improve the impurity profile compared to conventional routes?
A: By avoiding high-temperature heating steps (60-70°C) during crystallization, this method significantly reduces the formation of oxidative degradation products. Additionally, the specific solvent polarity helps remove organic impurities that are difficult to eliminate in purely aqueous salting-out processes.
Q: Is this synthesis route scalable for commercial production?
A: Yes, the process utilizes standard reagents like sodium hydroxide and tosic acid and operates under mild conditions (room temperature stirring), making it highly suitable for commercial scale-up of complex oncology intermediates without requiring specialized high-pressure or cryogenic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pemetrexed Disodium Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced manufacturing processes is key to securing a competitive edge in the global pharmaceutical market. Our technical team has extensively analyzed the pathway described in CN102911176A and possesses the expertise to execute this room-temperature crystallization strategy with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this novel method are fully realized at an industrial scale. Our facilities are equipped with rigorous QC labs and state-of-the-art analytical instrumentation to verify that every batch meets stringent purity specifications, consistently delivering material with the high HPLC purity profiles demonstrated in the patent examples. We are committed to being a reliable pemetrexed disodium supplier who not only provides the product but also guarantees the process integrity behind it.
We invite procurement leaders and R&D directors to engage with us to explore how this technology can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential operational efficiencies specific to your volume requirements. Our technical procurement team is ready to provide specific COA data and route feasibility assessments to demonstrate how our implementation of this low-temperature crystallization method can serve as a cornerstone for your oncology API sourcing strategy. Contact us today to discuss your requirements and secure a supply of high-purity pemetrexed disodium that meets the highest standards of quality and reliability.
