Advanced Manufacturing of 2-Amino-3-Chloro-5-Trifluoromethylpyridine for Global Agrochemical Supply Chains
The global demand for high-performance fungicides continues to drive innovation in the synthesis of key agrochemical intermediates. Patent CN112321496A introduces a transformative method for synthesizing 2-amino-3-chloro-5-trifluoromethylpyridine, a critical precursor for the broad-spectrum bactericide fluazinam. This technical breakthrough addresses long-standing challenges in the industry by replacing hazardous high-pressure ammonolysis with a safer, three-step sequence involving bromination, chlorination, and coupling. By starting from the readily available 2-aminopyridine, this route offers a compelling alternative for manufacturers seeking to optimize their supply chains. The process is characterized by mild reaction conditions, exceptional controllability, and the ability to produce high-purity products suitable for sensitive pharmaceutical and agrochemical applications. For R&D directors and procurement specialists, understanding this pathway is essential for securing a reliable agrochemical intermediate supplier capable of meeting rigorous quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 2-amino-3-chloro-5-trifluoromethylpyridine has relied heavily on the reaction of 2,3-dichloro-5-trifluoromethylpyridine with sodium amide or ammonia water. This traditional approach is fraught with significant operational hazards, primarily due to the requirement for high-temperature or high-pressure reaction environments. Such extreme conditions not only elevate the risk of industrial accidents but also impose stringent requirements on reactor specifications and safety protocols, thereby inflating capital expenditure. Furthermore, the selectivity of nucleophilic substitution under these harsh conditions can be difficult to control, often leading to complex impurity profiles that necessitate costly and time-consuming purification steps. The reliance on hazardous reagents and energy-intensive processes makes the conventional route less attractive in an era where green chemistry and operational safety are paramount concerns for modern chemical enterprises.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN112321496A presents a sophisticated solution that circumvents these inherent dangers by utilizing a stepwise functionalization strategy. Starting from 2-aminopyridine, the process employs selective electrophilic substitutions followed by a transition-metal catalyzed coupling, all conducted under significantly milder conditions. The elimination of high-pressure ammonia reactions drastically reduces the safety burden on manufacturing facilities, allowing for more flexible plant operations. Moreover, the use of common organic solvents such as MIBK, THF, and dioxane simplifies the solvent recovery and waste management processes. This novel approach not only enhances the safety profile of the manufacturing process but also improves the overall economic feasibility by utilizing low-cost, commercially abundant raw materials. It represents a paradigm shift towards safer, more sustainable cost reduction in fluazinam manufacturing.
Mechanistic Insights into Regioselective Halogenation and Pd-Catalyzed Coupling
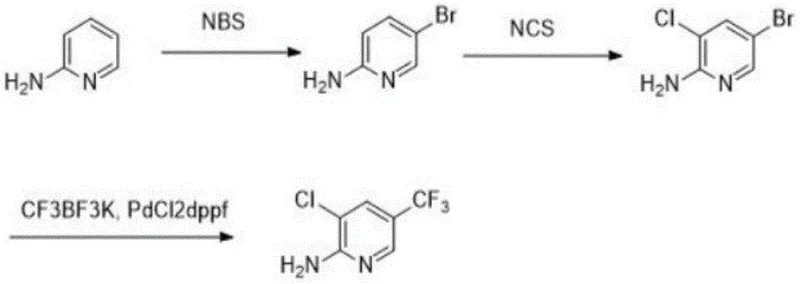
The core of this synthetic strategy lies in its precise control over regioselectivity during the halogenation phases. The first step involves the reaction of 2-aminopyridine with N-bromosuccinimide (NBS) at low temperatures ranging from -5 to 0°C. Under these controlled thermal conditions, the amino group directs the electrophilic bromination specifically to the 5-position of the pyridine ring, minimizing the formation of unwanted isomers. Following this, the intermediate undergoes chlorination using N-chlorosuccinimide (NCS) at elevated temperatures between 80 and 100°C. This second halogenation step is equally critical, as it selectively installs the chlorine atom at the 3-position. The sequential nature of these reactions ensures that the halogen atoms are placed exactly where needed for the subsequent coupling step, thereby maintaining a clean impurity profile throughout the synthesis.
The final transformation is a palladium-catalyzed cross-coupling reaction that introduces the trifluoromethyl group, a moiety essential for the biological activity of the final fungicide. Using PdCl2dppf as the catalyst and potassium trifluoromethyl trifluoroborate (CF3BF3K) as the trifluoromethyl source, the reaction proceeds efficiently in solvents like dioxane or DMSO. This step replaces the bromine atom at the 5-position with the trifluoromethyl group with high fidelity. The choice of ligand and catalyst loading is optimized to ensure complete conversion while minimizing the residual metal content in the final product. This mechanistic precision is vital for R&D teams focused on purity, as it directly correlates to the ease of downstream purification and the quality of the commercial scale-up of complex agrochemical intermediates.
How to Synthesize 2-Amino-3-Chloro-5-Trifluoromethylpyridine Efficiently
Implementing this synthesis requires careful attention to temperature control and stoichiometry to maximize yield and purity. The process begins with the dissolution of 2-aminopyridine in a suitable solvent like MIBK, followed by the slow addition of NBS while maintaining the temperature below 0°C to prevent side reactions. Once the bromination is complete, the reaction mixture is heated for the chlorination step with NCS, ensuring thorough conversion before proceeding to the coupling stage. The final coupling reaction demands an inert atmosphere and precise catalyst loading to facilitate the introduction of the trifluoromethyl group effectively. For detailed operational parameters and specific workup procedures, please refer to the standardized guide below.
- Perform selective 5-position bromination of 2-aminopyridine using NBS in solvents like MIBK or THF at low temperatures (-5 to 0°C).
- Conduct selective 3-position chlorination by reacting the brominated intermediate with NCS at elevated temperatures (80-100°C).
- Execute palladium-catalyzed trifluoromethylation using potassium trifluoromethyl trifluoroborate (CF3BF3K) and PdCl2dppf to yield the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits beyond mere technical feasibility. The shift away from high-pressure reactors to standard glass-lined or stainless steel vessels significantly lowers the barrier to entry for production, allowing for greater flexibility in sourcing manufacturing partners. The use of inexpensive starting materials like 2-aminopyridine and common succinimide reagents stabilizes the raw material cost base, insulating the supply chain from volatile price fluctuations associated with specialized precursors. Furthermore, the mild reaction conditions reduce energy consumption and extend the lifespan of production equipment, contributing to long-term operational efficiency. These factors collectively enhance the reliability of the supply chain, ensuring consistent delivery of high-purity intermediates to downstream formulators.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the elimination of expensive high-pressure infrastructure and the utilization of low-cost commodity chemicals. By avoiding the need for specialized autoclaves required for ammonia reactions, manufacturers can significantly reduce capital depreciation costs. Additionally, the high selectivity of the reaction minimizes the loss of valuable materials to byproducts, thereby improving the overall mass balance and reducing waste disposal costs. The simplified purification process, which relies on standard recrystallization techniques rather than complex chromatography, further drives down the cost of goods sold, making the final product more competitive in the global market.
- Enhanced Supply Chain Reliability: Sourcing reliability is greatly improved because the key raw materials, including 2-aminopyridine and NBS, are produced on a massive industrial scale globally. This abundance ensures that production schedules are not disrupted by shortages of niche reagents. The robustness of the reaction conditions also means that the process is less susceptible to minor variations in utility supplies, such as cooling water temperature fluctuations, which can often halt more sensitive processes. Consequently, suppliers adopting this method can offer more consistent lead times and maintain higher inventory levels of finished goods, providing a buffer against market demand spikes.
- Scalability and Environmental Compliance: Scaling this process from pilot plant to commercial tonnage is straightforward due to the absence of exothermic runaway risks associated with high-pressure ammonolysis. The ability to conduct reactions at atmospheric pressure simplifies the engineering requirements for scale-up, reducing the time and cost needed to bring new capacity online. From an environmental perspective, the process generates less hazardous waste and avoids the release of ammonia, aligning with increasingly stringent global environmental regulations. This compliance reduces the regulatory burden on manufacturers and minimizes the risk of production shutdowns due to environmental non-compliance, ensuring a stable long-term supply.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of this key fluazinam intermediate. These answers are derived directly from the technical specifications and experimental data provided in the patent literature, ensuring accuracy and relevance for industry professionals. Understanding these details is crucial for evaluating the feasibility of integrating this intermediate into your existing supply chain or product portfolio.
Q: What are the safety advantages of this new synthesis route compared to traditional methods?
A: Traditional methods often require high-temperature or high-pressure reactions with sodium amide or ammonia water, posing significant industrial safety risks. This patented method utilizes mild reaction conditions throughout the three steps, eliminating the need for extreme pressure and enhancing operational safety.
Q: How does this process ensure high purity for fluazinam production?
A: The process achieves high purity (up to 99.85%) through precise regioselective halogenation and a final recrystallization treatment. The use of specific solvents like MIBK and dioxane allows for effective impurity removal during the workup phases.
Q: Is this method suitable for large-scale commercial production?
A: Yes, the patent explicitly states that the raw materials are inexpensive and easily obtainable, and the reaction conditions are easy to control. These factors, combined with high yields (71-76%), make the route highly viable for scaling up to meet increasing market demand for fluazinam intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-3-Chloro-5-Trifluoromethylpyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of agrochemical formulations. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-amino-3-chloro-5-trifluoromethylpyridine meets the highest industry standards. Our commitment to technical excellence allows us to navigate the complexities of fine chemical synthesis, delivering products that support the efficacy and safety of your final applications.
We invite you to collaborate with us to optimize your sourcing strategy for this essential building block. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced manufacturing capabilities can add value to your supply chain. Let us be your partner in driving efficiency and innovation in the agrochemical sector.
