Advanced Synthesis of 2-Amino-3-Chloro-5-Trifluoromethylpyridine for Scalable Fluazinam Production
The global demand for high-performance fungicides continues to drive innovation in the synthesis of critical agrochemical intermediates, specifically within the fluazinam production value chain. Patent CN112321496B introduces a transformative methodology for synthesizing 2-amino-3-chloro-5-trifluoromethylpyridine, a pivotal building block that addresses longstanding challenges in process safety and purity control. This technical breakthrough shifts the paradigm from hazardous high-pressure amination protocols to a streamlined, three-step sequence initiated from the commercially abundant 2-aminopyridine. By leveraging selective halogenation strategies followed by advanced palladium-catalyzed coupling, this route offers a robust alternative for manufacturers seeking to optimize their supply chains for next-generation crop protection agents. The significance of this development extends beyond mere chemical novelty; it represents a strategic opportunity for reliable agrochemical intermediate supplier networks to enhance production stability while mitigating the risks associated with traditional high-energy synthetic pathways.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of 2-amino-3-chloro-5-trifluoromethylpyridine has relied heavily on the nucleophilic substitution of 2,3-dichloro-5-trifluoromethylpyridine using ammonia water or sodium amide. These conventional processes are fraught with significant operational hazards, primarily due to the requirement for extreme reaction conditions involving high temperatures and elevated pressures. Such environments not only increase the risk of thermal runaway and equipment failure but also necessitate specialized, costly reactor infrastructure that limits production flexibility. Furthermore, the harsh conditions often lead to the formation of complex impurity profiles, including over-amination byproducts and dehalogenated species, which complicate downstream purification and reduce overall yield. The reliance on these energy-intensive and dangerous protocols creates a bottleneck for cost reduction in fluazinam manufacturing, forcing producers to absorb higher safety compliance costs and face potential supply disruptions due to rigorous regulatory scrutiny of high-pressure operations.
The Novel Approach
In stark contrast, the patented methodology delineates a sophisticated yet practical route that circumvents these dangers by constructing the target molecule through sequential functionalization of a simpler pyridine scaffold. Starting from 2-aminopyridine, the process employs a controlled bromination followed by a selective chlorination, effectively installing the necessary halogen handles under mild, atmospheric conditions before introducing the trifluoromethyl group via cross-coupling. This stepwise assembly allows for precise control over regioselectivity, ensuring that the amino group remains intact while the halogen atoms are positioned exactly where needed for subsequent biological activity. The elimination of high-pressure amination steps fundamentally alters the risk profile of the manufacturing process, enabling safer scale-up and reducing the capital expenditure required for specialized containment systems. By utilizing readily available solvents and reagents, this approach not only simplifies the operational workflow but also enhances the economic viability of producing high-purity pyridine derivatives on a commercial scale.
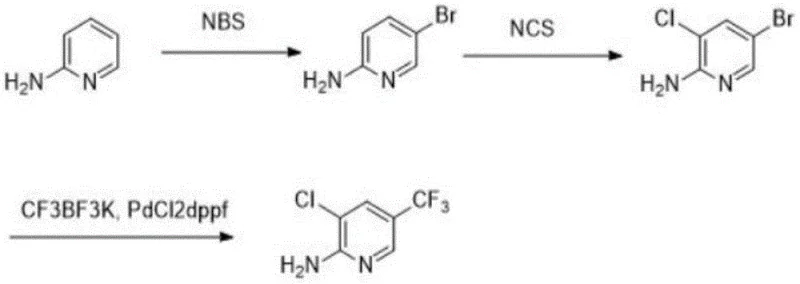
Mechanistic Insights into Selective Halogenation and Pd-Catalyzed Coupling
The core of this synthetic strategy lies in the exquisite control of electrophilic aromatic substitution and transition metal catalysis. The initial step utilizes N-bromosuccinimide (NBS) at low temperatures ranging from -5 to 0°C to achieve selective bromination at the 5-position of the pyridine ring. This low-temperature regime is critical for suppressing poly-bromination and ensuring that the electron-donating amino group directs the incoming electrophile to the desired position without compromising the integrity of the heterocyclic core. Following isolation or in-situ processing, the intermediate undergoes chlorination using N-chlorosuccinimide (NCS) at elevated temperatures between 80 and 100°C. This thermal activation facilitates the introduction of the chlorine atom at the 3-position, completing the di-halogenated scaffold required for the final transformation. The mechanistic precision here prevents the formation of isomeric impurities that typically plague non-selective halogenation reactions, thereby streamlining the purification process.
The final transformation involves a palladium-catalyzed cross-coupling reaction, specifically utilizing PdCl2dppf as the catalyst and potassium trifluoromethyl trifluoroborate (CF3BF3K) as the trifluoromethyl source. This step replaces the bromine atom at the 5-position with a trifluoromethyl group, a modification that is essential for the lipophilicity and metabolic stability of the final fungicide. The choice of CF3BF3K is particularly advantageous as it serves as a stable, non-gaseous source of the CF3 moiety, avoiding the handling difficulties associated with trifluoromethyl iodide or gas. The catalytic cycle likely proceeds through oxidative addition of the aryl bromide to the palladium center, transmetallation with the trifluoroborate salt, and reductive elimination to release the product. This mechanism ensures high conversion rates and minimizes the presence of residual heavy metals in the final API intermediate, adhering to stringent purity specifications required for agrochemical registration.
How to Synthesize 2-Amino-3-Chloro-5-Trifluoromethylpyridine Efficiently
The execution of this synthesis requires careful attention to temperature gradients and stoichiometric ratios to maximize yield and purity. The process begins with the dissolution of 2-aminopyridine in a solvent such as MIBK or THF, followed by the controlled addition of NBS to effect bromination. Subsequent chlorination with NCS demands precise thermal management to drive the reaction to completion without degradation. Finally, the coupling step necessitates an inert atmosphere and the correct loading of the palladium catalyst to ensure efficient trifluoromethylation. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized protocol below.
- Perform selective 5-position bromination of 2-aminopyridine using NBS in organic solvents like MIBK or THF at low temperatures (-5 to 0°C).
- Conduct selective 3-position chlorination by reacting the brominated intermediate with NCS at elevated temperatures (80-100°C).
- Execute palladium-catalyzed cross-coupling using potassium trifluoromethyl trifluoroborate (CF3BF3K) to introduce the trifluoromethyl group.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented route offers substantial strategic benefits by decoupling production from hazardous high-pressure infrastructure. The shift to atmospheric pressure reactions significantly lowers the barrier to entry for contract manufacturing organizations, thereby increasing the pool of qualified suppliers and enhancing supply chain resilience. By utilizing common organic solvents like MIBK, ethanol, and dioxane, the process leverages existing solvent recovery systems found in most fine chemical facilities, eliminating the need for specialized solvent procurement or disposal protocols. This compatibility with standard industrial equipment translates directly into reduced capital expenditure and faster time-to-market for new production lines. Furthermore, the use of stable, solid reagents like NBS and NCS simplifies logistics and storage requirements compared to gaseous or highly reactive liquid alternatives, ensuring a more reliable flow of materials throughout the manufacturing lifecycle.
- Cost Reduction in Manufacturing: The economic advantages of this route are driven primarily by the use of inexpensive starting materials and the elimination of energy-intensive high-pressure steps. 2-Aminopyridine is a commodity chemical with a stable global supply, insulating manufacturers from the price volatility often seen with specialized fluorinated precursors. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, while the high selectivity of the halogenation steps minimizes the loss of valuable intermediates to side reactions. The ability to achieve high purity through simple recrystallization rather than complex chromatographic separation further drives down processing costs, making the overall cost structure highly competitive for large-volume agrochemical production.
- Enhanced Supply Chain Reliability: Supply continuity is significantly bolstered by the robustness of the reaction conditions and the availability of raw materials. Unlike processes that rely on scarce or regulated reagents, this method utilizes widely accessible chemicals that can be sourced from multiple vendors globally. The operational safety profile reduces the likelihood of unplanned shutdowns due to safety incidents or regulatory inspections, providing buyers with greater confidence in delivery schedules. Moreover, the scalability of the process means that production volumes can be ramped up quickly to meet seasonal demand spikes in the agricultural sector without requiring extensive lead times for equipment installation or validation.
- Scalability and Environmental Compliance: The environmental footprint of this synthesis is favorable due to the avoidance of hazardous high-pressure waste streams and the use of recyclable solvents. The process generates fewer byproducts that require complex treatment, simplifying wastewater management and reducing the burden on environmental compliance teams. The solid nature of key reagents and the final product facilitates easier handling and packaging, reducing the risk of spills or leaks during transport. This alignment with green chemistry principles not only meets current regulatory standards but also future-proofs the supply chain against tightening environmental regulations, ensuring long-term viability for commercial scale-up of complex heterocycles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and process descriptions outlined in the patent documentation, providing clarity on yield expectations, purity profiles, and operational safety. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this new methodology for their specific production needs.
Q: What are the safety advantages of this new synthesis route compared to traditional methods?
A: Traditional methods often involve high-temperature or high-pressure reactions between 2,3-dichloro-5-trifluoromethylpyridine and ammonia or sodium amide, which pose significant industrial safety risks. The patented route utilizes mild conditions with standard atmospheric pressure and moderate temperatures, significantly reducing operational hazards.
Q: How does this process ensure high purity for the final agrochemical intermediate?
A: The process achieves high purity through precise regioselective halogenation steps followed by a recrystallization treatment. The use of specific solvents like MIBK and ethanol/water mixtures during workup allows for effective removal of impurities, resulting in product purity exceeding 99.5%.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the route is designed for scalability. It utilizes inexpensive and readily available raw materials like 2-aminopyridine and common organic solvents. The reaction conditions are easy to control, making it highly adaptable for tonnage-level manufacturing of fluazinam intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-3-Chloro-5-Trifluoromethylpyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the efficacy and safety of final agrochemical products. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to industrial manufacturing is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of 2-amino-3-chloro-5-trifluoromethylpyridine meets the exacting standards required for fluazinam synthesis. Our infrastructure is designed to handle complex halogenation and coupling chemistries safely, providing a secure foundation for your supply chain.
We invite you to collaborate with us to leverage this advanced synthesis technology for your upcoming projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can drive value and reliability in your agrochemical manufacturing operations.
