Advanced Aminolysis Technology for High-Purity Fluorinated Pyridine Intermediates in Agrochemical Manufacturing
The global demand for fluorinated heterocyclic compounds in the agrochemical sector has driven significant innovation in synthetic methodologies, particularly for key intermediates like 2-amino-3-chloro-5-trifluoromethylpyridine. Patent CN100497311C introduces a robust and environmentally superior preparation method that addresses critical bottlenecks in traditional manufacturing. This technology utilizes a high-pressure aminolysis reaction to convert 2,3-dichloro-5-trifluoromethylpyridine into the target amino-chloro derivative with exceptional efficiency. By operating under controlled thermal and pressure conditions ranging from 123-135°C and 2.0-3.5 MPa, the process ensures complete conversion while maintaining strict control over byproduct formation. For R&D directors and procurement specialists seeking a reliable agrochemical intermediate supplier, this patent represents a pivotal shift towards greener, high-yield chemistry that aligns with modern sustainability mandates.
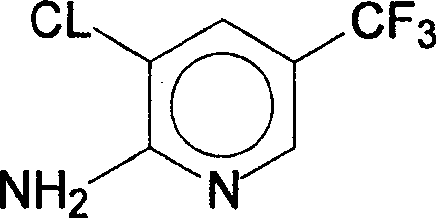
The structural integrity of the final product is paramount for downstream pesticide efficacy, and the method described ensures the preservation of the sensitive trifluoromethyl group while selectively introducing the amino functionality. This specific substitution pattern is crucial for the biological activity of next-generation fungicides and insecticides used in major crop protection programs. The ability to source this high-purity agrochemical intermediate through a streamlined single-step reaction significantly de-risks the supply chain for multinational agriscience companies. Furthermore, the elimination of complex purification steps typically required in older synthetic routes translates directly into operational excellence and reduced time-to-market for new active ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fluorinated pyridine derivatives often relied on harsh nitration protocols involving mixed acids, which presented severe logistical and environmental challenges for large-scale agrochemical intermediate manufacturing. Traditional routes frequently utilized 3-chloro-5-trifluoromethylpyridine as a starting material, subjecting it to aggressive nitration conditions using concentrated sulfuric and nitric acid mixtures. This approach not only posed significant safety risks due to the exothermic nature of nitration but also generated substantial quantities of hazardous spent acid and wastewater that were difficult and costly to treat. The environmental burden of disposing of these acidic effluents often negated the economic benefits of the raw materials, creating a bottleneck for cost reduction in agrochemical manufacturing. Additionally, the separation of isomers and the removal of trace metal impurities from acid-catalyzed reactions often required multiple recrystallization steps, further depressing overall yields and increasing production lead times.
The Novel Approach
In stark contrast, the novel aminolysis pathway detailed in the patent data offers a transformative solution by utilizing 2,3-dichloro-5-trifluoromethylpyridine as a more reactive and selective precursor. This method bypasses the need for corrosive nitrating agents entirely, replacing them with liquid ammonia in a common organic solvent system such as methanol or ethanol. The reaction proceeds via a clean nucleophilic substitution mechanism that inherently produces fewer byproducts, thereby simplifying the downstream workup to merely solvent removal and water washing. This simplicity is a game-changer for commercial scale-up of complex fluorinated intermediates, as it allows for continuous processing in standard high-pressure autoclaves without the need for specialized acid-resistant lining or extensive neutralization infrastructure. The result is a process that is not only chemically elegant but also economically superior, delivering yields around 90% with purity levels exceeding 99%.
Mechanistic Insights into High-Pressure Aminolysis
The core of this technological breakthrough lies in the precise exploitation of nucleophilic aromatic substitution (SnAr) dynamics on the electron-deficient pyridine ring. The presence of the strongly electron-withdrawing trifluoromethyl group at the 5-position, combined with the electronegative ring nitrogen, significantly activates the carbon-chlorine bonds towards nucleophilic attack. However, the reaction exhibits remarkable regioselectivity, preferentially displacing the chlorine atom at the 2-position over the one at the 3-position. This selectivity is driven by the inductive effect of the ring nitrogen, which renders the adjacent C2 position more electrophilic and thus more susceptible to attack by the ammonia nucleophile. Understanding this mechanistic nuance is vital for R&D teams aiming to optimize reaction parameters, as slight deviations in temperature or pressure could theoretically influence the ratio of mono-substituted to di-substituted products.
Furthermore, the use of liquid ammonia in a pressurized environment ensures a high local concentration of the nucleophile, driving the equilibrium towards the desired amine product despite the potential reversibility of aminolysis reactions. The choice of solvent plays a critical role in stabilizing the transition state; polar protic solvents like methanol or ethanol facilitate the dissolution of ammonia and stabilize the developing negative charge in the Meisenheimer complex intermediate. From an impurity control perspective, this mechanism minimizes the formation of tar-like polymerization byproducts often seen in high-temperature acid catalysis. The resulting crude product is remarkably clean, requiring only a simple aqueous wash to remove residual ammonium salts and solvent, which explains the consistently high purity profiles reported in the patent embodiments.
How to Synthesize 2-Amino-3-Chloro-5-Trifluoromethylpyridine Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for industrial implementation, emphasizing safety and reproducibility in high-pressure environments. The process begins with the charging of the dichloro precursor and a selected alcohol solvent into a sealed autoclave, followed by the careful introduction of liquid ammonia to achieve the specified molar excess. Maintaining the reaction temperature between 125-135°C and pressure between 2.0-3.5 MPa is critical for ensuring complete conversion within the 8 to 15-hour timeframe. Detailed standardized synthesis steps see the guide below.
- Charge 2,3-dichloro-5-trifluoromethylpyridine and an organic solvent (methanol, ethanol, or isopropanol) into a high-pressure autoclave reactor.
- Introduce liquid ammonia at a molar ratio of 1: 10 to 1:18 relative to the substrate, then heat to 125-135°C under 2.0-3.5 MPa pressure for 8-15 hours.
- Distill off the solvent, wash the residue with water, centrifuge to isolate the solid, and dry to obtain the product with >99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this aminolysis technology offers tangible strategic advantages beyond mere chemical yield. The shift away from corrosive acid-based chemistry fundamentally alters the cost structure of production by eliminating the need for expensive acid-resistant equipment and complex waste treatment facilities. This simplification of the manufacturing infrastructure allows for greater flexibility in plant utilization and significantly lowers the barrier to entry for scaling production volumes. Moreover, the reliance on commodity chemicals like liquid ammonia and methanol ensures a stable and predictable raw material supply chain, insulating manufacturers from the volatility often associated with specialized reagents.
- Cost Reduction in Manufacturing: The elimination of heavy metal catalysts and corrosive mineral acids removes the necessity for costly downstream purification steps such as ion exchange or activated carbon treatment. By streamlining the workup to simple distillation and filtration, the process drastically reduces utility consumption (steam and water) and labor hours per batch. This operational efficiency translates into substantial cost savings, allowing suppliers to offer more competitive pricing structures for long-term contracts without compromising on margin. The high yield of approximately 90% further maximizes raw material utilization, ensuring that every kilogram of expensive fluorinated starting material is converted into saleable product with minimal waste.
- Enhanced Supply Chain Reliability: The robustness of the high-pressure aminolysis reaction makes it highly resilient to minor fluctuations in feedstock quality, ensuring consistent output even when sourcing raw materials from diverse suppliers. The simplified process flow reduces the number of unit operations, thereby decreasing the probability of mechanical failure or batch rejection due to human error. This reliability is crucial for maintaining just-in-time inventory levels for downstream pesticide formulators who cannot afford interruptions in their production schedules. Additionally, the absence of hazardous waste generation simplifies logistics and regulatory compliance, reducing the risk of shipment delays caused by environmental inspections.
- Scalability and Environmental Compliance: As regulatory scrutiny on chemical manufacturing intensifies globally, this green chemistry approach positions manufacturers favorably for future compliance audits. The process generates negligible wastewater and no solid waste residues, aligning perfectly with zero-discharge initiatives adopted by many industrial parks. This environmental stewardship not only mitigates regulatory risk but also enhances the brand reputation of the supply chain partners. The technology is readily scalable from pilot plants to multi-ton reactors using standard pressure vessel technology, facilitating rapid capacity expansion to meet surging market demand for fluorinated agrochemicals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this fluorinated pyridine intermediate. These insights are derived directly from the patented methodology and are intended to clarify the operational benefits for potential partners. Understanding these details is essential for evaluating the feasibility of integrating this intermediate into your existing supply chain.
Q: What is the primary advantage of the aminolysis route over traditional nitration methods for this intermediate?
A: The aminolysis route described in patent CN100497311C eliminates the use of corrosive sulfuric and nitric acid mixtures, thereby avoiding the generation of large volumes of spent acid and wastewater associated with traditional nitration processes, significantly reducing environmental compliance costs.
Q: How is regioselectivity controlled to ensure the amino group substitutes only at the 2-position?
A: The reaction leverages the inherent electronic activation of the pyridine ring; the chlorine atom at the 2-position (alpha to the ring nitrogen) is significantly more susceptible to nucleophilic attack by ammonia than the chlorine at the 3-position, ensuring high regioselectivity without needing protecting groups.
Q: What are the typical yield and purity specifications achievable with this process?
A: According to the patent data, this optimized high-pressure aminolysis process consistently achieves product yields of approximately 90% with a final purity exceeding 99%, making it highly suitable for direct use in sensitive agrochemical formulations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-3-Chloro-5-Trifluoromethylpyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation crop protection solutions. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 2-amino-3-chloro-5-trifluoromethylpyridine meets the exacting standards required for pharmaceutical and agrochemical applications. Our commitment to quality assurance extends beyond the final product, encompassing full traceability of raw materials and adherence to international safety protocols.
We invite you to collaborate with us to leverage this advanced aminolysis technology for your specific project needs. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your volume forecasts, demonstrating how our optimized process can improve your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate why we are the preferred partner for complex fluorinated chemistry.
