Advanced Three-Step Synthesis Strategy for High-Purity Carbapenem Antibiotic Intermediates
Advanced Three-Step Synthesis Strategy for High-Purity Carbapenem Antibiotic Intermediates
The escalating crisis of bacterial resistance has positioned carbapenem antibiotics as a critical line of defense in modern medicine, necessitating robust and efficient supply chains for their key precursors. In response to the industry demand for streamlined manufacturing processes, patent CN101367831B discloses a highly efficient methodology for synthesizing a pivotal carbapenem intermediate, specifically (3S,4R)-3-[(1R)-1-(tert-butyldimethylsilyloxy)ethyl]-4-{2-[2-(tert-butoxycarbonylamino)ethylthio]carboxyethyl}-2-azetidinone. This molecule serves as a fundamental building block for next-generation beta-lactamase inhibitors and broad-spectrum antibiotics. The structural complexity of this intermediate, featuring both TBDMS and Boc protecting groups alongside a sensitive azetidinone core, traditionally poses significant challenges in terms of stability and purification. However, the disclosed innovation offers a pathway that mitigates these risks through a concise three-step sequence starting from commercially available 4-acetoxy-2-azetidinone derivatives. 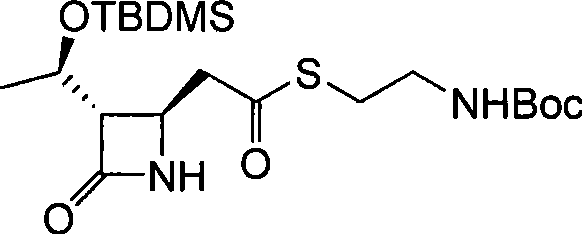
For procurement managers and supply chain directors, the implications of this technology extend far beyond mere chemical novelty; it represents a tangible opportunity for cost reduction in pharmaceutical manufacturing and enhanced supply security. By reducing the number of unit operations and eliminating the need for exotic reagents, this process directly addresses the pain points of long lead times and volatile raw material costs. As a reliable carbapenem intermediate supplier, understanding the nuances of such patented routes allows us to offer clients not just a product, but a validated, scalable solution that aligns with stringent regulatory requirements for API production. The following analysis dissects the technical merits and commercial viability of this synthesis, providing a comprehensive view for stakeholders involved in the sourcing of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the total synthesis of carbapenem antibiotics has been plagued by excessively long reaction sequences that often exceed ten discrete steps, leading to cumulative yield losses and substantial waste generation. Traditional routes frequently rely on precious metal catalysts, such as palladium or rhodium complexes, which not only drive up the raw material costs but also introduce complex downstream processing requirements to meet residual metal specifications. Furthermore, many conventional methodologies necessitate cryogenic conditions or highly hazardous reagents that complicate safety protocols and increase the capital expenditure required for specialized reactor infrastructure. These factors collectively contribute to a fragile supply chain where minor disruptions in reagent availability can halt production for weeks, creating significant bottlenecks for downstream API manufacturers. The environmental footprint of these older methods is also considerable, often generating large volumes of heavy metal-contaminated waste that requires expensive treatment before disposal.
The Novel Approach
In stark contrast, the methodology outlined in the patent data presents a streamlined three-step protocol that dramatically simplifies the production landscape while maintaining high stereochemical fidelity. By utilizing a commercially available acetoxy-azetidinone starting material, the process bypasses the need for constructing the beta-lactam ring from scratch, thereby saving considerable time and resources. The reaction conditions are notably mild, operating at near-ambient temperatures for the allylation step and utilizing common oxidants like potassium permanganate, which are inexpensive and easy to source globally. This approach eliminates the dependency on scarce transition metals, replacing them with activated zinc, a commodity metal that is both cost-effective and easier to remove from the final product matrix. The result is a synthesis that is not only chemically elegant but also industrially pragmatic, offering a clear path toward cost reduction in pharmaceutical manufacturing without compromising on the purity or quality of the final intermediate.
Mechanistic Insights into Zinc-Mediated Allylation and Oxidative Functionalization
The cornerstone of this synthesis is the initial allylation reaction, where the acetoxy group of the starting material is displaced by an allyl moiety using activated zinc powder. This transformation is critical as it establishes the carbon framework necessary for subsequent side-chain elaboration while preserving the delicate stereochemistry at the C3 and C4 positions of the azetidinone ring. The mechanism likely proceeds through the formation of an organozinc species in situ, which acts as a nucleophile to attack the electrophilic center, facilitated by the Lewis acidic nature of the zinc surface. The use of solvents such as tetrahydrofuran or acetonitrile ensures adequate solubility of the organic substrates while maintaining the stability of the organometallic intermediate. Strict control of the reaction temperature between 30°C and 35°C is essential to prevent thermal degradation of the beta-lactam ring and to minimize side reactions such as polymerization of the allyl bromide. 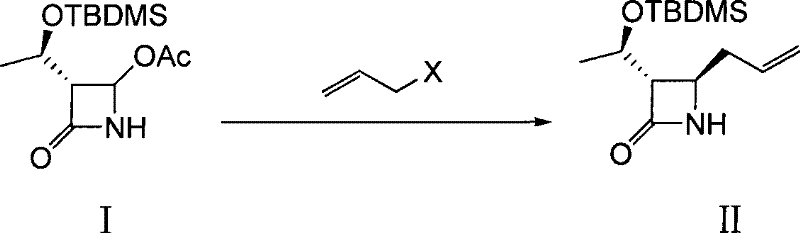
Following the successful installation of the allyl group, the process moves to an oxidative cleavage step using potassium permanganate to convert the terminal alkene into a carboxylic acid. This oxidation is performed in a biphasic system of acetone and water, buffered to a pH of 2 to 4 to optimize the oxidizing power of the permanganate while preventing over-oxidation or ring opening. The final step involves the coupling of this newly formed acid with N-tert-butoxycarbonyl cysteamine to install the requisite thioether side chain. This esterification is mediated by carbodiimide coupling agents like DCC or EDCI in the presence of catalytic DMAP, ensuring high conversion rates under mild conditions. The choice of protecting groups, specifically the TBDMS ether and the Boc amine, provides orthogonal stability, allowing for selective deprotection in later stages of the antibiotic synthesis without affecting other functional groups. 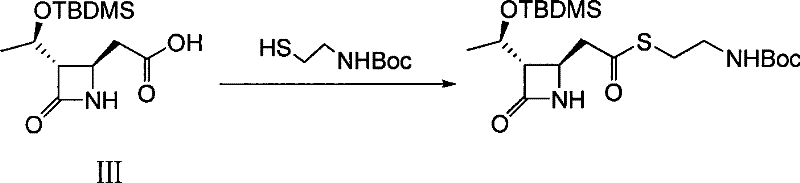
How to Synthesize Carbapenem Intermediate Efficiently
The execution of this synthesis requires precise adherence to the specified reaction parameters to ensure optimal yield and purity, particularly regarding the activation of the zinc powder and the control of pH during oxidation. The process is designed to be robust, utilizing standard laboratory and plant equipment without the need for specialized high-pressure or cryogenic apparatus. Detailed operational guidelines regarding reagent stoichiometry, addition rates, and workup procedures are critical for reproducing the high yields reported in the patent examples, which range from 60% to 78% for individual steps. For technical teams looking to implement this route, the following standardized procedure outlines the key operational milestones derived from the proprietary data.
- Perform zinc-mediated allylation of 4-acetoxy-2-azetidinone derivative with allyl bromide in THF at 30-35°C to form the allyl intermediate.
- Oxidize the resulting allyl compound using potassium permanganate in acetone/water mixture at pH 2-4 and temperature -5 to 2°C to generate the carboxylic acid.
- Conduct esterification of the carboxylic acid with N-tert-butoxycarbonyl cysteamine using DCC and DMAP catalyst in dichloromethane to yield the final thioether intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this synthesis route offers compelling advantages that directly impact the bottom line and operational resilience of pharmaceutical supply chains. The shift away from precious metal catalysts to base metals like zinc represents a fundamental change in the cost structure of the intermediate, removing exposure to the volatile pricing of platinum group metals. Additionally, the use of commodity chemicals such as allyl bromide, potassium permanganate, and cysteamine hydrochloride ensures that raw material availability is not a bottleneck, as these are produced in massive quantities for various industrial applications. The simplified workup procedures, which rely on filtration and liquid-liquid extraction rather than complex chromatographic separations, significantly reduce the cycle time per batch and lower the consumption of silica gel and solvents. These factors combine to create a manufacturing process that is inherently more stable and predictable, reducing the risk of supply interruptions for downstream API producers.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of activated zinc significantly lowers the direct material costs associated with the synthesis. Furthermore, the mild reaction temperatures reduce energy consumption for heating and cooling, contributing to lower utility costs per kilogram of product. The high atom economy of the allylation and coupling steps minimizes waste generation, which in turn reduces the costs associated with waste disposal and environmental compliance. By streamlining the process to only three steps, labor costs and equipment occupancy time are drastically reduced compared to longer conventional routes.
- Enhanced Supply Chain Reliability: Reliance on widely available commodity reagents ensures that the supply chain is resilient to geopolitical or market-specific shortages that often affect specialty fine chemicals. The robustness of the reaction conditions means that the process can be easily transferred between different manufacturing sites or scaled up without requiring extensive re-optimization. This flexibility allows for dual-sourcing strategies and inventory buffering, ensuring continuous availability of the intermediate even during periods of high market demand. The simplicity of the purification steps also reduces the lead time for quality control testing, accelerating the release of batches for shipment.
- Scalability and Environmental Compliance: The process avoids the use of chlorinated solvents in the oxidation step and utilizes aqueous workups where possible, aligning with green chemistry principles and reducing the environmental burden. The absence of heavy metal residues simplifies the regulatory filing process for the final drug product, as extensive cleaning validation and metal scavenging steps are not required. The reaction exotherms are manageable, allowing for safe scale-up in standard glass-lined or stainless steel reactors without the need for specialized containment systems. This ease of scale-up facilitates rapid response to market needs, enabling the production of multi-ton quantities to support commercial API manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, based on the detailed experimental data provided in the patent documentation. These insights are intended to clarify the operational feasibility and quality attributes of the intermediate for potential partners and licensees.
Q: What are the key advantages of this synthesis route over conventional methods?
A: This route significantly shortens the synthetic sequence to only three steps from a commercially available starting material, avoiding harsh reaction conditions and expensive transition metal catalysts typically required in traditional carbapenem synthesis.
Q: How is stereochemical integrity maintained during the allylation step?
A: The process utilizes activated zinc powder under strictly anhydrous and anaerobic conditions at controlled temperatures (30-35°C), which ensures high stereoselectivity and prevents epimerization of the sensitive beta-lactam ring.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method employs readily available reagents like allyl bromide and potassium permanganate, and the workup procedures involve standard filtration and extraction techniques, making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbapenem Intermediate Supplier
The technical elegance of this three-step synthesis underscores the potential for efficient, large-scale production of critical antibiotic precursors, a capability that NINGBO INNO PHARMCHEM is uniquely positioned to deliver. As a seasoned CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and compliant. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, including residual solvent analysis and chiral purity assessment, to guarantee that every batch meets the exacting standards required for GMP API synthesis. We understand that consistency is key in the pharmaceutical supply chain, and our process engineering team is dedicated to optimizing every parameter to maximize yield and minimize variability.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By leveraging this optimized synthesis route, we can help you secure a stable supply of high-quality intermediates while simultaneously driving down your overall cost of goods. Please contact our technical procurement team to request specific COA data, route feasibility assessments, and a comprehensive proposal for your upcoming projects. Let us collaborate to strengthen your supply chain and accelerate the delivery of life-saving medications to patients worldwide.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
