Advanced Synthesis of Imiquimod Intermediates via Novel Cyanation Route for Commercial Scale-up
Introduction to Patent CN1684963A and Technological Breakthroughs
The pharmaceutical landscape for immunomodulators has been significantly advanced by the disclosures within patent CN1684963A, which details a robust and innovative synthetic pathway for preparing 1H-imidazo[4,5-c]quinoline-4-amines, most notably the critical active pharmaceutical ingredient (API) Imiquimod. This patent represents a pivotal shift away from legacy manufacturing techniques that often relied on hazardous high-pressure conditions or complex multi-step protection-deprotection sequences. Instead, it introduces a streamlined approach utilizing novel 1H-imidazo[4,5-c]quinoline-4-cyano and 4-carboxamide intermediates. These intermediates serve as the cornerstone for a more efficient synthesis, offering superior control over regioselectivity and impurity profiles. For R&D directors and process chemists, the significance of this methodology lies in its ability to bypass the thermodynamic barriers associated with direct nucleophilic aromatic substitution on the quinoline ring, thereby opening a viable route for high-purity production.
Furthermore, the strategic implementation of these intermediates addresses long-standing supply chain vulnerabilities associated with the production of immune response modifiers used in treating viral infections such as genital warts. By establishing a reliable synthetic sequence that transitions smoothly from an N-oxide precursor through a cyano intermediate to the final amine, the technology ensures a consistent supply of high-quality material. This is particularly vital for generic manufacturers and CDMOs aiming to secure market share in the dermatological and oncological sectors. The process described not only enhances the chemical elegance of the synthesis but also aligns with modern green chemistry principles by reducing the reliance on extreme reaction parameters, thus positioning it as a preferred method for industrial application.
![Chemical structure of Imiquimod (1-isobutyl-1H-imidazo[4,5-c]quinolin-4-amine)](/insights/img/imiquimod-intermediate-synthesis-pharma-supplier-20260305124455-01.png)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of the 4-amino group onto the imidazoquinoline scaffold has been fraught with significant technical challenges that hindered efficient commercial manufacturing. Traditional methods predominantly relied on nucleophilic substitution reactions where a leaving group, such as chlorine or triflate, was displaced by ammonia or amines under forcing conditions. For instance, prior art often necessitated the use of high temperatures and high pressures to drive the reaction of 4-chloro derivatives with ammonia, which posed severe safety risks and required specialized, expensive reactor infrastructure capable of withstanding such extremes. Additionally, alternative routes involving the reaction of N-oxides with benzoyl isocyanate or the use of azide chemistry introduced complex purification burdens due to the formation of toxic byproducts and difficult-to-separate isomers.
Moreover, the reliance on dibenzylamine followed by catalytic hydrogenation, as seen in some earlier patents, added unnecessary steps and cost to the overall process. The removal of benzyl groups typically requires precious metal catalysts and rigorous hydrogenation protocols, which not only increase the capital expenditure but also introduce potential heavy metal contamination issues that must be meticulously managed to meet stringent pharmacopeial standards. These conventional pathways often resulted in lower overall yields and broader impurity spectra, necessitating extensive recrystallization efforts that further eroded process efficiency. Consequently, there was a pressing industry need for a method that could operate under milder conditions while delivering superior purity without the logistical nightmare of high-pressure processing.
The Novel Approach
The methodology outlined in CN1684963A presents a transformative solution by leveraging a cyanation-hydrolysis-rearrangement sequence that fundamentally alters the reaction landscape. This novel approach initiates with the conversion of a 1H-imidazo[4,5-c]quinoline-N-oxide into a 4-cyano intermediate using alkali metal cyanides, a reaction that proceeds efficiently at near-ambient or slightly cooled temperatures. This eliminates the need for the extreme thermal energy inputs required by traditional nucleophilic substitutions. The subsequent hydrolysis of the nitrile group to a carboxamide, followed by a Hofmann rearrangement, provides a highly controlled pathway to the target amine. This sequence is not only chemically elegant but also operationally simpler, allowing for better heat management and safer handling of reagents on a large scale.
![Overall reaction process flow for the preparation of 1H-imidazo[4,5-c]quinoline-4-amine](/insights/img/imiquimod-intermediate-synthesis-pharma-supplier-20260305124455-05.webp)
By adopting this route, manufacturers can achieve a drastic simplification of the production workflow. The avoidance of high-pressure autoclaves reduces the barrier to entry for production facilities and lowers the associated insurance and maintenance costs. Furthermore, the intermediates generated, specifically the 4-cyano and 4-carboxamide derivatives, are stable and crystalline, facilitating easy isolation and purification between steps. This modularity allows for flexible manufacturing strategies where intermediates can be stockpiled or shipped if necessary, enhancing supply chain resilience. The transition from an N-oxide to a cyano group is particularly advantageous as it utilizes the activating nature of the N-oxide to facilitate nucleophilic attack at the 4-position, a transformation that is both rapid and selective, minimizing the formation of regioisomeric impurities that plague other synthetic strategies.
Mechanistic Insights into Alkali Metal Cyanide Mediated Cyclization and Rearrangement
The core of this innovative synthesis lies in the precise mechanistic execution of the cyanation step, where the 1H-imidazo[4,5-c]quinoline-N-oxide reacts with an alkali metal cyanide, preferably sodium cyanide. This transformation is conducted in a biphasic solvent system, typically comprising methylene dichloride and water, which is crucial for solubilizing both the organic substrate and the inorganic cyanide salt. The reaction is promoted by the presence of an organic acyl halide, such as benzoyl chloride, which acts as a dehydrating agent and activates the N-oxide for nucleophilic attack. Maintaining the reaction temperature between -5°C and 5°C is critical; this thermal control suppresses side reactions, particularly the formation of 4-hydroxy derivatives, which are common byproducts in N-oxide chemistry. The stoichiometry is carefully managed, often using a significant excess of cyanide (e.g., a 3:1 molar ratio), to drive the equilibrium towards the desired nitrile and ensure complete consumption of the starting material.
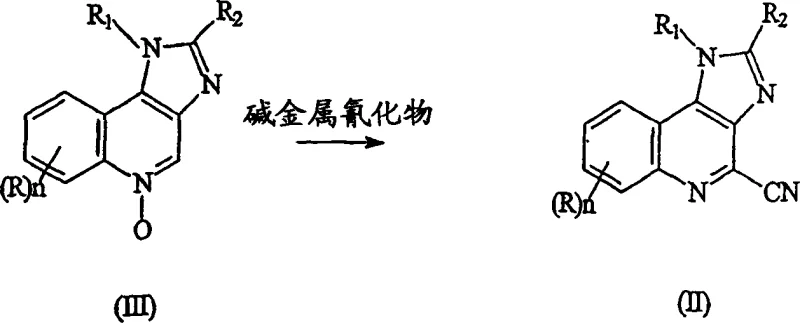
Following the successful formation of the 4-cyano intermediate, the process employs a classic yet optimized hydrolysis step using a strong acid aqueous solution, such as 30% hydrochloric acid. Heating this mixture to moderate temperatures (40-60°C) facilitates the conversion of the nitrile to the corresponding primary carboxamide. This step is remarkably clean, with the patent data indicating that the resulting carboxamide intermediate can achieve HPLC purities of at least 99%. The final transformation involves a Hofmann rearrangement, where the carboxamide is treated with a hypohalous acid salt (like sodium hypochlorite) in the presence of a strong base (such as sodium hydroxide). This degradation reaction shortens the carbon chain by one atom, effectively converting the carbonyl carbon into an amine nitrogen. The use of glycol dimethyl ether as a co-solvent in this step enhances the solubility of the organic substrate in the aqueous basic medium, ensuring homogeneous reaction conditions and high conversion rates to the final 1H-imidazo[4,5-c]quinoline-4-amine.
How to Synthesize 1-Isobutyl-1H-imidazo[4,5-c]quinoline-4-amine Efficiently
The synthesis of this high-value immunomodulator intermediate requires strict adherence to the optimized parameters defined in the patent to ensure maximum yield and purity. The process begins with the preparation of the N-oxide precursor, which is then subjected to the critical cyanation step under biphasic conditions. Operators must monitor the addition of the acyl halide carefully to manage the exotherm and maintain the low-temperature window essential for selectivity. Once the cyano intermediate is formed and isolated, it undergoes acid hydrolysis to yield the carboxamide, which serves as the direct precursor for the final amination. The detailed standardized synthesis steps, including specific reagent grades, stirring rates, and workup procedures, are outlined below to guide process engineers in replicating this high-efficiency route.
- React 1H-imidazo[4,5-c]quinoline-N-oxide with alkali metal cyanide (e.g., NaCN) in a biphasic system with an organic acyl halide at low temperature (-5 to 5°C) to form the 4-cyano intermediate.
- Hydrolyze the 4-cyano intermediate using a strong acid aqueous solution (e.g., 30% HCl) at moderate temperatures (40-60°C) to yield the 4-carboxamide intermediate.
- Subject the 4-carboxamide intermediate to Hofmann rearrangement conditions using a hypohalous acid salt (e.g., NaOCl) and strong base (e.g., NaOH) to generate the final 4-amine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers profound strategic benefits that extend beyond mere chemical yield. The primary advantage lies in the substantial cost reduction in pharmaceutical intermediate manufacturing achieved by eliminating the need for high-pressure reactor vessels. Traditional methods requiring ammonia under pressure demand specialized infrastructure that is capital intensive to build and maintain; by shifting to a cyanation-based approach that operates at atmospheric pressure and mild temperatures, facilities can utilize standard glass-lined or stainless steel reactors, significantly lowering capital expenditure and operational overheads. Additionally, the reagents employed, such as sodium cyanide and sodium hypochlorite, are commodity chemicals available from a broad global supplier base, reducing the risk of supply bottlenecks associated with exotic or proprietary catalysts.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the simplification of the purification train. Because the intermediates (cyano and carboxamide) are obtained with high purity directly from the reaction mixture, the need for extensive chromatographic purification or multiple recrystallizations is drastically reduced. This reduction in downstream processing translates directly into lower solvent consumption, reduced waste disposal costs, and shorter cycle times per batch. Furthermore, the avoidance of precious metal catalysts, which are often required for hydrogenation steps in alternative routes, removes a significant variable cost component and eliminates the need for expensive metal scavenging resins to meet residual metal specifications.
- Enhanced Supply Chain Reliability: From a logistics perspective, the robustness of this chemistry ensures consistent batch-to-batch quality, which is critical for maintaining regulatory compliance and avoiding costly production delays. The use of stable, isolable intermediates allows for a decoupled manufacturing strategy, where the cyano or carboxamide intermediates can be produced at one location and shipped to another for final conversion, providing flexibility in capacity planning. This modularity mitigates the risk of a single point of failure in the supply chain. Moreover, the raw materials are not subject to the same geopolitical restrictions or scarcity issues as some specialized organometallic reagents, ensuring a steady and predictable flow of inputs for continuous commercial production.
- Scalability and Environmental Compliance: Scaling this process from pilot plant to multi-ton commercial production is inherently safer and more straightforward due to the manageable thermal profile of the reactions. The exotherms are controlled through slow addition and cooling, rather than relying on the massive heat removal capacity needed for high-pressure exothermic reactions. Environmentally, the process generates less hazardous waste compared to routes involving heavy metals or azides. The aqueous waste streams from the hydrolysis and rearrangement steps can be treated using standard neutralization and oxidation protocols, simplifying effluent management and helping facilities meet increasingly stringent environmental regulations without investing in specialized treatment infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and claims within the patent documentation, providing clarity on safety, purity, and scalability concerns that often arise during technology transfer. Understanding these nuances is essential for project managers evaluating the feasibility of adopting this method for their specific production lines.
Q: How does this novel cyanation route improve impurity profiles compared to traditional chlorination methods?
A: The novel route avoids the formation of difficult-to-remove chlorinated byproducts and eliminates the need for high-pressure ammonia substitutions. By utilizing a controlled cyanation followed by hydrolysis, the process achieves HPLC purities exceeding 99% for the carboxamide intermediate, significantly reducing the burden on downstream purification steps.
Q: Is the use of sodium cyanide safe for large-scale commercial manufacturing?
A: Yes, while sodium cyanide requires strict safety protocols, the process utilizes a biphasic system (DCM/Water) at low temperatures (-5 to 5°C), which effectively controls the reaction exotherm and minimizes the release of hazardous gases. Furthermore, the subsequent acid hydrolysis step consumes the cyano group completely, ensuring no residual cyanide carries over into the final API.
Q: What are the scalability advantages of the Hofmann rearrangement step in this process?
A: The Hofmann rearrangement in this protocol uses commercially available bleach (NaOCl) and caustic soda (NaOH) in a glycol ether/water solvent system. This avoids the use of hazardous reagents like lead tetraacetate or elemental bromine often found in older literature, making the reaction safer and more straightforward to scale from kilogram to multi-ton production without specialized corrosion-resistant equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1H-imidazo[4,5-c]quinoline-4-amine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the competitive landscape of pharmaceutical intermediates. Our team of expert process chemists has extensively evaluated the technology disclosed in CN1684963A and possesses the technical capability to execute this complex sequence with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless. Our state-of-the-art facilities are equipped with the necessary containment systems for handling cyanides and the corrosion-resistant reactors required for the acid hydrolysis steps, guaranteeing both safety and quality. We adhere to stringent purity specifications and operate rigorous QC labs to ensure every batch meets the highest international standards.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis can be integrated into your supply chain. By leveraging our expertise, you can secure a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us to request specific COA data from our pilot runs and comprehensive route feasibility assessments. Let us collaborate to optimize your production of Imiquimod intermediates, ensuring reliability, cost-efficiency, and uninterrupted supply for your global operations.
