Advanced Manufacturing of Imiquimod Intermediates via Novel Phthalimide Protection Strategy
Introduction to the Novel Phthalimide Protection Strategy
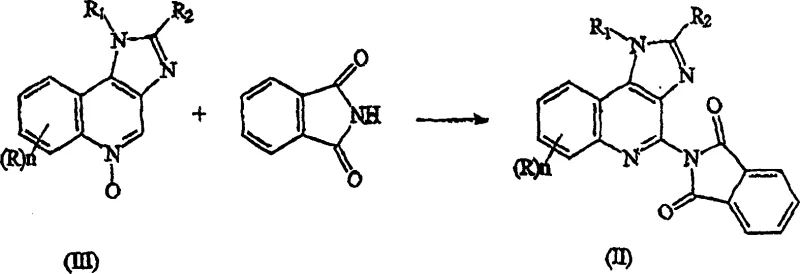
The pharmaceutical industry constantly seeks robust synthetic pathways that balance efficiency with safety, particularly for high-volume immunomodulators like Imiquimod. Patent CN1681814A introduces a transformative approach to synthesizing 1H-imidazo[4,5-C]quinoline-4-amines by utilizing a novel 1H-imidazo[4,5-C]quinoline-4-phthalimide intermediate. This methodology represents a significant departure from traditional routes that often rely on hazardous reagents or extreme reaction conditions. By employing a phthalimide protecting group strategy, the process effectively masks the reactive amino functionality during the critical cyclization and substitution phases, thereby enhancing control over regioselectivity and minimizing the formation of stubborn by-products. For R&D directors and process chemists, this patent offers a compelling solution to the longstanding challenges associated with introducing the 4-amino group on the quinoline ring system.
![Chemical structure of the key 1H-imidazo[4,5-C]quinoline-4-phthalimide intermediate (Formula II)](/insights/img/imiquimod-phthalimide-intermediate-synthesis-supplier-20260306214354-01.webp)
The core innovation lies in the stability and reactivity profile of the phthalimide intermediate, which serves as a versatile precursor for the final active pharmaceutical ingredient. Unlike direct nucleophilic substitutions that can suffer from poor yields and require rigorous purification, this two-step sequence involving N-oxide activation followed by hydrazinolysis provides a cleaner trajectory towards the target molecule. The ability to isolate and characterize the phthalimide intermediate allows for stringent quality control checkpoints before the final deprotection step, ensuring that only high-quality material proceeds to the final stage. This level of process control is essential for meeting the increasingly strict regulatory standards imposed on generic and branded antiviral agents in global markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-amino-substituted imidazoquinolines has been plagued by significant operational hurdles that impact both cost and safety profiles. Traditional methods often involve the direct nucleophilic displacement of a leaving group, such as chlorine or triflate, using ammonia under high temperature and high pressure conditions. These harsh environments necessitate the use of specialized autoclaves and pose substantial safety risks related to pressurized ammonia handling. Furthermore, alternative routes utilizing azide chemistry, while effective in some contexts, introduce severe safety concerns regarding the potential explosivity of organic azides and the complexity of their subsequent reduction to amines. Such processes often result in broad impurity profiles, including hydroxylated derivatives and unreacted starting materials, which are notoriously difficult to separate from the final API, driving up purification costs and reducing overall throughput.
The Novel Approach
In stark contrast, the methodology disclosed in CN1681814A operates under remarkably mild conditions that mitigate these historical risks. The key coupling reaction between the quinoline N-oxide and phthalimide is conducted at a controlled low temperature range of 0-10°C, eliminating the thermal stress on the molecular framework and preventing degradation. This gentle approach not only preserves the integrity of the sensitive imidazoquinoline core but also simplifies the engineering requirements for the reactor setup, as there is no need for high-pressure containment systems. The subsequent deprotection step utilizes hydrazine hydrate at standard reflux temperatures, a well-understood and easily manageable unit operation in any standard multipurpose pharmaceutical plant. This shift from extreme to moderate conditions fundamentally alters the economic equation of manufacturing, offering a pathway that is inherently safer and more energy-efficient.
Mechanistic Insights into Phthalimide-Mediated Amination
The success of this synthetic route hinges on the precise activation of the phthalimide nucleophile and its subsequent attack on the electrophilic center of the quinoline N-oxide. In the presence of an activating agent like benzoyl chloride and a base such as tri-n-butylamine, the phthalimide is converted into a highly reactive N-acyl species or a similar activated complex. This activated nucleophile then selectively attacks the 4-position of the 1H-imidazo[4,5-C]quinoline-N-oxide, displacing the oxygen functionality in a concerted manner that preserves the aromaticity of the system. The use of the N-oxide is critical here, as it enhances the electrophilicity of the C-4 position, facilitating the substitution without requiring the extreme forcing conditions needed for neutral quinoline substrates. This mechanistic nuance ensures that the reaction proceeds with high regioselectivity, minimizing the risk of attack at other positions on the heterocyclic ring.
Furthermore, the phthalimide group acts as an excellent protecting group that stabilizes the newly formed C-N bond against hydrolysis or other side reactions during workup. The steric bulk and electronic properties of the phthalimide moiety prevent over-reaction or polymerization, which are common pitfalls in direct amination strategies. Once the intermediate is formed, the removal of the phthalimide group via hydrazinolysis is a classic and highly efficient transformation. The hydrazine attacks the carbonyl carbons of the phthalimide ring, leading to the formation of phthalhydrazide and the release of the free primary amine. This cleavage is thermodynamically driven and proceeds to completion, ensuring that the final product is obtained with minimal residual protecting group contaminants, thus simplifying the downstream purification process significantly.
How to Synthesize 1-Isobutyl-1H-Imidazo[4,5-C]quinoline-4-Amine Efficiently
Implementing this synthesis requires careful attention to the activation step and the subsequent hydrazinolysis to maximize yield and purity. The process begins with the preparation of the N-oxide, followed by the critical coupling with phthalimide in a solvent system such as dichloromethane or ethyl acetate. Maintaining the temperature strictly between 0-10°C during the addition of benzoyl chloride is paramount to controlling the exotherm and preventing the formation of hydroxylated by-products. After isolation of the solid phthalimide intermediate, which can be purified by simple resuspension in methanol, the final step involves heating with hydrazine hydrate in water. The detailed standardized operating procedures for scaling this reaction from laboratory to commercial production are outlined below.
- React 1H-imidazo[4,5-C]quinoline-N-oxide with phthalimide and benzoyl chloride in the presence of a base like tri-n-butylamine at 0-10°C to form the phthalimide intermediate.
- Isolate the 1H-imidazo[4,5-C]quinoline-4-phthalimide intermediate through filtration and washing with dichloromethane and methanol to ensure high purity.
- Perform hydrazinolysis by reacting the isolated phthalimide intermediate with hydrazine hydrate in water at 94-95°C to yield the final 1H-imidazo[4,5-C]quinoline-4-amine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this phthalimide-based route offers tangible benefits that extend beyond mere chemical elegance. The elimination of high-pressure reactors and hazardous azide reagents translates directly into reduced capital expenditure (CAPEX) and lower insurance premiums for manufacturing facilities. The reliance on commodity chemicals like phthalimide, hydrazine, and common organic solvents ensures a stable and resilient supply chain, mitigating the risk of raw material shortages that can plague specialty reagent-dependent processes. Moreover, the high purity of the intermediate allows for more flexible sourcing of starting materials, as the robust purification steps inherent in this route can tolerate slight variations in feedstock quality without compromising the final API specification.
- Cost Reduction in Manufacturing: The process significantly lowers operational costs by removing the need for energy-intensive high-temperature and high-pressure reactions. The mild conditions allow for the use of standard glass-lined or stainless steel reactors, avoiding the specialized metallurgy required for corrosive high-pressure ammonia systems. Additionally, the high selectivity of the reaction reduces the burden on purification units, leading to lower solvent consumption and waste disposal costs, which are major drivers of overall manufacturing expenses in the fine chemical sector.
- Enhanced Supply Chain Reliability: By utilizing widely available reagents such as phthalimide and hydrazine hydrate, the supply chain becomes less vulnerable to geopolitical disruptions or vendor monopolies often associated with exotic catalysts or specialized leaving groups. The robustness of the intermediate also allows for strategic stockpiling; the phthalimide derivative is stable and can be manufactured in large batches during off-peak times, decoupling the production of the intermediate from the final formulation schedule and providing greater flexibility in meeting market demand fluctuations.
- Scalability and Environmental Compliance: The reaction generates fewer hazardous by-products compared to azide-based routes, simplifying wastewater treatment and aligning better with modern environmental regulations. The aqueous workup in the final step reduces the volume of organic waste, and the solid by-product, phthalhydrazide, can often be recovered or disposed of with minimal environmental impact. This green chemistry profile facilitates faster regulatory approvals and reduces the long-term liability associated with hazardous waste management, making it an attractive option for sustainable manufacturing initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. Understanding these nuances is critical for project managers evaluating the feasibility of technology transfer. The answers provided are derived directly from the experimental data and claims within the patent documentation, ensuring accuracy and relevance for decision-making processes.
Q: What are the primary advantages of the phthalimide route over traditional ammonolysis for Imiquimod synthesis?
A: The phthalimide route avoids the need for high-temperature and high-pressure conditions required for direct ammonia substitution, significantly reducing safety risks and energy consumption while improving regioselectivity and product purity.
Q: How does the new method impact the impurity profile of the final API?
A: By utilizing a protected intermediate, the process minimizes side reactions such as hydroxylation at the 4-position, resulting in a cleaner crude product with significantly lower levels of difficult-to-remove impurities compared to direct substitution methods.
Q: Is this synthesis route scalable for commercial production?
A: Yes, the reaction conditions are mild (0-10°C for the coupling step and standard reflux for hydrazinolysis), utilizing common solvents like dichloromethane and water, which makes the process highly amenable to large-scale commercial manufacturing without specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imiquimod Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of adopting advanced synthetic methodologies like the phthalimide protection route to maintain competitiveness in the global antiviral market. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch against the highest industry standards.
We invite you to collaborate with us to leverage this innovative technology for your supply chain needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this route can optimize your total cost of ownership. Please contact us today to request specific COA data and route feasibility assessments, and let us partner with you to secure a reliable and cost-effective supply of critical pharmaceutical intermediates.
