Optimizing Imiquimod Production: A Novel Halogen Exchange and Purification Strategy
Optimizing Imiquimod Production: A Novel Halogen Exchange and Purification Strategy
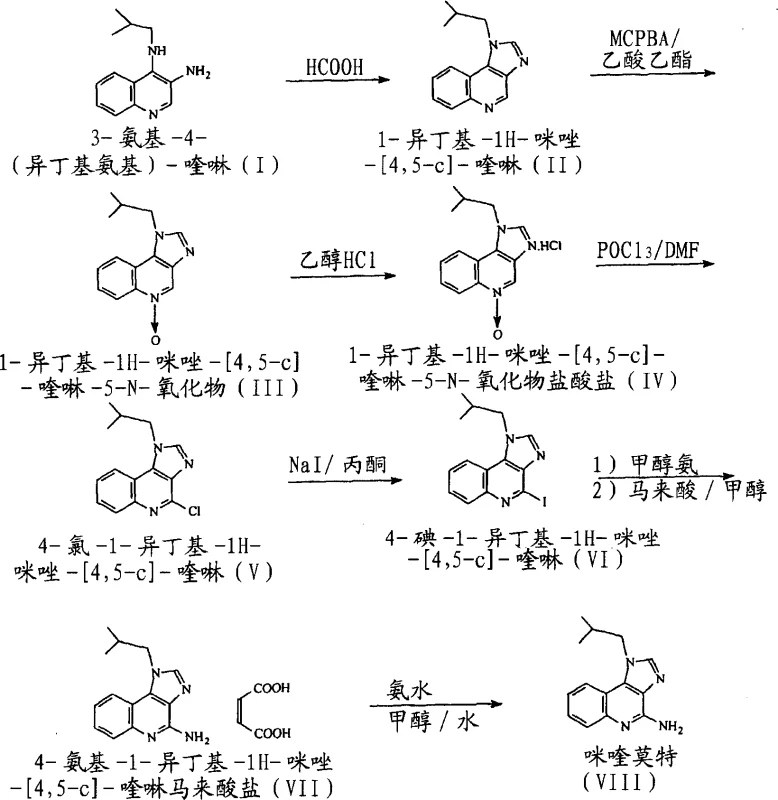
The pharmaceutical industry continuously seeks robust synthetic pathways for immune response modifiers like Imiquimod, specifically 4-amino-1-isobutyl-1H-imidazo[4,5-c]-quinoline. Patent CN101048404A introduces a transformative methodology that addresses long-standing challenges in yield optimization and impurity control during the manufacturing of this critical active pharmaceutical ingredient (API). By leveraging a novel halogen exchange strategy and a unique maleate salt purification protocol, this process establishes a new benchmark for producing high-purity pharmaceutical intermediates. The technical significance of this patent lies in its ability to bypass the harsh conditions typically associated with nucleophilic aromatic substitution, thereby enhancing both safety and scalability for commercial production.
This comprehensive analysis delves into the mechanistic advantages and commercial implications of the disclosed synthesis route. For R&D directors and process chemists, the shift from direct chloro-substitution to an iodo-intermediate pathway represents a significant leap in reaction efficiency. Furthermore, the integration of a maleate salt crystallization step ensures that the final product meets stringent regulatory standards for purity, a critical factor for procurement managers evaluating potential suppliers. As a leading entity in fine chemical manufacturing, understanding these nuanced process improvements is essential for maintaining a competitive edge in the global supply chain of antiviral therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Imiquimod has relied heavily on the direct nucleophilic substitution of a 4-chloro leaving group with ammonia or amine sources. While conceptually straightforward, this conventional approach is plagued by significant kinetic barriers due to the electron-deficient nature of the heterocyclic ring system. Prior art methods, such as those described in various US patents, often necessitate extreme reaction conditions, including high temperatures and elevated pressures in autoclaves, to drive the displacement of the chloride ion. These harsh parameters not only increase energy consumption and operational risks but also promote the formation of degradation by-products and regioisomers that are difficult to separate. Additionally, traditional oxidation steps using peracetic acid in toluene have been reported to suffer from incomplete conversions and inconsistent yields, creating bottlenecks in the upstream supply of key intermediates.
The Novel Approach
The methodology outlined in patent CN101048404A circumvents these kinetic limitations through a strategic halogen exchange maneuver. Instead of forcing the displacement of a chlorine atom, the process converts the 4-chloro intermediate into a 4-iodo derivative using sodium iodide in acetone. Iodine serves as a superior leaving group due to its larger atomic radius and weaker bond strength with carbon, dramatically lowering the activation energy required for the subsequent amination step. This modification allows the introduction of the amino group under comparatively milder conditions, reducing thermal stress on the molecule and minimizing side reactions. Coupled with the use of m-chloroperbenzoic acid (MCPBA) for the initial oxidation and a final purification via maleate salt formation, this novel approach delivers a cleaner reaction profile and superior overall yield compared to legacy technologies.
Mechanistic Insights into Halogen Exchange and Oxidative Activation
The core innovation of this synthesis lies in the precise control of electronic properties at the C-4 position of the imidazoquinoline ring. The initial oxidation of 1-isobutyl-1H-imidazo[4,5-c]-quinoline to its 5-N-oxide using MCPBA in ethyl acetate is a critical activation step. The N-oxide functionality increases the electrophilicity of the adjacent carbon atoms, facilitating the subsequent replacement of the oxygen with a chlorine atom upon treatment with phosphorus oxychloride (POCl3) in dimethylformamide (DMF). This sequence generates the 4-chloro-1-isobutyl-1H-imidazo[4,5-c]-quinoline intermediate, which serves as the pivot point for the novel halogen exchange. The transformation of the chloro-derivative to the iodo-analog proceeds via a Finkelstein-type reaction mechanism, where the equilibrium is driven by the precipitation of sodium chloride in acetone, effectively pushing the reaction towards the formation of the more reactive 4-iodo species.
From an impurity control perspective, this mechanistic pathway offers distinct advantages. The use of MCPBA avoids the radical side reactions often associated with peracetic acid, resulting in a cleaner oxidation profile with fewer tarry by-products. Furthermore, the isolation of intermediates as stable salts, such as the hydrochloride of the N-oxide and the maleate of the final amine, provides multiple opportunities for purification throughout the synthetic sequence. The maleate salt formation, in particular, exploits differences in solubility to exclude structurally similar impurities that might co-elute during standard chromatographic or crystallization processes. This multi-stage purification strategy ensures that the final free base, obtained by neutralizing the maleate salt with ammonia, possesses a purity profile suitable for direct formulation without extensive additional refining.
How to Synthesize 4-Amino-1-isobutyl-1H-imidazo[4,5-c]-quinoline Efficiently
The execution of this synthesis requires careful attention to solvent selection and temperature control to maximize the benefits of the halogen exchange mechanism. The process begins with the cyclization of 3-amino-4-(isobutylamino)-quinoline using formic acid, followed by the critical MCPBA oxidation in ethyl acetate at controlled temperatures between 60-80°C. Following the isolation of the N-oxide hydrochloride, the chlorination step utilizes POCl3 in DMF, after which the crude chloro-intermediate is subjected to sodium iodide in acetone to generate the reactive iodo-species. The final amination is conducted in methanolic ammonia within a pressure reactor, followed by the distinctive maleate salt crystallization from aqueous methanol. Detailed standardized operating procedures for each unit operation are essential to maintain consistency.
- Oxidize 1-isobutyl-1H-imidazo[4,5-c]-quinoline using m-chloroperbenzoic acid in ethyl acetate, followed by isolation as the hydrochloride salt.
- Convert the N-oxide hydrochloride to the 4-chloro derivative using phosphorus oxychloride in DMF, then perform halogen exchange with sodium iodide in acetone.
- React the 4-iodo intermediate with methanolic ammonia under pressure, purify the crude product as the maleate salt, and finally liberate the free base.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented process translates into tangible operational efficiencies and risk mitigation. The shift to a milder amination protocol reduces the dependency on specialized high-pressure equipment capable of withstanding extreme temperatures, thereby lowering capital expenditure requirements for manufacturing facilities. Moreover, the enhanced reactivity of the iodo-intermediate leads to shorter reaction times and higher conversion rates, which directly improves throughput and asset utilization. The robust purification capability of the maleate salt step minimizes the risk of batch rejection due to purity failures, ensuring a more reliable supply of material for downstream formulation teams. These factors collectively contribute to a more resilient and cost-effective supply chain for this high-value pharmaceutical intermediate.
- Cost Reduction in Manufacturing: The elimination of extreme reaction conditions significantly lowers energy consumption and maintenance costs associated with high-pressure reactors. By utilizing a more reactive iodo-intermediate, the process achieves higher yields with fewer recycle loops, reducing the overall consumption of raw materials and solvents. The ability to purify via simple crystallization of the maleate salt avoids the need for expensive and time-consuming chromatographic separations, further driving down the cost of goods sold (COGS) while maintaining high quality standards.
- Enhanced Supply Chain Reliability: The use of commercially available and stable reagents like MCPBA and sodium iodide ensures a consistent supply of inputs without reliance on exotic or hazardous custom chemicals. The improved stability of intermediates, particularly when isolated as salts, allows for greater flexibility in production scheduling and inventory management. This stability reduces the risk of degradation during storage and transport, ensuring that the material delivered to customers meets specifications regardless of logistical delays, thus strengthening the overall reliability of the supply network.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing common organic solvents like ethyl acetate and acetone which are easier to recover and recycle compared to chlorinated solvents often used in older methods. The cleaner reaction profile generates less hazardous waste, simplifying effluent treatment and reducing the environmental footprint of the manufacturing site. This alignment with green chemistry principles not only ensures compliance with increasingly strict environmental regulations but also enhances the sustainability profile of the final product for eco-conscious pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this specific synthetic route. Understanding these details is crucial for technical teams evaluating the feasibility of technology transfer or scale-up. The answers provided are derived directly from the experimental data and claims within the patent documentation, ensuring accuracy and relevance for process development discussions.
Q: Why is the 4-iodo intermediate preferred over the 4-chloro derivative for amination?
A: The 4-iodo derivative exhibits significantly higher reactivity towards nucleophilic substitution by ammonia compared to the chloro analog. This allows the amination step to proceed under milder conditions with improved conversion rates, avoiding the extreme temperatures and pressures often required for direct chloro-displacement.
Q: What is the advantage of using the maleate salt for purification?
A: Formation of the maleate salt provides a highly crystalline solid that can be easily recrystallized from aqueous alcohol mixtures. This step effectively removes trace organic impurities and residual solvents, ensuring the final free base achieves pharmaceutical-grade purity exceeding 99.5% by HPLC.
Q: How does the MCPBA oxidation method compare to traditional peracetic acid routes?
A: Using m-chloroperbenzoic acid (MCPBA) in ethyl acetate offers a cleaner reaction profile with higher selectivity for the N-oxide formation. Unlike peracetic acid methods which can suffer from incomplete reactions and lower yields, the MCPBA route facilitates easier workup and higher isolation yields of the critical N-oxide intermediate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imiquimod Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving demands of the pharmaceutical market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the halogen exchange route for Imiquimod can be seamlessly transitioned from the laboratory to full-scale manufacturing. We are committed to delivering high-purity pharmaceutical intermediates that adhere to stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch.
We invite you to collaborate with us to leverage these process improvements for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing capabilities can enhance your project's success and ensure a steady supply of high-quality Imiquimod intermediates.
