Advanced Industrial Synthesis of Tranexamic Acid via Selective Oxidation and Ammonolysis
The pharmaceutical industry continuously seeks robust synthetic pathways that balance high purity with economic viability, particularly for essential hemostatic agents like tranexamic acid. Patent CN110156620B introduces a groundbreaking preparation method that fundamentally shifts the paradigm from hazardous cyanide-based chemistries to a safer, high-yield oxidation-ammonolysis sequence. This innovative approach utilizes 1,4-cyclohexanedimethanol as a readily accessible starting material, reacting it with hydrohalic acids to form key halo-intermediates before undergoing selective oxidation and subsequent ammonolysis. By leveraging oxygen gas atmospheres and controlled high-pressure conditions, this technology achieves a total yield ranging from 55% to 70%, a substantial improvement over the dismal 13% yields reported in prior art involving bromination and TEMPO oxidation. For R&D directors and procurement specialists, this patent represents a critical opportunity to secure a reliable tranexamic acid supplier capable of delivering high-purity pharmaceutical intermediates without the baggage of toxic waste streams or prohibitive raw material costs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of tranexamic acid has been plagued by significant safety hazards and inefficiencies that hinder cost reduction in pharmaceutical intermediates manufacturing. The traditional methyl acrylate method, for instance, relies heavily on highly toxic cyanide reagents and heavy metal copper catalysts, creating severe environmental pollution and demanding complex waste treatment protocols that inflate operational expenditures. Alternative routes utilizing p-aminomethyl benzoic acid require expensive noble metal catalysts such as platinum, rhodium, or ruthenium for catalytic hydrogenation, which not only drives up the cost of goods sold but also introduces challenges in removing trace metal impurities to meet stringent regulatory standards. Furthermore, existing methods based on 1,4-cyclohexanedimethanol, such as those disclosed in Indian patent IN2010CH03367, suffer from poor reaction selectivity during the initial halogenation step, leading to a proliferation of by-products and necessitating rigorous recrystallization steps that decimate the overall yield to merely 13%. These legacy processes create bottlenecks in the supply chain, resulting in inconsistent batch quality and extended lead times for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast, the novel methodology described in CN110156620B offers a streamlined, three-step trajectory that maximizes atom economy and operational simplicity. The process initiates with a highly selective halogenation of 1,4-cyclohexanedimethanol using HX acid, where reaction conditions are meticulously tuned to prevent the formation of dihalogenated by-products, thereby preserving the integrity of the mono-substituted intermediate. This is followed by an elegant oxidation step utilizing sodium nitrite under a controlled oxygen atmosphere, which efficiently converts the alcohol moiety to a carboxylic acid without the need for stoichiometric amounts of hazardous oxidants. The final stage involves a high-pressure ammonolysis reaction that directly installs the amine functionality, followed by a straightforward alkali transformation to yield the target tranexamic acid. This route not only bypasses the need for toxic cyanides and precious metals but also demonstrates exceptional scalability, making it an ideal candidate for the commercial scale-up of complex pharmaceutical additives.
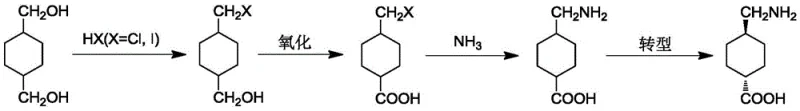
Mechanistic Insights into Selective Aerobic Oxidation and Ammonolysis
The core chemical innovation lies in the precise control of the oxidation and ammonolysis mechanisms, which dictates the final purity and stereochemical outcome of the API intermediate. During the oxidation phase, the introduction of gas with an oxygen content of 21-100% acts as the terminal oxidant in the presence of sodium nitrite, facilitating a catalytic cycle that selectively targets the primary alcohol group while leaving the halomethyl group intact. This selectivity is paramount; uncontrolled oxidation could lead to over-oxidation or degradation of the sensitive cyclohexane ring, generating difficult-to-remove impurities that would compromise the fibrinolytic activity of the final drug substance. The reaction temperature is maintained between 0-50°C, a range that kinetically favors the formation of the desired 4-halomethyl cyclohexyl formic acid while suppressing side reactions. Following this, the ammonolysis step leverages high-pressure conditions (0.1-4.0 MPa) and elevated temperatures (90-130°C) to drive the nucleophilic substitution of the halogen atom by ammonia. This high-energy environment ensures complete conversion of the halo-acid to the amino-acid precursor, minimizing the retention of unreacted starting materials that often plague lower-pressure batch processes.
Impurity control is further enhanced by the inherent selectivity of the initial halogenation step, which avoids the generation of dihalogenated species that are common in radical halogenation processes. By utilizing specific molar ratios of 1,4-cyclohexanedimethanol to HX acid (preferably 1:2-3), the process ensures that the mono-halogenated intermediate is the dominant species in the reaction mixture. This purity at the early stage cascades through the subsequent oxidation and ammonolysis steps, significantly reducing the burden on downstream purification units. For the trans-specific route, starting with pure trans-1,4-cyclohexanedimethanol allows for the direct synthesis of the biologically active trans-tranexamic acid without the need for a final isomerization or resolution step, which is typically a major source of yield loss in other synthetic strategies. This mechanistic elegance translates directly into a more robust and predictable manufacturing process.
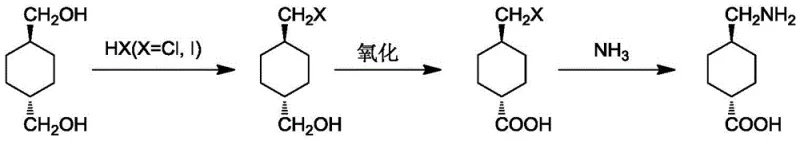
How to Synthesize Tranexamic Acid Efficiently
The implementation of this synthesis route requires careful attention to reaction parameters, particularly during the high-pressure ammonolysis and the aerobic oxidation stages. Operators must ensure strict control over the oxygen flow rate and pressure to maintain the optimal oxidation potential without risking safety incidents associated with oxygen-enriched atmospheres. Similarly, the ammonolysis step demands specialized high-pressure reactor equipment capable of withstanding the corrosive nature of ammonia at elevated temperatures. The following guide outlines the standardized operational framework derived from the patent examples, providing a clear roadmap for process engineers to replicate these high-yield results in a pilot or production setting. Detailed standardized synthesis steps are provided in the guide below.
- React 1,4-cyclohexanedimethanol with HX acid (Cl or I) at 50-100°C to generate 4-halomethyl cyclohexyl methanol with high selectivity.
- Oxidize the intermediate using sodium nitrite under an oxygen atmosphere (21-100% O2) at 0-50°C to form the corresponding carboxylic acid.
- Perform ammonolysis in a high-pressure reactor with liquid ammonia or ammonia water at 90-130°C, followed by alkali transformation to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers transformative benefits that extend far beyond simple yield improvements. The elimination of toxic cyanide reagents and expensive noble metal catalysts fundamentally alters the cost structure of tranexamic acid production, removing significant line items related to hazardous waste disposal and catalyst recovery. This shift enables a drastic simplification of the supply chain, as the reliance on specialized, high-cost reagents is replaced by commodity chemicals like hydrochloric acid, sodium nitrite, and ammonia, which are globally available and subject to less volatile pricing dynamics. Consequently, manufacturers can achieve substantial cost savings in API intermediate manufacturing while simultaneously mitigating the risk of supply disruptions caused by the scarcity of exotic catalysts. The robustness of the process also implies a more reliable tranexamic acid supplier profile, capable of meeting large-volume demands with consistent quality.
- Cost Reduction in Manufacturing: The economic advantage of this process is driven primarily by the substitution of high-value inputs with low-cost commodities. By avoiding the use of platinum, rhodium, or ruthenium catalysts, the capital tied up in catalyst inventory is liberated, and the recurring cost of catalyst replenishment is eliminated entirely. Furthermore, the high selectivity of the halogenation step means that raw material utilization is maximized, with minimal loss to dihalogenated by-products that would otherwise represent a sunk cost. The simplified work-up procedures, which do not require complex extraction or chromatographic purification to remove heavy metals, further reduce the consumption of solvents and energy, leading to a leaner and more cost-effective production model that enhances overall profit margins.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of 1,4-cyclohexanedimethanol as the foundational building block. Unlike specialized precursors that may have limited global suppliers, this diol is a mature chemical product with a stable and diversified supply base. The reagents used in the subsequent steps, such as sodium nitrite and ammonia, are produced on a massive industrial scale worldwide, ensuring that production schedules are not held hostage by the availability of niche chemicals. This ubiquity of raw materials allows for strategic stocking and long-term contracting, effectively insulating the manufacturing process from market fluctuations and ensuring continuous production capacity even during periods of global supply chain stress.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this route is exceptionally well-suited for expansion from pilot batches to multi-ton commercial production. The absence of cyanide removes a major regulatory hurdle, simplifying the permitting process for new production lines and reducing the liability associated with handling extremely toxic substances. The waste streams generated are predominantly aqueous salts and organic solvents that are easier to treat compared to heavy metal-laden sludge, aligning with increasingly stringent global environmental regulations. This compliance ease facilitates faster scale-up timelines, allowing companies to respond rapidly to market demand surges for hemostatic agents without the delays typically associated with environmental impact assessments for hazardous chemistries.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this tranexamic acid synthesis technology. These answers are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing clarity on yield expectations, safety profiles, and scalability. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for their own manufacturing portfolios or for procurement teams assessing the long-term viability of suppliers utilizing this method.
Q: How does this synthesis route improve upon traditional cyanide-based methods?
A: This novel route eliminates the use of highly toxic cyanide and heavy metal copper catalysts required in the methyl acrylate method. By utilizing a selective halogenation and aerobic oxidation strategy, it significantly reduces environmental pollution and safety risks while achieving a total yield of 55-70%, compared to merely 13% in older bromination-oxidation protocols.
Q: What are the key advantages regarding raw material availability and cost?
A: The process utilizes 1,4-cyclohexanedimethanol as a starting material, which is cheap and easily obtained commercially. Furthermore, the avoidance of expensive noble metal catalysts (like Pt, Rh, Ru) used in hydrogenation methods drastically lowers the production cost, making it highly suitable for large-scale industrial manufacturing.
Q: Is this method scalable for commercial API production?
A: Yes, the method is explicitly designed for industrial scalability. The reactions involve standard unit operations such as halogenation, oxidation with gas introduction, and high-pressure ammonolysis. The high selectivity minimizes by-product formation, simplifying purification and ensuring consistent quality suitable for pharmaceutical grade standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tranexamic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of deploying efficient and safe synthetic routes to meet the growing global demand for essential pharmaceutical ingredients. Our team of expert process chemists has extensively evaluated the oxidation-ammonolysis pathway described in CN110156620B and possesses the technical expertise to optimize this chemistry for your specific production needs. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped with high-pressure reactors and advanced QC labs capable of maintaining stringent purity specifications, guaranteeing that every batch of tranexamic acid meets the rigorous standards required for API applications.
We invite you to collaborate with us to leverage this advanced technology for your supply chain optimization initiatives. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this cyanide-free route for your specific volume requirements. We encourage you to reach out today to obtain specific COA data from our recent pilot runs and to discuss route feasibility assessments tailored to your project timelines. Let us help you secure a sustainable, cost-effective, and high-quality supply of tranexamic acid that supports your long-term business goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
