Scalable Copper-Catalyzed Synthesis of Montelukast Sodium Intermediates for Global Pharma Supply Chains
The pharmaceutical industry continuously seeks robust synthetic pathways that balance high purity with economic viability, particularly for high-volume asthma medications like Montelukast Sodium. Patent CN112724082B introduces a transformative preparation method for the critical intermediate (E)-2-[3-[3-[2-(7-chloro-2-quinolinyl)vinyl]phenyl]-3-oxopropyl]benzoate, addressing long-standing inefficiencies in traditional manufacturing. This innovation replaces expensive noble metal catalysts with accessible copper-based systems, fundamentally altering the cost structure and environmental footprint of the supply chain. By leveraging mild reaction conditions between 80°C and 150°C, the process ensures exceptional selectivity, minimizing hazardous by-products while maximizing throughput. For global procurement leaders, this represents a strategic opportunity to secure a reliable montelukast sodium intermediate supplier capable of delivering consistent quality at reduced operational expenditures.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this key pharmaceutical building block relied heavily on palladium-catalyzed cross-coupling reactions, which, while effective, impose severe economic and logistical burdens on large-scale production. As illustrated in the prior art reaction scheme, the conventional route utilizes palladium catalysts to couple (E)-1-(3-(2-(7-chloro-2-quinolinyl)vinylphenyl)-2-propen-1-ol with methyl 2-iodobenzoate. Although chemically feasible, the reliance on palladium introduces significant volatility in raw material costs due to the scarcity of this precious metal. Furthermore, the removal of residual palladium from the final active pharmaceutical ingredient (API) requires complex and costly purification steps to meet strict regulatory limits on heavy metals. The difficulty in recovering and recycling the palladium catalyst further exacerbates waste generation, creating a bottleneck for sustainable commercial scale-up of complex pharmaceutical intermediates.
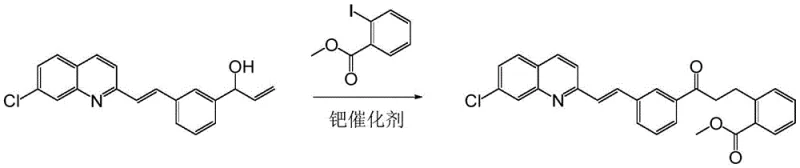
The Novel Approach
In stark contrast, the methodology disclosed in CN112724082B utilizes copper halides such as CuCl, CuBr, or CuI to drive the coupling reaction with remarkable efficiency. This novel approach eliminates the dependency on precious metals, substituting them with abundant and inexpensive copper sources that do not compromise reaction kinetics. The reaction proceeds smoothly in solvents like N-methylpyrrolidone or toluene, facilitated by common bases such as potassium carbonate or triethylamine. Crucially, the copper catalyst can be recovered through simple filtration after the reaction is complete, allowing for multiple reuse cycles without significant degradation in performance. This shift not only drastically simplifies the downstream processing workflow but also aligns with green chemistry principles by reducing heavy metal waste, making it an ideal solution for cost reduction in pharmaceutical intermediates manufacturing.
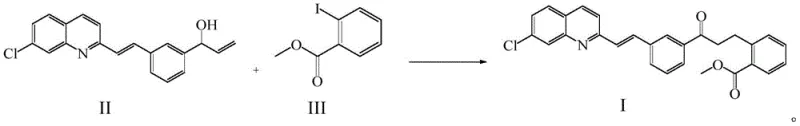
Mechanistic Insights into Copper-Catalyzed Coupling
The success of this synthesis hinges on the precise optimization of reaction parameters, particularly temperature control, which governs the activation energy and selectivity of the copper-catalyzed cycle. The patent specifies a critical temperature window of 80°C to 150°C, with an optimal range of 90°C to 120°C, to balance reaction rate against thermal degradation. Operating below this threshold results in incomplete conversion even with extended reaction times, leading to lower yields and accumulation of unreacted starting materials. Conversely, excessive temperatures promote the formation of undesired side products and decomposition of sensitive functional groups, compromising the purity profile required for downstream API synthesis. Maintaining the reaction at approximately 100°C ensures that the copper species remain active throughout the 8 to 10-hour duration, facilitating the efficient formation of the carbon-carbon bond between the quinoline and benzoate moieties.
Impurity control is another cornerstone of this mechanistic design, achieved through the careful selection of stoichiometry and solvent systems. The molar ratio of the alcohol substrate to the iodobenzoate is maintained between 1.0:1 and 1.8:1, preferably 1.4:1, to drive the equilibrium toward the desired ketone product while minimizing homocoupling side reactions. The use of polar aprotic solvents like N-methylpyrrolidone enhances the solubility of the inorganic base and the copper catalyst, ensuring a homogeneous reaction environment that promotes consistent kinetics. Post-reaction workup involves a sophisticated crystallization protocol where the crude product is washed with ethanol and subjected to water-methanol treatments, effectively stripping away trace organic impurities and residual catalyst. This rigorous purification strategy guarantees a final purity exceeding 99%, satisfying the stringent specifications demanded by high-purity pharmaceutical intermediates.
How to Synthesize (E)-2-[3-[3-[2-(7-chloro-2-quinolinyl)vinyl]phenyl]-3-oxopropyl]benzoate Efficiently
To implement this technology in a pilot or production setting, operators must adhere to strict inert atmosphere protocols and precise thermal regulation to replicate the high yields reported in the patent literature. The process begins with the uniform mixing of the quinoline alcohol derivative, methyl 2-iodobenzoate, base, and copper catalyst under nitrogen protection to prevent oxidative deactivation of the catalytic species. Detailed standard operating procedures regarding addition rates, heating ramps, and filtration techniques are essential to ensure reproducibility across different batch sizes. For a comprehensive breakdown of the exact reagent quantities, timing, and isolation steps required to achieve >90% yield, please refer to the standardized synthesis guide below.
- Mix Compound II, Compound III, base (e.g., K2CO3), and CuI catalyst in a solvent like N-methylpyrrolidone under nitrogen protection.
- Heat the reaction mixture to a controlled temperature between 80°C and 150°C, preferably 100°C, and maintain for 8 to 10 hours.
- Filter to recover the catalyst, crystallize the filtrate, and purify the crude product via water/methanol washing to achieve >99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the transition from palladium to copper catalysis offers profound advantages that extend far beyond simple reagent substitution. This technological pivot directly addresses the volatility of precious metal markets, insulating the supply chain from price spikes that frequently disrupt production schedules for API manufacturers. By utilizing a catalyst that is both inexpensive and recyclable, production facilities can significantly reduce their overall cost of goods sold (COGS) without investing in new infrastructure. The ability to recover the catalyst via simple filtration means that waste disposal costs are minimized, and the environmental compliance burden is lightened, facilitating smoother regulatory audits and faster time-to-market for generic formulations.
- Cost Reduction in Manufacturing: The substitution of palladium with copper halides represents a fundamental shift in the economic model of intermediate production, eliminating the need for expensive noble metals that often account for a disproportionate share of raw material costs. Since copper is orders of magnitude cheaper than palladium and does not require complex ligand systems to function effectively, the direct material expenditure is drastically lowered. Furthermore, the catalyst's ability to be reused multiple times without replenishment means that the effective cost per kilogram of product decreases with every subsequent batch, compounding the financial benefits over the lifecycle of the drug product.
- Enhanced Supply Chain Reliability: Relying on abundant base metals like copper mitigates the geopolitical and supply risks associated with scarce precious metals, ensuring a more stable and predictable supply of critical reagents. The simplified purification process, which avoids the need for specialized scavengers to remove trace palladium, reduces the dependency on secondary suppliers and shortens the overall lead time for high-purity pharmaceutical intermediates. This robustness allows manufacturers to maintain higher inventory turnover rates and respond more agilely to fluctuations in market demand for asthma medications, securing the continuity of supply for downstream API producers.
- Scalability and Environmental Compliance: The mild reaction conditions and straightforward workup procedures make this process inherently scalable from laboratory benchtop to multi-ton industrial reactors without encountering the heat transfer or mixing limitations often seen with heterogeneous palladium systems. The reduction in heavy metal waste aligns with increasingly stringent global environmental regulations, reducing the liability and cost associated with hazardous waste treatment and disposal. This eco-friendly profile not only enhances the corporate sustainability metrics of the manufacturer but also simplifies the regulatory filing process for new drug applications by presenting a cleaner, more controlled impurity profile.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this copper-catalyzed synthesis route, derived directly from the experimental data and claims within the patent documentation. These insights are intended to clarify the operational feasibility and quality benchmarks associated with this advanced manufacturing technique for R&D and quality assurance teams evaluating potential technology transfers.
Q: Why is copper catalysis preferred over palladium for this intermediate?
A: Copper catalysts (CuCl, CuBr, CuI) are significantly more cost-effective than palladium and can be easily recovered and reused multiple times without significant loss of activity, whereas palladium is expensive and difficult to recycle.
Q: What is the expected purity and yield of this synthesis method?
A: The patented process consistently achieves yields exceeding 90% and purity levels greater than 99%, meeting stringent pharmaceutical quality standards.
Q: Can the catalyst be recycled in industrial production?
A: Yes, the copper catalyst can be recovered via simple filtration after the reaction and reused for multiple cycles (demonstrated up to 5 times) while maintaining high catalytic activity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Montelukast Sodium Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the adoption of innovative synthetic routes like the copper-catalyzed method described in CN112724082B requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless and risk-free. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify that every batch of intermediate meets the >99% purity threshold required for GMP-compliant API synthesis.
We invite global pharmaceutical partners to engage with our technical procurement team to discuss how this cost-optimized route can enhance your supply chain resilience and profitability. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic impact of switching to this copper-based methodology for your specific production volumes. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project timelines, ensuring that your Montelukast Sodium supply remains competitive and uninterrupted in a dynamic global market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
