Advanced Suzuki Coupling Strategy for High-Purity Aprepitant Intermediate Manufacturing
Advanced Suzuki Coupling Strategy for High-Purity Aprepitant Intermediate Manufacturing
The pharmaceutical industry continuously seeks robust synthetic routes that balance high purity with operational safety, particularly for complex chiral intermediates used in antiemetic therapies. Patent CN111004190A introduces a significant technological advancement in the preparation of a key morpholine derivative, specifically (2R,3S)-2-[(1R)-1-[3,5-bis(trifluoromethyl)phenyl]ethoxy]-3-(4-fluorophenyl)morpholine hydrochloride, which serves as the critical mother nucleus for the synthesis of Aprepitant. This NK-1 receptor antagonist is a vital medication for managing chemotherapy-induced nausea and vomiting, and its market demand necessitates a supply chain capable of delivering high-quality intermediates consistently. The disclosed invention replaces hazardous and sensitive traditional methodologies with a streamlined Suzuki-Miyaura cross-coupling approach, thereby addressing long-standing challenges regarding environmental control, byproduct management, and process scalability that have historically plagued the manufacturing of this specific chemical entity.
Aprepitant, chemically defined as 5-[[ (2R,3S)-2-[(1R)-1-[3,5-bis(trifluoromethyl)phenyl]ethoxy]-3-(4-fluorophenyl)-4-morpholinyl]methyl]-1,2-dihydro-3H-1,2,4-triazole-3-one, relies heavily on the stereochemical integrity of its morpholine precursor. The structural complexity involves three chiral centers that must be meticulously preserved throughout the synthesis to ensure biological efficacy. As illustrated in the molecular architecture, the presence of multiple trifluoromethyl groups and a fluorophenyl ring adds layers of synthetic difficulty, requiring precise control over reaction conditions to prevent defluorination or racemization. The ability to produce this intermediate with high optical purity and minimal impurities is not merely a technical achievement but a commercial imperative for any reliable pharmaceutical intermediate supplier aiming to support global API production without compromising on regulatory standards or patient safety profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
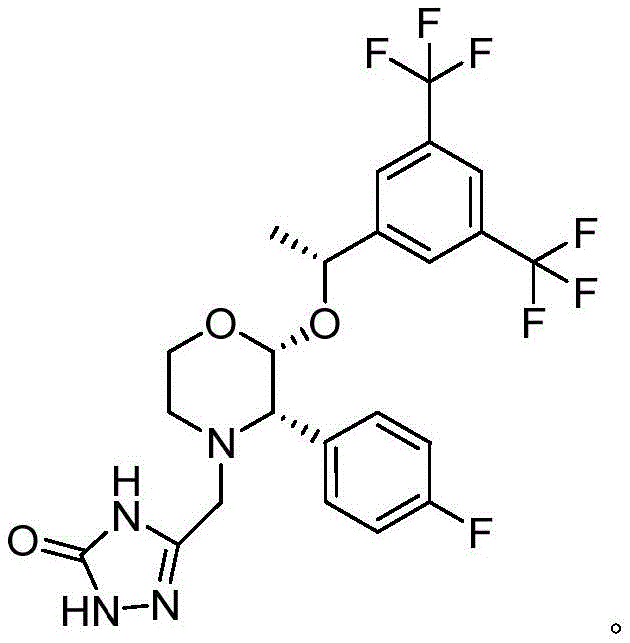
Historically, the synthesis of this pivotal intermediate has relied on a Grignard reaction pathway followed by catalytic hydrogenation, a process fraught with significant operational hazards and purity limitations that hinder efficient commercial scale-up. The conventional route typically involves the generation of a Grignard reagent from p-bromofluorobenzene, which necessitates strictly anhydrous and anaerobic environments to prevent reagent decomposition, thereby imposing severe constraints on reactor design and operational flexibility in a large-scale manufacturing setting. Furthermore, the subsequent hydrogenation step often utilizes p-toluenesulfonic acid as a pH regulator, which introduces the risk of forming potentially genotoxic sulfonate impurities that require rigorous and costly purification protocols to eliminate before the material can proceed to the next stage of API synthesis. Additionally, the intermediate compound generated prior to salification in this traditional route exhibits poor stability, leading to the unavoidable accumulation of excessive impurities even when employing one-pot continuous operation modes, ultimately compromising the overall yield and purity of the final active pharmaceutical ingredient.
The Novel Approach
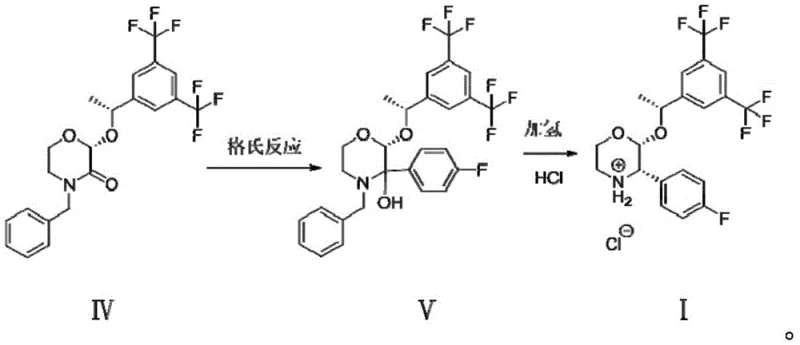
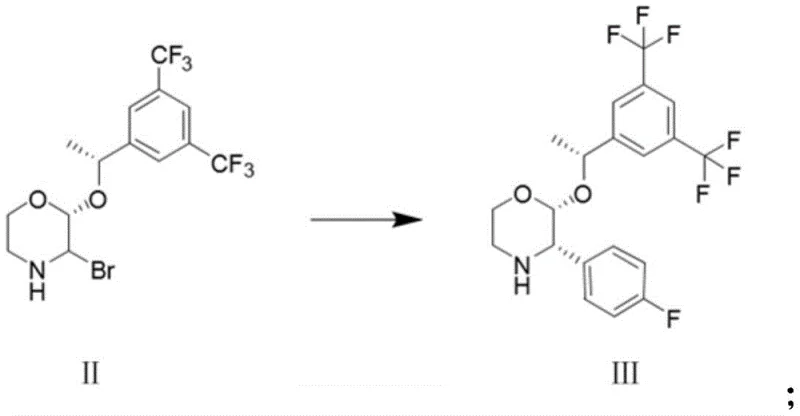
In stark contrast to the precarious nature of the Grignard methodology, the novel approach detailed in the patent utilizes a Suzuki coupling reaction that fundamentally alters the risk profile and efficiency of the manufacturing process. By reacting (2R)-2-[(1R)-1-[3,5-bis(trifluoromethyl)phenyl]ethoxy]-3-bromomorpholine with 4-fluorophenylboronic acid in the presence of a palladium catalyst, the process completely bypasses the need for sensitive organometallic reagents and dangerous high-pressure hydrogenation steps. This strategic shift allows the reaction to proceed under much more forgiving conditions where environmental humidity and oxygen content do not require stringent control, thus drastically simplifying the engineering requirements for production facilities and enhancing the inherent safety of the operation. The result is a cleaner reaction profile with significantly fewer byproducts, particularly avoiding the troublesome defluorinated impurities that are characteristic of the older methods, thereby ensuring a higher quality output that is easier to purify and more suitable for direct use in downstream API condensation reactions.
Mechanistic Insights into Pd-Catalyzed Suzuki Coupling
The core of this technological breakthrough lies in the mechanistic elegance of the palladium-catalyzed cross-coupling cycle, which facilitates the formation of the carbon-carbon bond between the morpholine scaffold and the fluorophenyl ring with exceptional stereocontrol. The reaction initiates with the oxidative addition of the palladium catalyst to the carbon-bromine bond of the morpholine substrate, followed by transmetallation with the activated boronic acid species in the presence of a base such as saturated sodium carbonate solution. This mechanism is inherently robust against moisture and oxygen compared to Grignard chemistry, allowing for a wider operating window that is crucial for maintaining consistent quality across different batch sizes. The choice of ligand and catalyst system, such as [1,1'-bis(diphenylphosphino)ferrocene]palladium dichloride, plays a pivotal role in stabilizing the catalytic cycle and preventing the leaching of palladium or the formation of homocoupling byproducts, ensuring that the chiral centers at the 2 and 3 positions of the morpholine ring remain intact throughout the transformation.
Impurity control is another critical aspect where this mechanism offers distinct advantages, specifically regarding the suppression of defluorination side reactions that plague alternative synthetic routes. In the conventional hydrogenation process, the conditions required to reduce the intermediate often lead to the cleavage of the carbon-fluorine bond, generating defluorinated analogs that are structurally similar to the desired product and notoriously difficult to separate via crystallization or chromatography. The Suzuki coupling conditions, operating at moderate temperatures between 80°C and 100°C in a biphasic system of toluene and aqueous base, provide a kinetic environment that favors the desired cross-coupling over bond cleavage. Furthermore, the subsequent salification step using hydrochloric acid in a solvent like methyl isobutyl ketone acts as a final polishing operation, where the specific solubility characteristics of the hydrochloride salt allow for the exclusion of remaining non-ionic organic impurities, resulting in a final product with HPLC purity levels that meet the stringent specifications required for GMP manufacturing of oncology supportive care drugs.
How to Synthesize Aprepitant Intermediate Efficiently
The practical execution of this synthesis involves a carefully optimized sequence of mixing, heating, and separation steps designed to maximize yield while minimizing waste and operational complexity. The process begins with the uniform mixing of the bromo-morpholine starting material, 4-fluorophenylboronic acid, and the base solution, followed by the addition of the palladium catalyst and heating to reflux in toluene to drive the coupling reaction to completion. Once the reaction is finished, the workup procedure is remarkably straightforward, involving cooling, filtration to recover the precious metal catalyst for potential recycling, and phase separation to isolate the organic layer containing the crude product. For a detailed breakdown of the specific parameters including molar ratios, solvent volumes, and temperature profiles that ensure optimal performance, please refer to the standardized protocol below.
- Perform Suzuki coupling reaction between (2R)-2-[(1R)-1-[3,5-bis(trifluoromethyl)phenyl]ethoxy]-3-bromomorpholine and 4-fluorophenylboronic acid using a palladium catalyst in toluene with saturated sodium carbonate.
- Separate the organic phase, wash with saturated brine, and concentrate to obtain the free base compound (2R,3S)-2-[(1R)-1-[3,5-bis(trifluoromethyl)phenyl]ethoxy]-3-(4-fluorophenyl)morpholine.
- Dissolve the free base in an organic solvent such as methyl isobutyl ketone, adjust pH to 1-2 with hydrochloric acid, concentrate, and cool to crystallize the final hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this Suzuki-based methodology translates directly into tangible improvements in supply reliability and cost structure, primarily driven by the elimination of high-risk unit operations. The removal of the hydrogenation step not only mitigates the safety hazards associated with high-pressure hydrogen gas but also eliminates the need for specialized hydrogenation reactors and the associated maintenance and certification costs, leading to a leaner and more flexible production infrastructure. Moreover, the tolerance of the Suzuki reaction to ambient moisture and oxygen means that the process does not require expensive inert gas blanketing systems or rigorously dried solvents, which significantly reduces the utility consumption and raw material preparation costs associated with maintaining anhydrous conditions. These factors combined create a manufacturing process that is inherently more resilient to supply chain disruptions and equipment failures, ensuring a more consistent flow of materials to the API production line.
- Cost Reduction in Manufacturing: The economic benefits of this route are substantial, stemming from the simplification of the process flow and the reduction in raw material costs associated with avoiding exotic reagents. By eliminating the Grignard reagent preparation, the process saves on the cost of magnesium metal and the energy-intensive drying procedures required for solvents and glassware, while the avoidance of hydrogenation removes the capital expenditure for high-pressure vessels and the operational costs of hydrogen gas. Additionally, the higher purity of the crude product reduces the burden on downstream purification steps, meaning less solvent is consumed for recrystallization and less material is lost during waste treatment, contributing to a lower overall cost of goods sold for the intermediate.
- Enhanced Supply Chain Reliability: From a logistics and planning perspective, the robustness of this chemistry ensures that production schedules are less likely to be derailed by environmental factors or minor deviations in process parameters. The ability to operate without strict anaerobic conditions means that batch turnover times can be optimized, as there is no need for prolonged purging cycles or complex leak testing of reactors, allowing for faster campaign changes and more responsive fulfillment of customer orders. This operational agility is crucial for maintaining continuity of supply in the volatile pharmaceutical market, where delays in intermediate delivery can cascade into significant bottlenecks for final drug product manufacturing and market availability.
- Scalability and Environmental Compliance: The environmental footprint of this process is markedly smaller than that of the conventional route, aligning with the increasing regulatory pressure for greener chemical manufacturing. The absence of sulfonate impurities reduces the toxicity of the waste stream, simplifying wastewater treatment and lowering the costs associated with hazardous waste disposal. Furthermore, the scalability of the Suzuki reaction is well-documented in the industry, and the specific conditions outlined in the patent demonstrate excellent performance at larger scales without the exothermic runaway risks often associated with Grignard additions, making it an ideal candidate for multi-ton production campaigns that meet both economic and sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, providing clarity on how this technology integrates into existing manufacturing frameworks. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, offering a transparent view of the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of technology transfer and for commercial teams negotiating supply agreements based on realistic performance metrics.
Q: Why is the Suzuki coupling method preferred over the traditional Grignard route for this intermediate?
A: The Suzuki coupling method eliminates the need for strict anhydrous and anaerobic conditions required by Grignard reagents, significantly improving operational safety and simplifying industrial scale-up while reducing the formation of defluorinated impurities.
Q: How does this new process address safety concerns related to hydrogenation?
A: Unlike the conventional route which requires catalytic hydrogenation posing explosion risks and potential genotoxic impurity formation from sulfonates, this novel method avoids hydrogenation entirely, relying instead on a robust cross-coupling reaction followed by simple acid salification.
Q: What represents the critical quality attribute for this intermediate in the final API synthesis?
A: The critical quality attribute is the minimization of defluorinated impurities, which are difficult to remove in downstream processing; the Suzuki route described in patent CN111004190A demonstrates superior control over these specific byproducts compared to prior art methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aprepitant Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes like the Suzuki coupling method described in CN111004190A requires a partner with deep technical expertise and proven scale-up capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel chemistry are fully realized in a GMP-compliant manufacturing environment. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the absence of critical impurities such as defluorinated byproducts, guaranteeing that every batch of intermediate we supply meets the exacting standards required for oncology drug development.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can enhance your supply chain resilience and reduce your overall manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic impact of switching to this safer and more efficient process. We encourage potential partners to contact us directly to obtain specific COA data from our pilot batches and to receive comprehensive route feasibility assessments tailored to your specific production volume requirements and timeline constraints.
