Advanced Catalytic Transfer Hydrogenation for Scalable Aprepitant Intermediate Manufacturing
Introduction to Next-Generation Aprepitant Synthesis
The pharmaceutical landscape for antiemetic therapies continues to evolve, with Aprepitant standing as a cornerstone NK1 receptor antagonist for managing chemotherapy-induced nausea and vomiting. The commercial viability of this critical API hinges on the efficient production of its complex chiral intermediates. Patent CN112174909A introduces a transformative preparation method for the key aprepitant intermediate, addressing long-standing challenges in stereocontrol and process safety. This innovation utilizes a sophisticated biomimetic catalytic system to achieve asymmetric transfer hydrogenation, bypassing the need for hazardous high-pressure hydrogenation equipment traditionally employed in this sector.
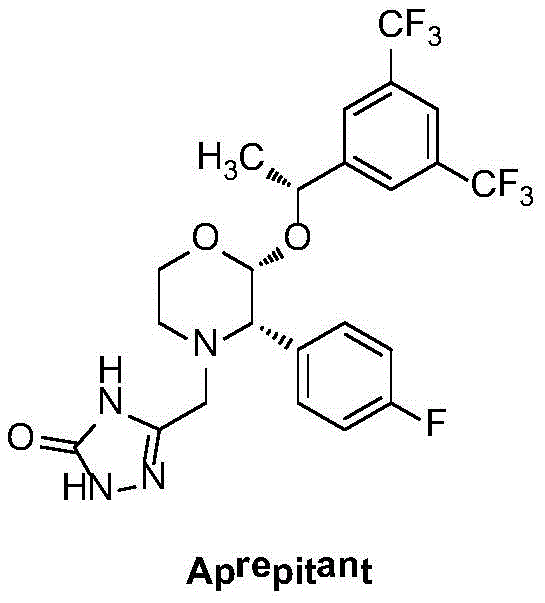
The target molecule, Aprepitant, possesses three distinct chiral centers, making its synthesis a formidable challenge for process chemists aiming for high optical purity. The intermediate described in this patent retains these critical stereocenters, serving as the pivotal building block for the final drug substance. By leveraging a novel chiral catalyst derived from 1,4-dihydropyridine scaffolds, this method offers a robust pathway that aligns with modern green chemistry principles while ensuring the stringent quality standards required by global regulatory bodies for reliable pharmaceutical intermediate supplier partnerships.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this specific morpholine-based intermediate has relied heavily on asymmetric hydrogenation utilizing high-pressure molecular hydrogen as the reductant. While effective in laboratory settings, these conventional routes impose severe constraints on industrial scalability. The requirement for specialized high-pressure reactors significantly increases capital expenditure (CAPEX) and operational complexity, creating bottlenecks in supply chain continuity. Furthermore, the use of transition metal catalysts, often based on precious metals like rhodium or ruthenium, introduces the risk of heavy metal contamination in the final product.
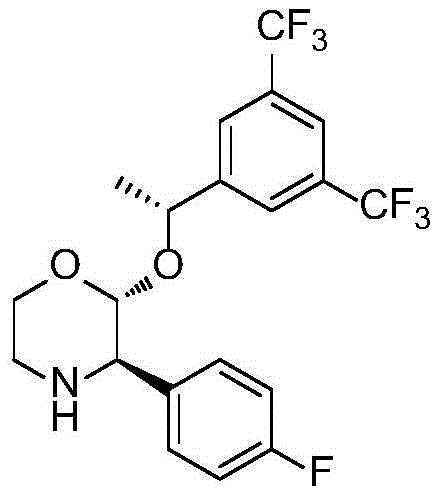
Removing these trace metal impurities to meet pharmacopeial limits necessitates additional downstream processing steps, such as scavenging and extensive purification, which invariably erode overall yield and drive up manufacturing costs. The harsh reaction conditions associated with high-pressure hydrogenation also pose safety risks, complicating hazard assessments and limiting the flexibility of production facilities to adapt to varying demand volumes without significant infrastructure investment.
The Novel Approach
The methodology disclosed in CN112174909A represents a paradigm shift by employing a biomimetic transfer hydrogenation strategy. Instead of gaseous hydrogen, the system utilizes a chiral 1,4-dihydropyridine derivative as a hydrogen donor, mimicking the function of the coenzyme NADH in biological systems. This approach allows the reduction of the imine substrate (Formula 1) to the amine intermediate (Formula 2) under remarkably mild conditions, specifically at temperatures around 45°C and atmospheric pressure.
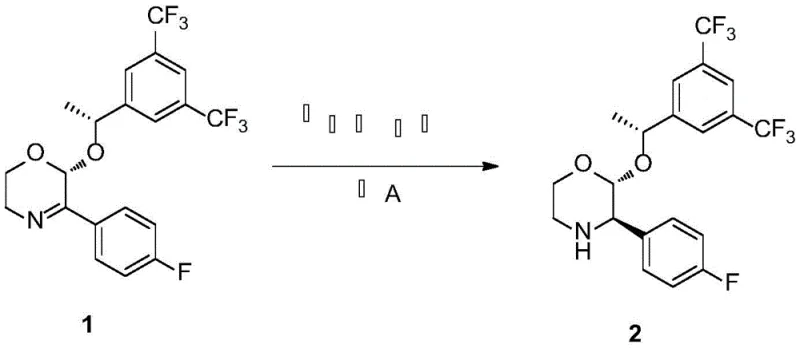
This transition eliminates the need for complex high-pressure equipment, drastically simplifying the reactor setup and enhancing operational safety. The process demonstrates exceptional efficiency, with experimental data indicating yields reaching up to 85% and enantiomeric excess (ee) values exceeding 99%. By replacing transition metals with an organocatalytic system, the method inherently reduces the environmental footprint and simplifies the purification workflow, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Biomimetic Transfer Hydrogenation
The core of this technological breakthrough lies in the design of the chiral catalyst, designated as Formula (I) in the patent documentation. This catalyst is a 3,5-amide-1,4-dihydropyridine derivative functionalized with chiral alpha-hydroxyphenylacetamide side chains. The mechanism operates through a concerted hydride transfer, where the dihydropyridine ring donates a hydride ion to the electrophilic carbon of the imine substrate, while a proton is simultaneously transferred from the acidic medium or the catalyst itself.
The stereochemical outcome is rigorously controlled by the chiral environment created by the alpha-hydroxyphenylacetamide moieties attached to the pyridine scaffold. These bulky chiral groups direct the approach of the substrate, ensuring high facial selectivity during the reduction step. This precise spatial arrangement is critical for achieving the observed >99% ee, as it effectively discriminates between the pro-chiral faces of the imine. The inclusion of a protic acid, such as hydrochloric acid or trifluoroacetic acid, further activates the imine substrate, facilitating the nucleophilic attack by the hydride source.
From an impurity control perspective, the absence of transition metals eliminates a major class of potential contaminants, specifically heavy metal residues that are notoriously difficult to purge. The reaction byproducts are primarily the oxidized pyridine forms, which are organic and generally easier to separate from the product via standard extraction and crystallization techniques. This mechanistic elegance translates directly into a cleaner crude profile, reducing the burden on downstream purification units and ensuring consistent batch-to-batch quality essential for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Aprepitant Intermediate Efficiently
The synthesis protocol outlined in the patent provides a clear, reproducible roadmap for producing the target intermediate with high fidelity. The process begins with the in-house preparation of the chiral catalyst, followed by the key reduction step. The operational simplicity of this route makes it highly attractive for technology transfer, as it relies on standard unit operations familiar to most fine chemical manufacturing sites. Detailed standardized synthetic steps see the guide below.
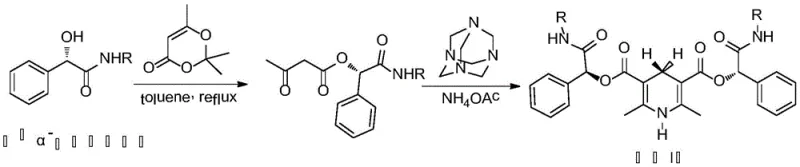
- Prepare the chiral catalyst by reacting chiral alpha-hydroxyphenylacetamide with 2,2,6-trimethyl-1,3-dioxin-4-one in toluene under reflux.
- Condense the resulting derivative with hexamethylenetetramine and ammonium acetate to form the 3,5-amide-1,4-dihydropyridine catalyst.
- Mix the imine substrate (Formula 1) with the catalyst and acid in THF at 45°C for 8 hours, followed by extraction and crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel catalytic system offers tangible strategic benefits beyond mere technical feasibility. The shift from high-pressure hydrogenation to atmospheric transfer hydrogenation fundamentally alters the cost structure and risk profile of the manufacturing process. By removing the dependency on specialized high-pressure vessels, manufacturers can utilize existing standard glass-lined or stainless steel reactors, thereby maximizing asset utilization and minimizing capital lock-up.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes the significant cost associated with purchasing precious metals and, more importantly, the expensive downstream processes required to scavenge and verify their removal. Additionally, the mild reaction temperature of 45°C significantly lowers energy consumption compared to high-temperature or high-pressure alternatives. The high yield of 85% reported in the examples ensures better material throughput, reducing the cost per kilogram of the active intermediate and optimizing raw material spend.
- Enhanced Supply Chain Reliability: Operating at atmospheric pressure inherently reduces safety risks, leading to fewer unplanned shutdowns due to safety interlocks or maintenance issues associated with high-pressure systems. The robustness of the catalyst system, which tolerates standard solvents like tetrahydrofuran and dichloromethane, ensures consistent production cycles. This reliability is crucial for maintaining continuous supply lines to downstream API manufacturers, preventing stockouts that could disrupt the availability of life-saving antiemetic medications.
- Scalability and Environmental Compliance: The process generates less hazardous waste, as it avoids heavy metal sludge and high-energy inputs. This aligns with increasingly stringent environmental regulations globally, reducing the compliance burden and waste disposal costs. The simplicity of the workup procedure—quenching with water, extracting with methyl isobutyl ketone, and freeze-crystallizing—is easily scalable from pilot plant to multi-ton production, ensuring that reducing lead time for high-purity pharmaceutical intermediates is achievable without compromising quality.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this specific preparation method. These insights are derived directly from the experimental data and claims presented in patent CN112174909A, providing clarity on the practical implementation of this technology.
Q: What are the advantages of this biomimetic catalyst over traditional hydrogenation?
A: Unlike traditional methods requiring high-pressure hydrogen and transition metals, this biomimetic system operates at atmospheric pressure and 45°C, eliminating safety risks and heavy metal contamination.
Q: What is the optical purity achieved with this method?
A: The process consistently achieves high enantiomeric excess (ee) values, with Example 1 reporting 99.2% ee and yields up to 85%.
Q: Is the catalyst reusable or easy to remove?
A: The organic nature of the catalyst avoids toxic transition metal residues, simplifying purification and reducing the need for expensive metal scavenging steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aprepitant Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the pharmaceutical value chain. Our team of expert process chemists has thoroughly analyzed the biomimetic catalytic technology described in CN112174909A and is fully prepared to implement this advanced methodology. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee >99% ee.
We invite you to collaborate with us to leverage this cost-effective and environmentally friendly synthesis route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can optimize your supply chain for this vital oncology supportive care intermediate.
