Advanced Manufacturing of Montelukast Sodium Intermediates: A Technical Breakdown for Global Supply Chains
Advanced Manufacturing of Montelukast Sodium Intermediates: A Technical Breakdown for Global Supply Chains
The pharmaceutical landscape for asthma and allergy treatments continues to evolve, driven by the demand for more efficient manufacturing of key active ingredients like Montelukast Sodium. A pivotal development in this sector is detailed in patent CN102442948B, which outlines a novel method for preparing the critical halogenated intermediate required for Montelukast synthesis. This technology represents a significant departure from legacy synthetic routes, offering a streamlined pathway that addresses long-standing challenges in yield optimization and impurity control. For R&D directors and procurement strategists, understanding this shift is essential for securing a competitive edge in the global pharmaceutical intermediates market. The patent describes a direct conversion strategy that bypasses cumbersome protection-deprotection sequences, thereby enhancing the overall economic viability of the supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Montelukast precursors has been plagued by excessive step counts and the reliance on sensitive protecting groups. Traditional routes often necessitate the installation and subsequent removal of bulky silyl ethers (such as TBS) or tetrahydropyranyl (THP) groups to mask reactive hydroxyl functionalities during carbon-sulfur bond formation. As illustrated in the complex reaction schemes of prior art, these multi-step sequences not only inflate material costs but also introduce significant opportunities for yield loss at each stage. Furthermore, the use of heavy metal catalysts or harsh conditions in older methodologies can lead to difficult-to-remove impurities, complicating the purification process and jeopardizing the strict quality standards required for high-purity API intermediates. The cumulative effect is a supply chain that is fragile, expensive, and environmentally burdensome.
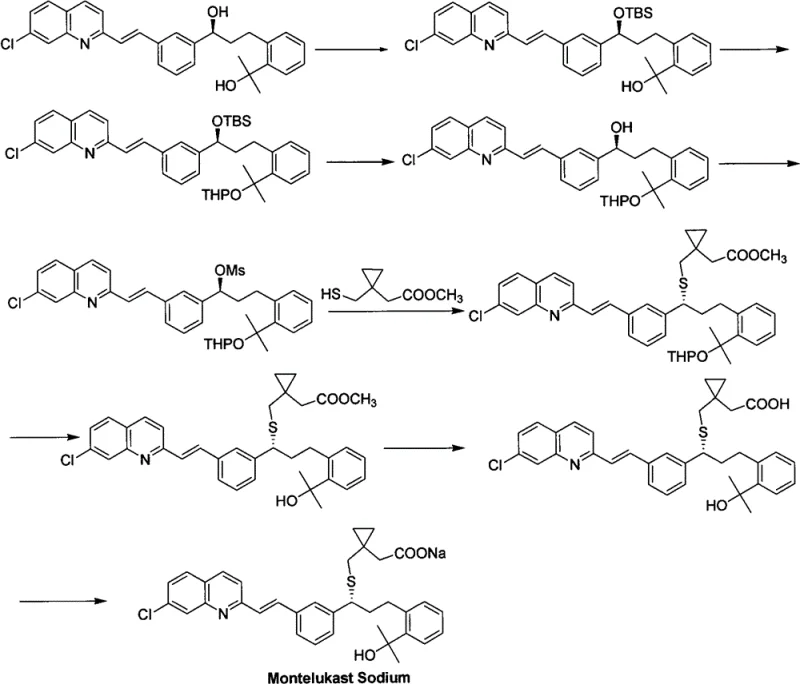
The Novel Approach
In stark contrast, the methodology disclosed in CN102442948B introduces a remarkably concise strategy centered on the direct activation of the secondary alcohol. Instead of navigating a labyrinth of protection steps, the new process employs a tandem sulfonylation-halogenation sequence. This approach converts the hydroxyl group of the precursor (Compound II) directly into a leaving group (sulfonate) and subsequently displaces it with a halide ion in a single operational pot. This innovation drastically reduces the number of unit operations, minimizes solvent consumption, and eliminates the waste associated with protecting group chemistry. By simplifying the molecular architecture early in the synthesis, manufacturers can achieve a more robust and predictable production profile, which is critical for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into One-Pot Sulfonylation-Halogenation
The core of this technological advancement lies in the precise control of reaction kinetics and thermodynamics during the activation phase. The process begins with the treatment of the chiral secondary alcohol with a sulfonylating agent, such as methanesulfonyl chloride (MsCl), in the presence of a non-nucleophilic base like diisopropylethylamine (DIPEA). This step generates a highly reactive mesylate intermediate in situ. Crucially, the reaction is conducted at low temperatures, typically ranging from -35°C to 0°C, to suppress potential side reactions and maintain the integrity of the sensitive olefinic and quinoline moieties present in the molecule. The choice of base and the stoichiometric ratio are optimized to ensure complete conversion without inducing elimination pathways that could degrade the product quality.
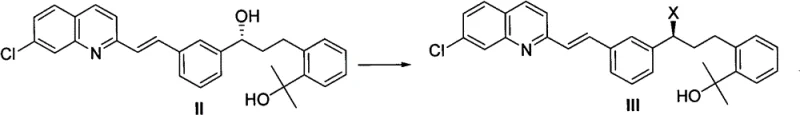
Following the formation of the sulfonate ester, the reaction mixture is treated directly with a halogenating source, such as lithium chloride or tetrabutylammonium chloride. This nucleophilic substitution proceeds smoothly, often requiring a gradual warming to room temperature to drive the reaction to completion. The mechanism likely follows an SN2 pathway, ensuring high stereochemical fidelity, which is paramount for the biological activity of the final drug substance. The patent highlights that this direct displacement avoids the isolation of unstable intermediates, thereby reducing exposure to moisture and air which could otherwise lead to hydrolysis or oxidation. This mechanistic elegance translates directly into operational simplicity, allowing for tighter control over the impurity profile and ensuring the resulting intermediate possesses the high optical purity demanded by regulatory bodies.
How to Synthesize Montelukast Sodium Intermediate Efficiently
Implementing this synthesis route requires careful attention to solvent selection and temperature management to maximize efficiency. The patent specifies the use of polar aprotic solvents like DMF or toluene, which facilitate the dissolution of both the organic substrate and the inorganic halide salts. Operators must maintain an inert atmosphere, typically using nitrogen or argon, to prevent oxidative degradation of the sensitive vinyl and thioether linkages. The procedure involves a sequential addition of reagents where the sulfonylation is allowed to reach completion before the introduction of the halide source, ensuring a clean transformation. For a detailed breakdown of the specific molar ratios, addition rates, and workup procedures, please refer to the standardized guide below.
- Dissolve the secondary alcohol precursor (Compound II) in a polar aprotic solvent like DMF or Toluene under inert gas protection.
- Add a base such as DIPEA and a sulfonylating agent (e.g., MsCl) at low temperatures (-35°C to 0°C) to activate the hydroxyl group.
- Directly introduce a halogenating source like Lithium Chloride to the reaction mixture, allowing the temperature to rise for substitution to occur.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this streamlined synthesis offers tangible benefits that extend beyond mere technical novelty. By collapsing multiple synthetic steps into a single pot operation, the process inherently reduces the consumption of raw materials, solvents, and energy. This reduction in process intensity directly correlates to a lower cost of goods sold (COGS), providing a buffer against volatile raw material pricing. Moreover, the simplified workflow shortens the overall manufacturing cycle time, enabling faster turnaround for batch production and improved responsiveness to market demand fluctuations. These factors collectively enhance the resilience of the supply chain, making it less susceptible to disruptions caused by complex logistics or equipment bottlenecks.
- Cost Reduction in Manufacturing: The elimination of protecting group reagents and the associated deprotection steps removes a significant cost center from the production budget. Traditional routes require expensive silyl chlorides and acidic or fluoride conditions for removal, along with the disposal of the resulting waste streams. By bypassing these steps, the new method achieves substantial cost savings through reduced reagent procurement and lower waste treatment expenses. Additionally, the higher yield and purity reduce the need for extensive chromatographic purification, further driving down operational expenditures.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as lithium chloride and methanesulfonyl chloride ensures a stable supply of inputs, unlike specialized catalysts that may face sourcing constraints. The robustness of the reaction conditions means that production is less likely to be halted by minor deviations in temperature or pressure, leading to more consistent batch success rates. This reliability is crucial for maintaining continuous supply to downstream API manufacturers, reducing the risk of stockouts and ensuring timely delivery of high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing solvents and conditions that are easily managed in large-scale reactors. The reduction in step count inherently lowers the environmental footprint by decreasing the total volume of solvent waste and hazardous byproducts generated per kilogram of product. This aligns with increasingly stringent global environmental regulations, facilitating smoother regulatory approvals and reducing the liability associated with hazardous waste disposal. The method supports the commercial scale-up required to meet the growing global demand for Montelukast formulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear picture of the technology's capabilities. Understanding these details is vital for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers reflect the balance between chemical efficiency and practical operability in a commercial setting.
Q: What are the primary advantages of this new halogenation method over traditional routes?
A: The patented method eliminates the need for complex protecting group strategies (like TBS or THP ethers) used in conventional syntheses. By converting the hydroxyl group directly to a halide via a sulfonate intermediate in a one-pot sequence, the process significantly reduces step count, minimizes waste generation, and simplifies purification, leading to higher overall throughput.
Q: How does this process ensure high optical purity for the final API?
A: The reaction conditions are meticulously controlled, specifically maintaining low temperatures during the activation phase to prevent racemization. The use of mild bases and specific halogenating agents ensures that the stereocenter is preserved or inverted predictably without degradation, resulting in intermediates with high Ee values suitable for stringent pharmaceutical standards.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the protocol utilizes commercially available reagents like methylsulfonyl chloride and lithium chloride in common solvents such as DMF or Toluene. The absence of sensitive catalysts and the robustness of the reaction conditions make it highly amenable to scale-up from pilot batches to multi-ton commercial manufacturing without significant engineering hurdles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Montelukast Sodium Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and reliable synthesis routes in the modern pharmaceutical supply chain. Our team of expert chemists has extensively evaluated the methodology described in CN102442948B and possesses the technical capability to implement this advanced process at scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Montelukast intermediate meets the highest international standards for safety and efficacy.
We invite you to collaborate with us to leverage these process improvements for your specific production requirements. Our technical sales team is prepared to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating exactly how this optimized route can impact your bottom line. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Let us partner with you to secure a sustainable and cost-effective supply of this vital pharmaceutical building block.
