Advanced Ru-Catalyzed Synthesis of Chiral Montelukast Intermediates for Commercial Scale-Up
Advanced Ru-Catalyzed Synthesis of Chiral Montelukast Intermediates for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for high-value active pharmaceutical ingredients (APIs) and their precursors. A pivotal development in this domain is documented in patent CN103073492A, which discloses an innovative synthesis method for 2-[3-(S)-[3-[2-(7-chloro-2-quinolyl)vinyl]phenyl]-3-hydroxypropyl]benzoate. This compound serves as a critical chiral intermediate in the manufacturing of Montelukast Sodium, a widely prescribed leukotriene receptor antagonist used for the maintenance treatment of asthma and the relief of symptoms of seasonal allergic rhinitis. The technical breakthrough presented in this patent addresses long-standing challenges regarding cost efficiency and environmental safety in the production of this key pharmaceutical building block. By leveraging asymmetric transfer hydrogenation technology, the disclosed method offers a pathway that significantly mitigates the reliance on hazardous solvents and expensive stoichiometric reagents, thereby aligning with modern green chemistry principles while ensuring high purity standards required for global regulatory compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this specific chiral alcohol intermediate has relied heavily on methods that present substantial economic and environmental drawbacks for large-scale manufacturers. Prior art, such as that described in WO2008032099A2 and EP20081988079A1, predominantly utilized (-)-DIP-Chloride as a stoichiometric reducing agent to achieve the necessary stereoselectivity. The fundamental flaw in this approach is the requirement for equivalent amounts of the reducing agent, which is not only prohibitively expensive but also generates significant quantities of boron-containing waste that requires complex disposal protocols. Furthermore, alternative asymmetric hydrogenation routes often necessitated the use of chlorobenzene as a primary solvent. Chlorobenzene is classified as a toxic solvent with severe implications for operator health and environmental safety, creating significant regulatory hurdles and increasing the cost of waste treatment and emission control in a commercial manufacturing setting. These factors collectively render conventional methods suboptimal for cost-sensitive and sustainability-focused supply chains.
The Novel Approach
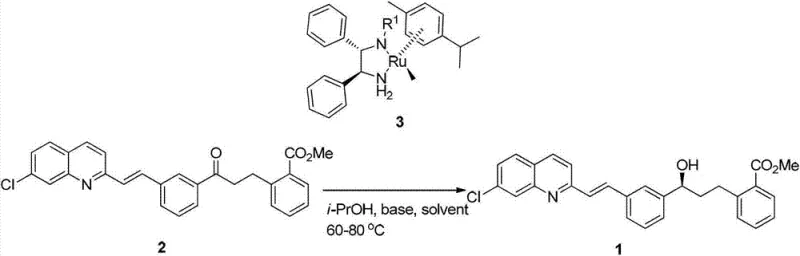
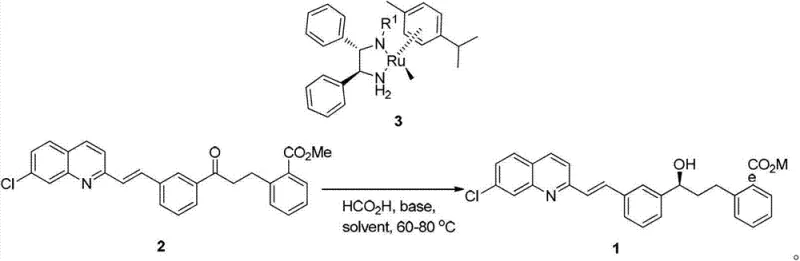
In stark contrast to these legacy techniques, the novel approach detailed in the patent utilizes a highly efficient asymmetric transfer hydrogenation strategy driven by a chiral Ruthenium catalyst. This method fundamentally shifts the paradigm from stoichiometric consumption to catalytic turnover, drastically reducing the material cost per kilogram of the final product. The process employs either isopropanol or formic acid as the hydrogen source, both of which are commodity chemicals that are inexpensive, readily available, and far less toxic than chlorobenzene. Specifically, the use of isopropanol allows it to function dually as both the reaction solvent and the hydrogen donor, simplifying the reaction matrix and downstream processing. Alternatively, the formic acid system offers a potent hydrogen source that operates effectively in polar aprotic solvents like DMF. This flexibility allows process chemists to optimize conditions based on solubility and throughput requirements without compromising the stereochemical integrity of the product.
Mechanistic Insights into Ru-Catalyzed Asymmetric Transfer Hydrogenation
The core of this technological advancement lies in the sophisticated mechanism of the Noyori-type Ruthenium catalyst, specifically the (S,S)-N-(R)-1,2-diphenylethylenediamine (p-cymene)-ruthenium(II) chloride complex. This catalyst operates through a metal-ligand bifunctional mechanism, where both the metal center and the ligand participate actively in the bond-breaking and bond-forming events. During the catalytic cycle, the ruthenium hydride species transfers a hydride ion to the carbonyl carbon of the ketone substrate, while simultaneously, the amine proton on the ligand is transferred to the carbonyl oxygen. This concerted six-membered transition state ensures a highly organized spatial arrangement that dictates the stereochemical outcome of the reduction. The chiral environment provided by the diphenylethylenediamine ligand effectively shields one face of the prochiral ketone, forcing the hydride attack to occur from the preferred trajectory, thus yielding the desired (S)-enantiomer with high optical purity. This mechanistic elegance allows the reaction to proceed with high turnover numbers, meaning a very small amount of the precious metal catalyst can convert a large quantity of substrate.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or harsh chemical reductions. Because the reaction proceeds under mild thermal conditions (60-80°C) and neutral to slightly basic pH, there is minimal risk of side reactions such as double bond isomerization or ester hydrolysis, which are common pitfalls in the synthesis of complex molecules containing sensitive functional groups like the vinyl quinoline moiety. The selectivity of the catalyst ensures that the reduction is chemoselective for the ketone carbonyl over other potential reducible groups, thereby simplifying the impurity profile of the crude reaction mixture. This high level of chemoselectivity reduces the burden on downstream purification steps, such as column chromatography or crystallization, leading to higher overall yields and reduced solvent consumption during the isolation phase. Consequently, the final product exhibits a cleaner impurity spectrum, which is a critical quality attribute for pharmaceutical intermediates destined for GMP production.
How to Synthesize 2-[3-(S)-[3-[2-(7-chloro-2-quinolyl)vinyl]phenyl]-3-hydroxypropyl]benzoate Efficiently
The execution of this synthesis requires precise control over reaction parameters to maximize both yield and enantiomeric excess. The process begins with the preparation of the reaction vessel under an inert atmosphere, typically nitrogen or argon, to prevent oxidation of the sensitive catalyst species. The substrate, 2-[3-[3-[2-(7-chloro-2-quinolyl)-vinyl]phenyl]-(3-carbonyl)propyl]benzoate, is combined with the chiral Ruthenium catalyst and an inorganic base such as potassium carbonate. The choice of solvent system—either isopropanol for the transfer hydrogenation route or DMF with formic acid—dictates the specific thermal profile, though both generally operate effectively within the 60-80°C range. Reaction monitoring via Thin Layer Chromatography (TLC) is essential to determine the endpoint, which typically occurs within 0.2 to 5 hours depending on the specific catalyst loading and temperature. Upon completion, the workup involves a straightforward aqueous quench followed by organic extraction, drying, and concentration. For a comprehensive breakdown of the specific operational parameters and safety precautions, please refer to the standardized procedure outlined below.
- Prepare the reaction mixture by combining the ketone substrate, the chiral Ruthenium catalyst complex, and an inorganic base such as potassium carbonate in a suitable solvent like isopropanol or DMF under an inert atmosphere.
- Heat the reaction mixture to a temperature range of 60-80°C and maintain stirring for 0.2 to 5 hours until TLC analysis confirms complete conversion of the starting material.
- Quench the reaction with saturated ammonium chloride solution, extract the product using ethyl acetate, wash with brine, dry over anhydrous sodium sulfate, and purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route translates directly into tangible strategic benefits that enhance the resilience and profitability of the supply chain. The most immediate impact is observed in the raw material cost structure, where the elimination of expensive stoichiometric reducing agents like (-)-DIP-Chloride results in a drastic reduction in the Bill of Materials (BOM) cost. Furthermore, the substitution of toxic chlorobenzene with commodity solvents like isopropanol removes the need for specialized handling equipment and expensive hazardous waste disposal contracts, thereby lowering the overhead costs associated with Environmental, Health, and Safety (EHS) compliance. This shift not only reduces direct operational expenditures but also mitigates the risk of supply disruptions caused by regulatory crackdowns on hazardous chemical usage. The simplicity of the workup procedure, which avoids complex distillation or extensive purification steps, further contributes to reduced manufacturing cycle times and increased throughput capacity at existing facilities.
- Cost Reduction in Manufacturing: The transition from stoichiometric reagents to a catalytic system fundamentally alters the cost dynamics of the production process. By utilizing a Ruthenium catalyst that operates at low loading levels, the consumption of precious metals is minimized while maintaining high conversion rates. Additionally, the use of isopropanol or formic acid as hydrogen sources leverages the economies of scale associated with bulk commodity chemicals, which are significantly cheaper than specialized chiral reducing agents. This structural change in the reagent profile ensures that the cost of goods sold (COGS) remains stable even during periods of volatility in the specialty chemical market. The reduction in waste generation also lowers the hidden costs associated with waste treatment and disposal, contributing to a leaner and more cost-effective manufacturing operation that enhances overall margin potential.
- Enhanced Supply Chain Reliability: Reliance on exotic or highly regulated solvents like chlorobenzene introduces significant fragility into the supply chain, as availability can be impacted by changing environmental regulations and transportation restrictions. By switching to universally available solvents like isopropanol and DMF, the manufacturing process becomes decoupled from these supply risks, ensuring a continuous and reliable flow of materials. The robustness of the catalyst system also means that the process is less sensitive to minor variations in raw material quality, reducing the likelihood of batch failures and production delays. This stability allows for more accurate demand forecasting and inventory planning, enabling the supply chain team to maintain optimal stock levels without the need for excessive safety buffers. Ultimately, this leads to shorter lead times and improved on-time delivery performance for downstream API manufacturers.
- Scalability and Environmental Compliance: The design of this synthesis route inherently supports seamless scale-up from pilot plant to commercial production volumes. The reaction conditions are mild and do not require high-pressure equipment or cryogenic temperatures, which simplifies the engineering requirements for large-scale reactors. From an environmental perspective, the process aligns with the principles of Green Chemistry by minimizing the use of hazardous substances and reducing the generation of toxic byproducts. This alignment facilitates easier permitting and regulatory approval in jurisdictions with strict environmental standards, such as the EU and North America. The ability to demonstrate a commitment to sustainable manufacturing practices also enhances the corporate reputation of the supplier, making them a preferred partner for multinational pharmaceutical companies that prioritize ESG (Environmental, Social, and Governance) criteria in their vendor selection processes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for process development teams. Understanding these details is crucial for evaluating the feasibility of integrating this route into existing manufacturing workflows. We encourage technical stakeholders to review these points carefully to assess the compatibility of this method with their specific quality and throughput requirements.
Q: What are the primary advantages of this Ru-catalyzed method over traditional reduction techniques?
A: The primary advantages include the replacement of expensive stoichiometric reducing agents like (-)-DIP-Chloride with catalytic amounts of Ruthenium complexes, and the substitution of toxic chlorobenzene solvents with environmentally benign isopropanol or formic acid systems.
Q: What hydrogen sources are compatible with this asymmetric transfer hydrogenation process?
A: The patent specifies two effective hydrogen sources: isopropanol (i-PrOH) acting as both solvent and hydrogen donor, or formic acid (HCO2H) used in conjunction with a polar aprotic solvent like DMF.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method is explicitly designed for scalability, utilizing cheap and readily available raw materials, simple workup procedures involving standard extraction and drying, and avoiding hazardous reagents that complicate industrial safety compliance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Montelukast Sodium Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the successful development and commercialization of life-saving medications like Montelukast. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global pharmaceutical supply chains. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify identity, assay, and chiral purity. Our facility is equipped to handle complex catalytic reactions safely and efficiently, providing a secure and compliant source for your critical raw materials. By partnering with us, you gain access to a supply chain that is not only robust and reliable but also deeply knowledgeable about the nuances of chiral synthesis and process optimization.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific project needs. Whether you require a Customized Cost-Saving Analysis to evaluate the economic benefits of switching to this catalytic method or need specific COA data to validate our quality standards, we are ready to provide comprehensive support. We encourage you to request route feasibility assessments to explore how our manufacturing capabilities can accelerate your development timelines. Contact us today to initiate a dialogue about securing a sustainable and cost-effective supply of high-purity Montelukast intermediates for your upcoming production campaigns.
