Advanced Manufacturing Protocol for High-Purity Ceftazidime Intermediates and Impurity Control
The pharmaceutical industry continuously demands higher purity standards for critical antibiotic intermediates, particularly for third-generation cephalosporins like ceftazidime. A recent technological breakthrough detailed in patent CN109912625B introduces a refined process method specifically designed to drastically reduce the levels of Ceftazidime Impurity H, a toxic methyl ester derivative. This innovation addresses a longstanding challenge in organic synthesis where traditional solvent systems inadvertently promote the formation of hazardous byproducts. By re-engineering the solvent environment during the condensation of 7-APCA and ceftazidime active ester, manufacturers can now achieve impurity levels well below 0.1%, significantly enhancing the safety profile of the final Active Pharmaceutical Ingredient (API). For global procurement teams and R&D directors, this represents a pivotal shift towards safer, more reliable ceftazidime intermediate supplier networks that prioritize patient safety through rigorous chemical control.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of ceftazidime has relied heavily on methanol-based solvent systems during the formation of the ceftazidime tert-butyl ester. While methanol offers excellent solubility properties, its chemical reactivity poses a severe risk in this specific context. The presence of methanol facilitates a transesterification reaction where the tert-butyl group at the seven-position side chain is replaced by a methyl group, resulting in the formation of Ceftazidime Methyl Ester, known as Impurity H. This impurity is not merely a quality defect but a significant safety hazard; toxicological assessments using zebrafish embryos have demonstrated that Impurity H possesses a teratogenic effect 25 times greater and a lethal effect 8 times greater than ceftazidime itself. Conventional processes often struggle to keep this impurity below the 0.2% threshold, necessitating costly and yield-reducing purification steps post-synthesis. Furthermore, the reliance on methanol limits the ability to scale up production without compromising the stringent purity specifications required by major pharmacopoeias.
The Novel Approach
The patented methodology overcomes these intrinsic defects by fundamentally altering the reaction medium. Instead of relying on methanol as a primary or sole solvent, the new process employs a sophisticated mixed solvent system comprising main solvents such as dichloromethane, chloroform, tetrahydrofuran, or acetonitrile, combined with tert-butyl alcohol as a cosolvent. This strategic substitution effectively suppresses the transesterification pathway, thereby preventing the generation of the toxic methyl ester at the source. The process incorporates a critical water treatment step post-reaction, which facilitates the separation of impurities and inorganic salts before the final crystallization. By optimizing the volume ratio of the main solvent to the cosolvent between 10:1 and 25:1, the reaction maintains high efficiency while ensuring that the final ceftazidime tert-butyl ester contains less than 0.1% of Impurity H. This approach not only simplifies the downstream purification workflow but also ensures a consistent, high-quality output suitable for direct conversion into the final antibiotic product.
Mechanistic Insights into Solvent-Mediated Impurity Suppression
The core of this technological advancement lies in the precise control of nucleophilic substitution reactions during the acylation of the 7-APCA nucleus. In traditional methanol-rich environments, the methoxide ion or methanol molecules act as nucleophiles that attack the ester linkage, displacing the tert-butyl group. The novel process mitigates this by utilizing tert-butyl alcohol, which shares the same steric bulk as the protecting group, thus thermodynamically disfavoring any exchange reaction. The reaction is conducted under alkaline conditions, with the pH carefully maintained between 7 and 9 using agents like triethylamine or diethylamine. This pH window is critical; it is sufficiently basic to activate the amine group of 7-APCA for nucleophilic attack on the active ester, yet mild enough to prevent the hydrolysis of the beta-lactam ring or the decomposition of the sensitive thiazole side chain. The use of low temperatures, specifically between 0°C and 10°C, further kinetically controls the reaction, minimizing side reactions and ensuring the stereochemical integrity of the (6R,7R) configuration is preserved throughout the synthesis.
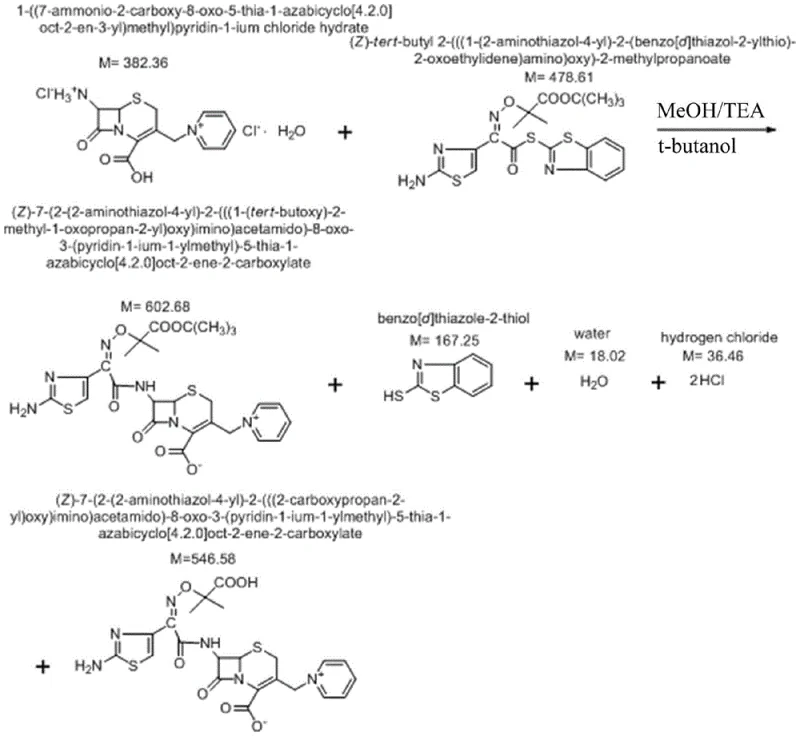
Following the condensation, the process introduces a unique water treatment phase. Adding pure water to the reaction mixture induces a phase separation or precipitation of specific impurities, allowing for their removal prior to crystallization. This step is instrumental in controlling unknown impurities to below 0.05%. The subsequent deprotection of the tert-butyl ester to form the hydrochloride salt is achieved using a mixture of formic acid and hydrochloric acid at mild temperatures of 15-20°C. This gentle deprotection avoids the harsh conditions that could otherwise degrade the molecule or regenerate impurities. Finally, the conversion to the pentahydrate form involves precise pH adjustment to 6.0-6.5 and controlled crystallization, ensuring the physical stability and bioavailability of the final drug substance. This comprehensive mechanistic control from intermediate to final API demonstrates a robust understanding of cephalosporin chemistry.
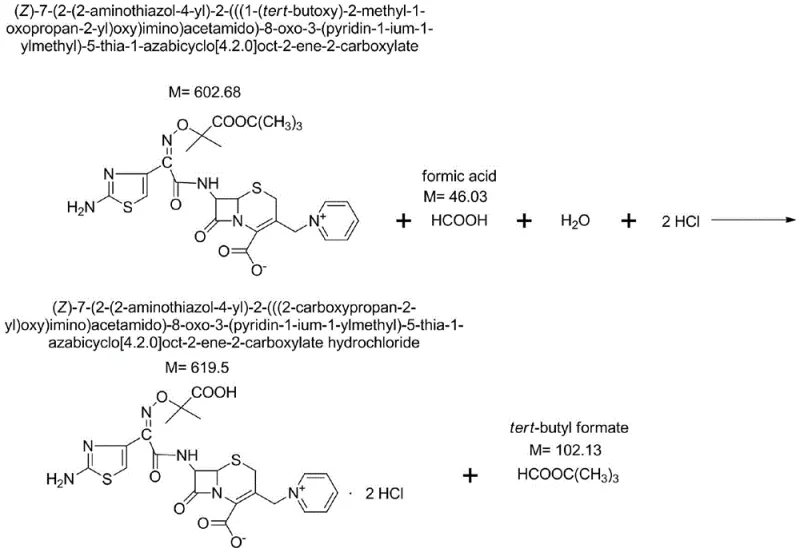
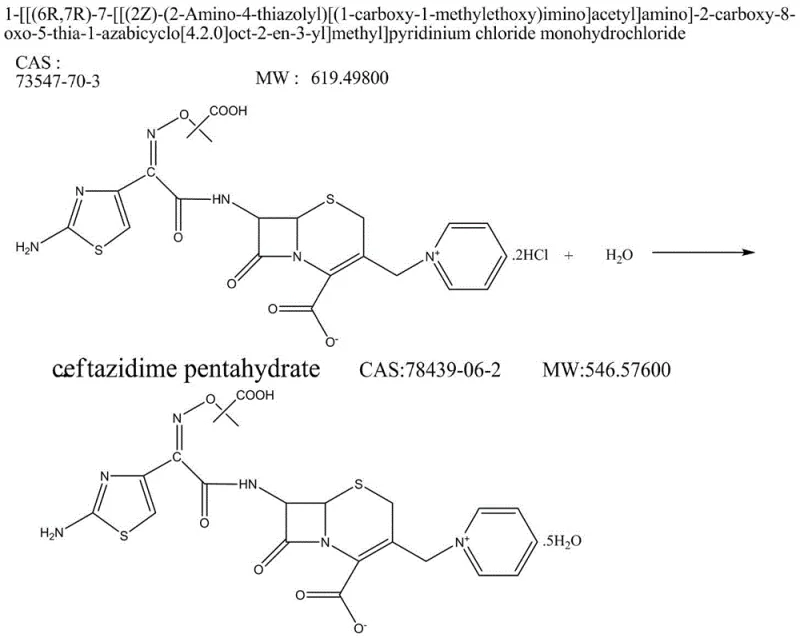
How to Synthesize Ceftazidime Tert-Butyl Ester Efficiently
Implementing this advanced synthesis route requires strict adherence to solvent ratios and temperature profiles to maximize yield and purity. The process begins with the preparation of the solvent matrix, followed by the controlled addition of reactants under inert atmosphere conditions to prevent oxidation. The key to success lies in the post-reaction workup, where the water treatment duration of 1 to 2 hours allows for complete equilibration and impurity partitioning. For detailed operational parameters, including specific stirring rates and filtration techniques, please refer to the standardized protocol below.
- Prepare a mixed solvent system comprising a main solvent (such as dichloromethane or tetrahydrofuran) and a cosolvent of tert-butyl alcohol, maintaining a volume ratio between 10: 1 and 25:1.
- Cool the solvent mixture to 0-10°C, add 7-APCA and ceftazidime active ester, and adjust pH to 7-9 using an alkaline agent like triethylamine.
- React for 20-24 hours, followed by water treatment for 1-2 hours to separate impurities, and finally perform solvent crystallization to isolate the pure tert-butyl ester.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this impurity-controlled process translates into tangible strategic benefits beyond mere regulatory compliance. The elimination of methanol-driven side reactions means that the manufacturing process is far more predictable and robust, reducing the risk of batch failures due to out-of-specification impurity profiles. This reliability is crucial for maintaining continuous supply lines for essential antibiotics. Furthermore, the simplified purification train—relying on crystallization and washing rather than complex chromatographic separations—drastically reduces the consumption of expensive stationary phases and solvents. This efficiency gain directly impacts the cost of goods sold (COGS), allowing for more competitive pricing in the global market without sacrificing quality. The use of common, industrially available solvents like dichloromethane and tert-butyl alcohol also ensures that raw material sourcing remains stable and unaffected by niche supply shocks.
- Cost Reduction in Manufacturing: The process eliminates the need for extensive reprocessing or recycling of batches contaminated with high levels of Impurity H. By preventing the formation of the toxic methyl ester at the source, manufacturers save significant resources on waste disposal and additional purification steps. The high yield of the tert-butyl ester intermediate, often exceeding theoretical expectations due to reduced side reactions, further enhances the overall economic viability of the production line. Additionally, the mild reaction conditions reduce energy consumption associated with heating or cryogenic cooling, contributing to a leaner manufacturing footprint.
- Enhanced Supply Chain Reliability: The robustness of the solvent system ensures consistent batch-to-batch quality, which is vital for long-term supply contracts with major pharmaceutical companies. The ability to consistently deliver ceftazidime intermediates with Impurity H levels below 0.1% builds trust and reduces the administrative burden of quality investigations. Moreover, the scalability of the process from laboratory to multi-ton production is proven, meaning that supply can be ramped up quickly to meet surges in demand without the need for re-validating complex new equipment or protocols.
- Scalability and Environmental Compliance: The reduction in hazardous waste generation is a significant advantage. By avoiding the formation of toxic byproducts, the environmental load of the manufacturing process is lightened, facilitating easier compliance with increasingly strict environmental regulations. The water treatment step allows for the recovery and reuse of organic solvents, aligning with green chemistry principles. This sustainability angle is increasingly important for multinational corporations aiming to reduce their carbon footprint and meet ESG (Environmental, Social, and Governance) goals while sourcing critical raw materials.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this low-impurity synthesis method. These insights are derived directly from the patent data and practical application scenarios, providing clarity for technical decision-makers evaluating this technology for their supply chains.
Q: Why is Impurity H (Ceftazidime Methyl Ester) critical to control in API manufacturing?
A: Impurity H exhibits significantly higher toxicity than the parent compound, with zebrafish embryotoxicity studies indicating it is 25 times more teratogenic and 8 times more lethal. Strict control below 0.1% is essential for patient safety and regulatory compliance.
Q: How does the novel solvent system prevent the formation of methyl ester impurities?
A: By replacing methanol with a mixed system of dichloromethane (or similar) and tert-butyl alcohol, the process eliminates the transesterification reaction pathway that typically converts the tert-butyl group into the toxic methyl ester during synthesis.
Q: What are the scalability advantages of this water-treatment crystallization method?
A: The method utilizes mild reaction temperatures (0-10°C) and common industrial solvents. The water treatment step effectively removes inorganic salts and polar byproducts without requiring complex chromatography, making it highly suitable for multi-ton commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ceftazidime Intermediate Supplier
At NINGBO INNO PHARMCHEM, we understand that the purity of intermediates like ceftazidime tert-butyl ester is the foundation of a safe and effective final drug product. Our technical team has extensively analyzed the implications of patent CN109912625B and integrated similar impurity-control strategies into our own manufacturing platforms. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to industrial reality is seamless. Our facilities are equipped with rigorous QC labs capable of detecting trace impurities at the ppm level, guaranteeing that every batch meets stringent purity specifications required by global pharmacopoeias.
We invite procurement leaders and R&D directors to collaborate with us to leverage these advanced synthesis capabilities for your specific project needs. By partnering with us, you gain access to a Customized Cost-Saving Analysis that evaluates how switching to this optimized solvent system can impact your overall budget and timeline. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and samples that demonstrate our commitment to delivering high-purity ceftazidime intermediates with superior impurity profiles.
