Scalable Manufacturing of Pyridine Quinazoline via Advanced Convergent Synthesis Routes
Scalable Manufacturing of Pyridine Quinazoline via Advanced Convergent Synthesis Routes
The global demand for high-efficiency, low-toxicity insecticides has driven intense research into novel quinazoline derivatives, specifically pyridine quinazoline (C19H15F7N4O2), which exhibits exceptional efficacy against stinkbugs, aphids, and whiteflies. A pivotal advancement in this domain is documented in patent CN111704604B, which discloses a robust preparation method that fundamentally shifts the production paradigm from inefficient linear sequences to a streamlined convergent synthesis. This technological breakthrough addresses critical bottlenecks in agrochemical manufacturing by optimizing atom economy and reducing the reliance on prohibitively expensive starting materials. By leveraging methyl acetyl (2-methyl-4-perfluoropropane phenyl) carbamate as a key building block, the process ensures a more stable supply chain and enhanced economic viability for large-scale production. The following analysis details the mechanistic superiority and commercial implications of this innovative route.
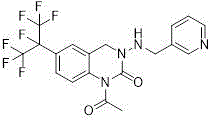
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of pyridine quinazoline has been plagued by complex, multi-step linear routes that impose severe economic and operational burdens on manufacturers. For instance, European patent EP1097932 outlines a cumbersome seven-step reaction sequence originating from 2-nitro-5-heptafluoroisopropylbenzaldehyde, a starting material notorious for its synthetic difficulty and high cost. Furthermore, this legacy method necessitates the use of N,N-carbonyldiimidazole (CDI), an expensive coupling reagent that significantly inflates the bill of materials without adding proportional value to the final active ingredient. Similarly, other reported methods, such as those in WO2005123695, rely on linear pathways starting from 2-bromoheptafluoropropane, where the overall production cost is inextricably linked to the low utilization rate of this fluorinated precursor. These conventional approaches not only suffer from low overall yields due to the accumulation of losses at each step but also generate substantial waste streams, complicating environmental compliance and increasing disposal costs for production facilities.
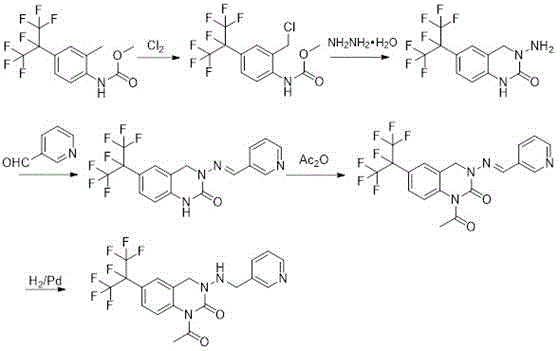
The Novel Approach
In stark contrast to these inefficient legacy processes, the methodology disclosed in CN111704604B introduces a highly efficient convergent synthesis strategy that dramatically simplifies the manufacturing landscape. This novel approach strategically decouples the synthesis into two parallel streams that converge at the cyclization stage, effectively reducing the longest linear sequence from five or seven steps down to just four critical operations. By selecting methyl acetyl (2-methyl-4-perfluoropropane phenyl) carbamate and 3-pyridine formaldehyde as the primary feedstocks, the process bypasses the need for difficult-to-source nitro-aldehydes and costly coupling agents. The convergence point utilizes a newly synthesized intermediate, 3-(hydrazinomethyl)pyridine, which is prepared in situ with high efficiency. This structural reorganization of the synthetic pathway not only enhances the overall throughput but also improves the utilization rate of the valuable perfluoropropane moiety, ensuring that raw material costs are minimized while maintaining rigorous quality standards suitable for agricultural applications.
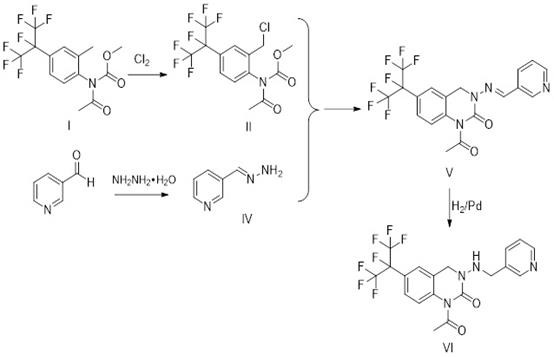
Mechanistic Insights into Convergent Cyclization and Hydrogenation
The core of this technological advancement lies in the precise control of reaction mechanisms across the four distinct stages, beginning with a free radical chlorination initiated by azobisisobutyronitrile (AIBN). In this initial phase, chlorine gas is introduced to methyl acetyl (2-methyl-4-perfluoropropane phenyl) carbamate at a controlled temperature range of 55-60°C, facilitating the selective substitution of the benzylic hydrogen to form the chloromethyl intermediate. This step is critical as it activates the aromatic ring for subsequent nucleophilic attack, and the use of chlorobenzene as a solvent ensures optimal solubility and heat transfer during the exothermic chlorination process. Following this, the parallel stream involves the N-alkylation of 3-pyridine formaldehyde with hydrazine hydrate in toluene at mild temperatures (30-35°C), generating the reactive hydrazine derivative required for ring closure. The convergence occurs when these two intermediates meet in a methanol medium with sodium carbonate acting as an acid scavenger, promoting the intramolecular cyclization that forms the dihydroquinazolinone core structure with remarkable selectivity and minimal byproduct formation.
The final transformation involves a catalytic hydrogenation step that is meticulously engineered to ensure both safety and high conversion rates. The cyclic imine intermediate is subjected to hydrogen pressure of 2 kg/cm² in the presence of 5% palladium on carbon (Pd/C) catalyst within a dichloroethane solvent system at 40-45°C. This reduction step is pivotal for saturating the double bond and establishing the final pharmacophore of the pyridine quinazoline molecule. The inclusion of concentrated sulfuric acid and potassium iodide (KI) in specific molar ratios acts as a promoter system, enhancing the activity of the palladium catalyst and preventing potential poisoning by sulfur or nitrogen impurities. Post-reaction, the crude product undergoes a rigorous recrystallization process using toluene, which effectively removes trace organic impurities and residual catalysts, resulting in a final product purity of 97.5-98.5%. This level of purity is essential for meeting the stringent regulatory requirements for agrochemical active ingredients, ensuring consistent field performance and minimizing phytotoxicity risks.
How to Synthesize Pyridine Quinazoline Efficiently
The synthesis of pyridine quinazoline via this patented convergent route represents a significant leap forward in process chemistry, offering a standardized protocol that balances reaction kinetics with operational safety. The procedure begins with the activation of the phenyl carbamate precursor through radical chlorination, followed by the independent preparation of the hydrazine linker, before merging these streams for cyclization and final reduction. This modular approach allows for better inventory management and quality control at each stage, as intermediates can be verified before proceeding to the next step. For R&D teams looking to implement this technology, the detailed reaction conditions regarding temperature gradients, molar ratios, and solvent volumes provided in the patent serve as a robust foundation for process optimization. The standardized synthesis steps outlined below provide a clear roadmap for replicating the high yields and purity profiles demonstrated in the experimental examples.
- Perform benzyl free radical chlorination on methyl acetyl (2-methyl-4-perfluoropropane phenyl) carbamate using chlorine and AIBN initiator.
- Synthesize 3-(hydrazinomethyl)pyridine via N-alkylation of 3-pyridine formaldehyde with hydrazine hydrate.
- Execute cyclization reaction between the chloromethyl intermediate and hydrazine derivative using sodium carbonate in methanol.
- Conduct catalytic hydrogenation using Pd/C under controlled pressure to obtain the final pyridine quinazoline product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this convergent synthesis route offers transformative advantages that directly impact the bottom line and operational resilience of agrochemical manufacturers. The most significant benefit stems from the drastic reduction in the number of reaction steps, which inherently lowers the consumption of solvents, energy, and labor hours required per kilogram of finished product. By eliminating the dependency on exotic and expensive reagents like N,N-carbonyldiimidazole or hard-to-synthesize nitro-aldehydes, the bill of materials is significantly streamlined, relying instead on commodity chemicals that are readily available in the global market. This shift not only reduces the direct cost of goods sold but also mitigates the risk of supply disruptions caused by the scarcity of specialized starting materials. Furthermore, the improved overall yield of 62.0-66.5% means that less raw material is wasted, translating to substantial cost savings in raw material procurement and waste treatment overheads.
- Cost Reduction in Manufacturing: The elimination of expensive coupling agents and the reduction of synthetic steps from seven to four creates a leaner manufacturing process that inherently drives down production costs. By avoiding the use of high-cost precursors like 2-bromoheptafluoropropane in a linear fashion, the process maximizes the value extraction from each mole of fluorinated starting material. The use of common solvents such as chlorobenzene, toluene, and methanol further reduces operational expenditures compared to processes requiring specialized or hazardous solvents. Additionally, the higher yield per batch means that fixed costs related to equipment depreciation and facility overhead are distributed over a larger output of saleable product, enhancing the overall margin profile for the manufacturer.
- Enhanced Supply Chain Reliability: Sourcing stability is greatly improved as the key raw materials, including methyl acetyl (2-methyl-4-perfluoropropane phenyl) carbamate and 3-pyridine formaldehyde, are established intermediates with mature supply chains. Unlike the niche reagents required by older patents, these materials can be procured from multiple qualified vendors, reducing the risk of single-source dependency. The robustness of the reaction conditions, which operate at moderate temperatures and pressures, also ensures that production can be maintained consistently without frequent shutdowns due to equipment stress or safety incidents. This reliability allows supply chain managers to forecast production schedules with greater accuracy and maintain lower safety stock levels, thereby freeing up working capital.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard unit operations such as chlorination, distillation, and filtration that are easily replicated from pilot plant to commercial scale. The reduction in step count inherently reduces the volume of chemical waste generated, simplifying effluent treatment and lowering the environmental footprint of the facility. The use of catalytic hydrogenation with recoverable palladium on carbon aligns with green chemistry principles, minimizing heavy metal contamination in the final product and waste streams. This environmental efficiency not only reduces compliance costs but also positions the manufacturer favorably in markets with strict regulatory oversight on pesticide residues and manufacturing emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this pyridine quinazoline synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in patent CN111704604B, providing clarity on yield expectations, purity profiles, and process robustness. Understanding these details is crucial for technical directors and procurement officers evaluating the feasibility of adopting this route for commercial production. The answers reflect the specific advantages of the convergent strategy over traditional linear methods, highlighting the tangible benefits for industrial application.
Q: What is the total yield advantage of the convergent synthesis route compared to linear methods?
A: The convergent synthesis route described in patent CN111704604B achieves a total yield of 62.0-66.5%, significantly outperforming traditional linear routes which often suffer from cumulative yield losses over 5 to 7 steps.
Q: How does this method improve raw material cost efficiency?
A: By utilizing methyl acetyl (2-methyl-4-perfluoropropane phenyl) carbamate and avoiding expensive reagents like N,N-carbonyldiimidazole or difficult-to-synthesize nitro-aldehydes, the process drastically reduces raw material costs.
Q: What purity levels can be achieved with this manufacturing process?
A: Through optimized recrystallization using toluene, the final pyridine quinazoline product consistently reaches a purity content of 97.5-98.5% as determined by liquid chromatography external standard methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyridine Quinazoline Supplier
As the agrochemical industry continues to evolve towards more sustainable and cost-effective manufacturing practices, the ability to execute complex convergent syntheses at scale becomes a key differentiator for CDMO partners. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific reaction conditions required for this process, including controlled chlorination and high-pressure hydrogenation, while adhering to stringent purity specifications and rigorous QC labs to guarantee product consistency. We understand that the reliability of your supply chain depends on the technical competence of your partner, and we are committed to delivering high-purity pyridine quinazoline that meets the exacting standards of global agrochemical formulators.
We invite you to leverage our technical expertise to optimize your supply chain and reduce your manufacturing costs for this critical insecticide intermediate. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how the adoption of this convergent route can improve your margins. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can support your long-term growth objectives in the agrochemical sector.
