Optimized Industrial Synthesis of Terbinafine Hydrochloride for Global Pharmaceutical Supply Chains
The global demand for effective antifungal agents continues to drive innovation in the synthesis of Terbinafine Hydrochloride, a cornerstone therapy for onychomycosis and other dermatophyte infections. Patent CN1293037C introduces a transformative manufacturing methodology that addresses critical bottlenecks in traditional production, specifically focusing on yield optimization and process safety. This technical insight report analyzes the proprietary route which utilizes a direct hydrobromination strategy followed by a clever selective crystallization technique. By bypassing the need for expensive catalysts and complex intermediate separations, this process offers a compelling value proposition for reliable terbinafine hydrochloride supplier networks seeking to enhance their portfolio efficiency. The core innovation lies in the stabilization of the E/Z isomer ratio during the initial bromination and the subsequent exploitation of solubility differences to isolate the pharmacologically active (E)-isomer directly from the crude reaction mixture.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of Terbinafine has relied on pathways that introduce significant operational complexity and environmental burden. Prior art, such as the methods disclosed in US5817875A, often utilized propylene oxide intermediates or Wittig olefination reactions. These conventional approaches are fraught with challenges, including the requirement for stringent anhydrous conditions, the use of stoichiometric amounts of phosphorus reagents which generate difficult-to-treat waste streams, and the handling of hazardous epoxide intermediates. Furthermore, traditional routes frequently necessitate the rigorous separation of geometric isomers at the intermediate stage, often requiring energy-intensive vacuum distillation of thermally sensitive allylic halides. This not only increases the risk of product decomposition and oxidation but also drastically inflates the cost of goods sold (COGS) due to yield losses during purification. The reliance on transition metal catalysts in some alternative routes further complicates the downstream processing, mandating expensive heavy metal scavenging steps to meet stringent regulatory limits for pharmaceutical ingredients.
The Novel Approach
In stark contrast, the methodology outlined in CN1293037C presents a streamlined, catalyst-free pathway that fundamentally simplifies the synthetic logic. The process initiates with the direct reaction of 6,6-dimethyl-1-hepten-4-yn-3-ol with hydrobromic acid in the presence of methanol. This step ingeniously generates a stable mixture of (E)- and (Z)-1-bromo-6,6-dimethyl-2-hepten-4-yne in a consistent 3:1 ratio without the need for any external catalyst. The true brilliance of this approach is revealed in the subsequent condensation step, where the crude bromide mixture is reacted directly with N-methyl-1-naphthylmethylamine. Instead of attempting to separate the isomers beforehand, the process leverages the distinct physicochemical properties of the final hydrochloride salts. 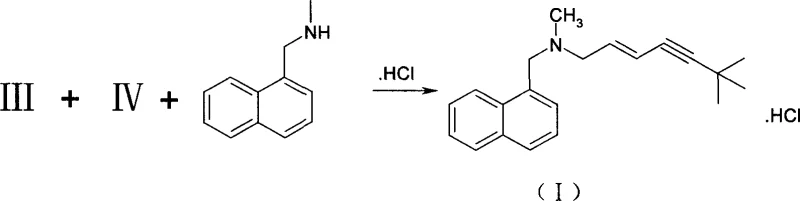 As illustrated in the reaction scheme, the introduction of hydrogen chloride gas followed by crystallization from an ethyl acetate-methanol system allows for the selective precipitation of the desired (E)-Terbinafine Hydrochloride. This eliminates the need for intermediate distillation, thereby preventing thermal degradation and significantly improving the overall mass balance of the production line.
As illustrated in the reaction scheme, the introduction of hydrogen chloride gas followed by crystallization from an ethyl acetate-methanol system allows for the selective precipitation of the desired (E)-Terbinafine Hydrochloride. This eliminates the need for intermediate distillation, thereby preventing thermal degradation and significantly improving the overall mass balance of the production line.
Mechanistic Insights into Direct Hydrobromination and Selective Salt Crystallization
The chemical elegance of this synthesis rests on the mechanistic behavior of the conjugated enyne system during the hydrobromination phase. When 6,6-dimethyl-1-hepten-4-yn-3-ol reacts with hydrobromic acid, the reaction proceeds through an allylic substitution mechanism that is facilitated by the conjugation between the double bond and the triple bond. The presence of methanol in the reaction medium plays a dual role: it acts as a co-solvent to improve the mutual solubility of the organic substrate and the aqueous acid, thereby enhancing the reaction kinetics, and it helps stabilize the transition state. The result is a stereoselective formation where the thermodynamically more stable (E)-isomer predominates over the (Z)-isomer in a reproducible 3:1 ratio. This consistency is vital for process control, as it ensures that the downstream condensation reactor always receives a feedstock of predictable composition, allowing for standardized operating parameters regardless of batch size. The absence of a catalyst not only reduces raw material costs but also removes a potential source of variable impurity profiles that could complicate regulatory filings.
Following the condensation with N-methyl-1-naphthylmethylamine, the purification mechanism shifts from kinetic control to thermodynamic solubility control. The crude reaction mixture contains both the (E)- and (Z)-isomers of the tertiary amine. Upon treatment with hydrogen chloride gas, both isomers form their respective hydrochloride salts. However, the crystal lattice energy and solvation properties of the (E)-isomer hydrochloride differ significantly from those of the (Z)-isomer in the specific solvent system of ethyl acetate and methanol. The (E)-isomer exhibits markedly lower solubility in this mixture, causing it to nucleate and grow as pure crystals while the (Z)-isomer remains dissolved in the mother liquor. This phenomenon effectively performs a 'chemical chromatography' on a macroscopic scale, achieving high purity levels without the need for silica gel columns or recrystallization from large volumes of solvent. This mechanism is critical for high-purity terbinafine hydrochloride production, ensuring that the final API meets the strict impurity thresholds required by pharmacopoeias worldwide.
How to Synthesize Terbinafine Hydrochloride Efficiently
The implementation of this synthesis route requires precise control over temperature and stoichiometry to maximize the benefits of the selective crystallization. The process is divided into two primary stages: the generation of the allylic bromide intermediate and the subsequent amine coupling followed by salt formation. Operators must maintain the bromination temperature strictly between 0°C and 50°C, with a preferred addition range of 5-10°C to manage exotherms, followed by a reaction hold at 35-40°C. For the detailed operational parameters, stoichiometry ratios, and specific workup instructions required for GMP manufacturing, please refer to the standardized protocol below.
- React 6,6-dimethyl-1-hepten-4-yn-3-ol with 48% hydrobromic acid and methanol at 0-50°C to generate a stable 3: 1 mixture of E/Z allylic bromides without catalyst.
- Condense the crude bromide mixture with N-methyl-1-naphthylmethylamine in chloroform using sodium hydroxide at 40-50°C.
- Treat the reaction mixture with hydrogen chloride gas and perform selective crystallization using an ethyl acetate-methanol system to isolate high-purity Terbinafine Hydrochloride.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patent-protected methodology translates into tangible strategic advantages beyond mere technical feasibility. The elimination of catalyst usage and the removal of intermediate distillation steps directly correlate to a reduction in utility consumption and equipment occupancy time. By simplifying the workflow from a multi-step purification sequence to a direct telescoped process, manufacturers can achieve a substantial decrease in production cycle times. This efficiency gain is crucial for cost reduction in antifungal API manufacturing, as it lowers the fixed cost allocation per kilogram of finished product. Furthermore, the use of common, readily available solvents like chloroform, ethyl acetate, and methanol mitigates supply chain risks associated with specialty reagents, ensuring a more resilient production schedule that is less susceptible to market volatility in raw material pricing.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the removal of expensive catalytic systems and the avoidance of energy-intensive separation techniques. Traditional routes often require vacuum distillation of heat-sensitive intermediates, which demands specialized glass-lined equipment and high energy inputs for heating and cooling cycles. By contrast, this novel approach relies on simple phase separation and crystallization, which are inherently lower-cost unit operations. Additionally, the high atom economy of the direct bromination step minimizes waste generation, reducing the financial burden associated with hazardous waste disposal and environmental compliance fees. The cumulative effect is a significantly leaner cost structure that allows for more competitive pricing in the generic pharmaceutical market.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the complexity of synthetic routes that depend on fragile intermediates or scarce reagents. This synthesis route enhances reliability by utilizing robust starting materials such as 6,6-dimethyl-1-hepten-4-yn-3-ol and N-methyl-1-naphthylmethylamine, which are commercially available from multiple global sources. The process tolerance is also improved; the ability to process a mixture of isomers without prior purification means that variations in the quality of the starting alcohol have less impact on the final outcome, provided the E/Z ratio remains within the expected range. This robustness reduces the frequency of batch failures and reworks, ensuring a steady flow of inventory to meet market demand without unexpected interruptions.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to multi-ton production often reveals hidden hazards, but this route is designed with scalability in mind. The reaction conditions are mild, operating near ambient pressures and moderate temperatures (below 60°C), which reduces the engineering controls required for high-pressure or cryogenic systems. From an environmental perspective, the avoidance of heavy metal catalysts and phosphorus waste aligns with modern green chemistry principles and increasingly stringent environmental regulations. The solvent system, while requiring standard recovery protocols, does not introduce exotic or persistent organic pollutants. This facilitates easier permitting for new production lines and ensures long-term operational sustainability, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is essential for stakeholders evaluating its integration into existing supply chains. The following questions address common inquiries regarding the purity profile, regulatory compliance, and operational specifics of the Terbinafine Hydrochloride process described in CN1293037C. These insights are derived directly from the patent specifications and are intended to clarify the practical implications of adopting this technology for large-scale manufacturing.
Q: How does this process avoid the need for separating E/Z isomers before the final step?
A: The process leverages the significant solubility difference between the hydrochloride salts of the E and Z isomers. By introducing hydrogen chloride gas directly into the reaction mixture and utilizing a specific ethyl acetate-methanol crystallization system, the desired (E)-isomer precipitates selectively while the (Z)-isomer remains in the mother liquor, eliminating the need for difficult distillation of the intermediate bromides.
Q: What are the safety advantages of this method compared to Wittig-based routes?
A: Unlike traditional Wittig reactions that often require strong bases and generate stoichiometric phosphorus waste, or routes using propylene oxide intermediates which involve hazardous epoxides, this method utilizes direct hydrobromination under mild conditions (0-50°C) without catalysts. This significantly reduces the handling of high-toxicity reagents and simplifies waste treatment protocols.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the route is specifically designed for industrial scalability. It features mild temperature controls (35-40°C for bromination, 40-50°C for condensation), uses common solvents like chloroform and ethyl acetate, and avoids complex purification steps like column chromatography or vacuum distillation of unstable intermediates, making it highly robust for multi-ton manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Terbinafine Hydrochloride Supplier
The technical advancements detailed in this report underscore the potential for significant efficiency gains in the production of antifungal APIs. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial realities. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of Terbinafine Hydrochloride meets the highest international standards. We understand that the transition to a new synthetic route requires confidence in the partner's capability to manage complex chemistry and deliver consistent quality.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic impact of switching to this catalyst-free methodology. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume needs, ensuring a seamless integration of high-quality Terbinafine Hydrochloride into your pharmaceutical portfolio.
