Advanced Preparation Method of Terbinafine Hydrochloride for Commercial Scale-up
Advanced Preparation Method of Terbinafine Hydrochloride for Commercial Scale-up
The pharmaceutical industry continuously seeks robust synthetic routes that balance high purity with economic efficiency, particularly for broad-spectrum antifungal agents like terbinafine hydrochloride. Patent CN114380696B introduces a significant advancement in the preparation method of this critical active pharmaceutical ingredient, addressing long-standing challenges related to isomer control and environmental impact. This innovative approach utilizes a tosylation strategy to achieve superior Z/E configuration selectivity, ensuring that the final product meets stringent quality standards required by global regulatory bodies. By optimizing reaction conditions and streamlining the workup process, this method offers a viable pathway for manufacturers aiming to enhance their supply chain stability while maintaining cost competitiveness in the market. The structural integrity of the final molecule is paramount for its efficacy in treating dermatophyte infections, and this patent provides a clear roadmap for achieving such consistency.
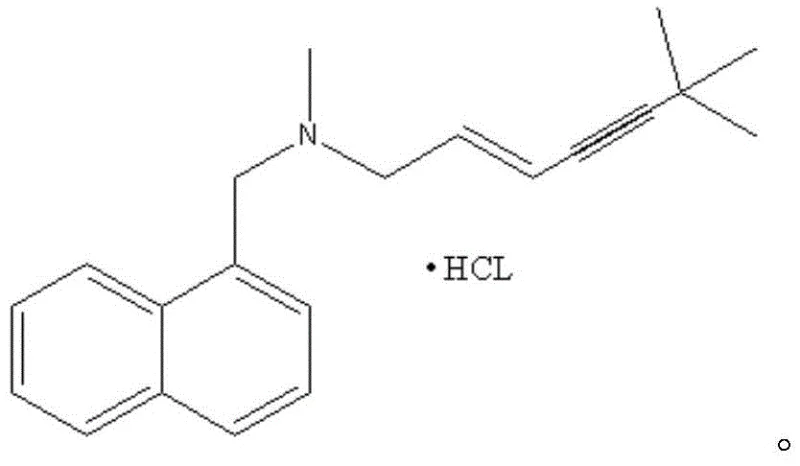
Terbinafine hydrochloride functions by inhibiting fungal squalene oxidase, leading to ergosterol deficiency and cell membrane rupture, a mechanism that requires high chemical purity to ensure patient safety and therapeutic effectiveness. The technical breakthrough described in this patent lies in the specific manipulation of the alkyne and alkene moieties within the side chain, which are critical for the drug's biological activity. Unlike previous methods that struggled with side reactions or required toxic reagents, this new protocol leverages the stability of the tosylate group to facilitate a cleaner nucleophilic substitution. For procurement managers and supply chain heads, understanding the underlying chemistry is essential, as it directly correlates to the reliability of the reliable terbinafine hydrochloride supplier partnership. The ability to consistently produce material with low isomer content translates to reduced risk of batch rejection and smoother regulatory filings for downstream drug product manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of terbinafine hydrochloride has been plagued by several technical bottlenecks that hinder efficient cost reduction in pharmaceutical intermediates manufacturing. Prior art, such as Patent CN111606811A, relies heavily on palladium chloride or cuprous iodide catalysts, which not only increase raw material costs but also introduce significant challenges in removing trace heavy metals from the final API. Furthermore, these transition metal-catalyzed reactions often require extended reaction times, sometimes exceeding 20 hours, which limits throughput and increases energy consumption in large-scale reactors. Another common issue found in older literature, like Patent CN031139198.7, is the formation of substantial amounts of Z-type cis-isomers, necessitating repeated and solvent-intensive recrystallization steps to achieve acceptable purity levels. Additionally, some routes involve the use of formaldehyde and sodium borohydride, reagents that pose significant safety hazards and environmental disposal concerns, making them less attractive for modern green chemistry initiatives. These cumulative inefficiencies result in higher production costs and longer lead times, creating vulnerabilities in the supply chain for high-purity antifungal API products.
The Novel Approach
The method disclosed in Patent CN114380696B represents a paradigm shift by eliminating the need for expensive transition metal catalysts and toxic reagents, thereby simplifying the overall process flow. By employing 4-methylbenzenesulfonyl chloride to convert the hydroxyl group into a tosylate, the reaction creates a superior leaving group that facilitates a more controlled nucleophilic attack by N-methyl-1-naphthylmethylamine. This strategic modification significantly enhances the selectivity of the reaction, ensuring that the desired E-configuration is preserved with minimal formation of unwanted isomers. The process allows for a telescoped operation where the intermediate solution can be used directly in the second step without rigorous drying, saving both time and solvent resources. This streamlined approach not only reduces the volume of wastewater generated but also minimizes the loss of intermediates, contributing to a higher overall yield and better atom economy. For industry stakeholders, this translates to a more sustainable and economically viable production model that aligns with current environmental regulations and cost-saving objectives.
Mechanistic Insights into Tosylate-Mediated Nucleophilic Substitution
The core of this synthetic innovation lies in the precise control of the nucleophilic substitution mechanism, which is critical for maintaining the stereochemical integrity of the allylamine side chain. In the first step, the reaction of 6,6-dimethyl-3-hydroxy-4-alkyne-1-heptene with 4-methylbenzenesulfonyl chloride in the presence of sodium bicarbonate generates the corresponding tosylate intermediate under mild conditions. The use of sodium bicarbonate as an acid scavenger ensures that the reaction medium remains neutral to slightly basic, preventing acid-catalyzed rearrangement or decomposition of the sensitive alkyne functionality. Cooling the reaction mixture to temperatures between -20°C and 10°C further suppresses side reactions, ensuring that the tosylate group is installed with high fidelity. This intermediate is inherently more stable and reactive towards the subsequent amine substitution compared to halide analogs, which often suffer from competing elimination reactions. The careful selection of solvents such as ethyl acetate or toluene provides an optimal environment for the dissolution of reactants while facilitating easy phase separation during the workup phase.
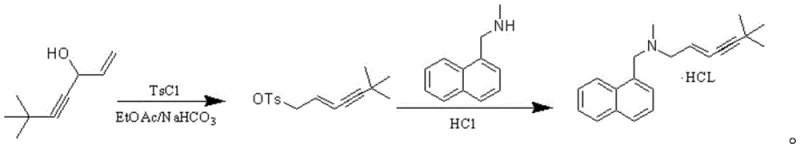
In the second step, the addition of potassium carbonate and N-methyl-1-naphthylmethylamine drives the substitution reaction to completion at elevated temperatures of 60-65°C. The carbonate base serves to deprotonate the amine, increasing its nucleophilicity and enabling it to effectively displace the tosylate group. This step is crucial for establishing the final carbon-nitrogen bond that defines the terbinafine structure, and the high selectivity of the tosylate leaving group ensures that the double bond geometry remains intact. The subsequent workup involves a simple acidification with hydrochloric acid to form the hydrochloride salt, followed by crystallization which further purifies the product. The ability to control the isomer content to within 0.05% demonstrates the robustness of this mechanistic pathway, providing a level of quality that is difficult to achieve with conventional halide-based methods. This deep understanding of the reaction mechanism allows process chemists to fine-tune parameters for commercial scale-up of complex pharmaceutical intermediates, ensuring consistent quality across different batch sizes.
How to Synthesize Terbinafine Hydrochloride Efficiently
Implementing this synthesis route requires careful attention to reaction parameters and material handling to maximize yield and purity while ensuring operator safety. The process begins with the preparation of the tosylate intermediate, where precise temperature control and stoichiometry are essential to prevent the formation of by-products. Following the formation of the intermediate, the reaction mixture can be carried forward directly, leveraging the telescoped nature of the process to minimize solvent exchange and drying steps. This operational efficiency is key to reducing the overall manufacturing footprint and energy consumption associated with the production of this antifungal agent. Detailed standard operating procedures regarding mixing speeds, addition rates, and crystallization cooling profiles are critical for reproducing the high success rates reported in the patent data. For technical teams looking to adopt this method, the following guide outlines the fundamental stages of the process.
- Prepare the tosylate intermediate by reacting 6,6-dimethyl-3-hydroxy-4-alkyne-1-heptene with 4-methylbenzenesulfonyl chloride in the presence of sodium bicarbonate at controlled low temperatures.
- Perform nucleophilic substitution by adding N-methyl-1-naphthylmethylamine and potassium carbonate to the intermediate solution, heating to 60-65°C to drive the reaction to completion.
- Isolate the final product through acidification, solvent removal, and recrystallization using isopropanol and ethyl acetate to achieve high purity terbinafine hydrochloride.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis method offers substantial strategic benefits for procurement managers and supply chain directors seeking to optimize their sourcing strategies. The elimination of transition metal catalysts removes the need for expensive scavenging resins and complex analytical testing for heavy metal residues, leading to significant cost savings in the downstream processing phase. Furthermore, the high selectivity of the reaction reduces the burden on purification units, allowing for faster batch turnover and increased production capacity without the need for additional capital investment. The use of readily available raw materials such as 6,6-dimethyl-3-hydroxy-4-alkyne-1-heptene ensures a stable supply base, mitigating the risk of raw material shortages that can disrupt production schedules. Additionally, the reduced generation of hazardous waste simplifies compliance with environmental regulations, lowering the overall cost of waste disposal and treatment. These factors collectively contribute to a more resilient and cost-effective supply chain for reliable terbinafine hydrochloride supplier partnerships.
- Cost Reduction in Manufacturing: The process achieves cost optimization by eliminating the need for precious metal catalysts and reducing the number of purification steps required to meet purity specifications. By avoiding the use of toxic reagents like formaldehyde, the facility also saves on safety equipment and specialized waste handling costs. The telescoped nature of the reaction saves solvent costs as the intermediate solution is used directly without isolation and drying. These cumulative efficiencies result in a lower cost of goods sold, allowing for more competitive pricing in the global market without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials reduces the risk of supply disruptions associated with specialized or custom-synthesized reagents. The robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality, ensuring consistent output even with fluctuating input specifications. This stability allows for more accurate production planning and inventory management, reducing the need for safety stock and freeing up working capital. Furthermore, the simplified workflow reduces the lead time for batch completion, enabling faster response to market demand fluctuations and urgent orders from pharmaceutical clients.
- Scalability and Environmental Compliance: The method is designed with industrial scalability in mind, utilizing standard reaction conditions and equipment that are easily adaptable to large-scale reactors. The reduction in wastewater volume and the absence of heavy metal contaminants make the process easier to treat and discharge, aligning with strict environmental protection standards. This green chemistry approach enhances the corporate social responsibility profile of the manufacturer, making it a preferred partner for multinational corporations with stringent sustainability goals. The ability to scale from pilot to commercial production with minimal process re-engineering ensures a smooth transition and rapid time-to-market for new product launches.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this preparation method. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this technology into their existing manufacturing portfolios. The answers are derived directly from the technical specifications and beneficial effects outlined in the patent documentation, ensuring accuracy and relevance. These insights help clarify the operational advantages and quality improvements associated with this novel synthetic route.
Q: How does this method improve Z/E configuration selectivity compared to prior art?
A: The use of the tosylate leaving group instead of halides significantly enhances stereochemical control, minimizing the formation of Z-type cis-isomers and reducing the need for complex recrystallization steps.
Q: What are the environmental benefits of this synthesis route?
A: The process eliminates the need for transition metal catalysts like palladium or copper, thereby reducing heavy metal waste and simplifying wastewater treatment protocols for industrial facilities.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the method utilizes readily available raw materials and avoids hazardous reagents like formaldehyde, making it robust and safe for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Terbinafine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving demands of the global pharmaceutical market. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the one described in Patent CN114380696B can be seamlessly translated into industrial reality. We are committed to delivering products with stringent purity specifications and maintaining rigorous QC labs to verify every batch against the highest international standards. Our infrastructure is designed to support the complex requirements of modern API manufacturing, providing a secure and reliable source for your critical raw materials. By partnering with us, you gain access to a wealth of technical expertise and production capacity that can drive your projects forward with confidence.
We invite you to engage with our technical procurement team to discuss how we can support your specific supply chain needs through a Customized Cost-Saving Analysis. Our experts are ready to provide specific COA data and route feasibility assessments tailored to your project requirements, ensuring that you have all the information needed to make informed decisions. Whether you are looking to optimize an existing supply chain or develop a new product line, our commitment to quality and efficiency makes us the ideal partner for your long-term success. Contact us today to explore how our advanced manufacturing capabilities can enhance your competitive edge in the marketplace.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
