Advanced Solvent-Free Synthesis of Terbinafine Hydrochloride for Global Pharmaceutical Manufacturing
Advanced Solvent-Free Synthesis of Terbinafine Hydrochloride for Global Pharmaceutical Manufacturing
The pharmaceutical industry continuously seeks robust, environmentally sustainable, and cost-efficient pathways for the production of critical antifungal agents. Patent CN111606811B, published in September 2022, introduces a groundbreaking preparation method for terbinafine hydrochloride that fundamentally alters the traditional manufacturing landscape. This innovation addresses the pressing need for greener chemistry by eliminating the reliance on volatile organic solvents, a common pain point in large-scale API intermediate production. The structural integrity of the final product, known chemically as (E)-N-(6,6-dimethyl-2-hepten-4-ynyl)-N-methyl-1-naphthalenemethylamine hydrochloride, is paramount for its efficacy in treating dermatophyte infections. As depicted in the chemical structure below, the molecule features a complex enyne side chain attached to a naphthalene ring, requiring precise stereochemical control during synthesis.
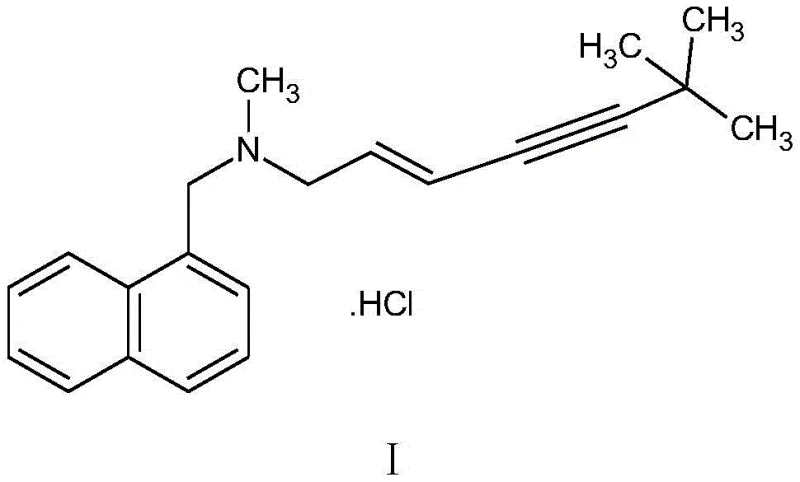
For R&D directors and process chemists, the significance of this patent lies in its ability to streamline the synthetic route while maintaining exceptional purity standards. The method utilizes a direct substitution followed by a condensation reaction, bypassing the isolation of unstable intermediates. This "one-pot" philosophy not only simplifies the operational workflow but also minimizes material loss typically associated with multiple work-up stages. By leveraging this technology, manufacturers can achieve a total molar yield exceeding 82%, a substantial improvement over conventional techniques that often struggle with lower conversion rates due to solvent interference or intermediate degradation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of terbinafine hydrochloride has been plagued by inefficiencies inherent to solvent-dependent processes. Prior art, such as the methods described in CN110423200A and CN101870655A, relies heavily on organic solvents like ethyl acetate, alcohols, or acetonitrile to facilitate the reaction between amine precursors and chloroalkynes. These traditional approaches generate substantial volumes of hazardous waste liquid, imposing heavy burdens on environmental compliance and waste treatment infrastructure. Furthermore, the necessity to isolate and purify intermediate compounds between reaction steps introduces significant opportunities for yield loss and contamination. In comparative studies, solvent-based methods have demonstrated total molar yields as low as 60.6%, indicating that nearly 40% of valuable raw materials are lost to side reactions or processing inefficiencies. The complexity of post-treatment procedures, including extraction, filtration, and solvent recovery, further escalates operational costs and extends production lead times, making these routes less attractive for modern, high-volume manufacturing.
The Novel Approach
In stark contrast, the novel approach detailed in CN111606811B revolutionizes the production workflow by adopting a solvent-free strategy. The process initiates with a substitution reaction between N-methyl-1-naphthylmethylamine and (E)-1,3-dichloropropene within an acid-binding agent medium, directly yielding the reactive intermediate without isolation. This reaction mixture is then immediately subjected to a condensation reaction with 3,3-dimethyl-1-butyne under the influence of a specialized composite catalyst. The elimination of organic solvents not only enhances the concentration of reactants, thereby driving the reaction kinetics forward, but also drastically simplifies the downstream processing. As illustrated in the reaction scheme, the transition from the starting materials to the final enyne structure is seamless, avoiding the pitfalls of intermediate handling. This methodology ensures a cleaner reaction profile, higher atom economy, and a significantly reduced environmental footprint, positioning it as the preferred choice for sustainable pharmaceutical manufacturing.
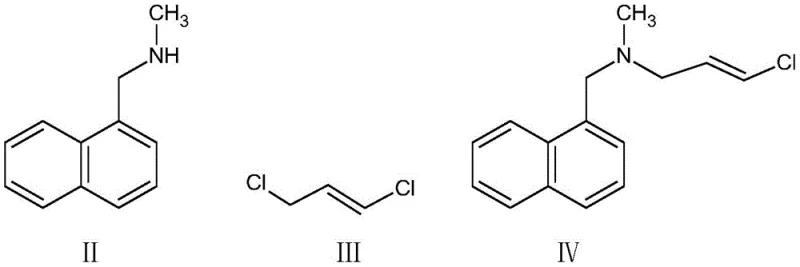
Mechanistic Insights into Pd/Cu-Catalyzed Condensation and Impurity Control
The core of this synthetic breakthrough lies in the sophisticated catalytic system employed during the condensation phase. The reaction utilizes a composite catalyst comprising palladium chloride, cuprous iodide, and triphenylphosphine, which facilitates a Sonogashira-type coupling between the chloroallyl intermediate and the terminal alkyne. In the absence of coordinating organic solvents, the acid-binding agent serves a dual purpose: neutralizing the generated hydrogen chloride and acting as the reaction medium. This unique environment promotes the formation of the carbon-carbon triple bond with high stereoselectivity, ensuring the preservation of the crucial (E)-configuration in the final product. The precise mass ratio of the catalyst components—specifically 1:10-15:3-5 for PdCl2, CuI, and PPh3—is critical for maximizing turnover frequency while minimizing the formation of homocoupling byproducts. The reaction temperature is carefully controlled between 60-65°C, a range that balances reaction rate with thermal stability, preventing the decomposition of the sensitive enyne moiety.
Impurity control is another pivotal aspect where this method excels, particularly through the innovative ammonia complexing and washing step. Following the condensation reaction, the crude mixture contains residual metal catalysts and unreacted amines which could compromise the quality of the final API. The protocol dictates concentrating the reaction solution and treating it with concentrated ammonia water. This step effectively complexes the copper and palladium species, rendering them water-soluble and easily separable from the organic oily layer containing the desired product. Subsequent washing removes these metal residues and excess acid-binding agents, resulting in an oily substance of high purity prior to salification. This rigorous purification strategy ensures that the final terbinafine hydrochloride meets stringent pharmacopeial standards for heavy metal content, a common failure point in transition-metal catalyzed syntheses. The subsequent salification in an aqueous hydrochloric acid solution at elevated temperatures further purifies the product by exploiting the differential solubility of the salt form.
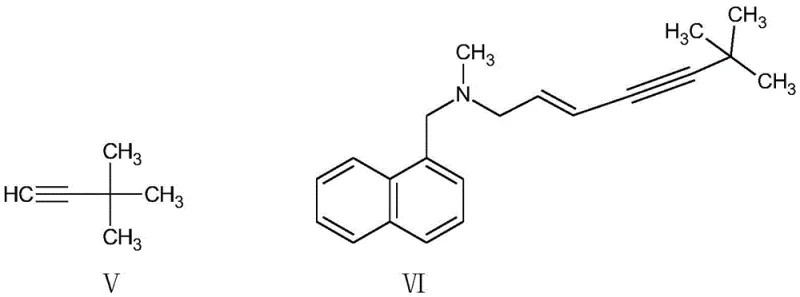
How to Synthesize Terbinafine Hydrochloride Efficiently
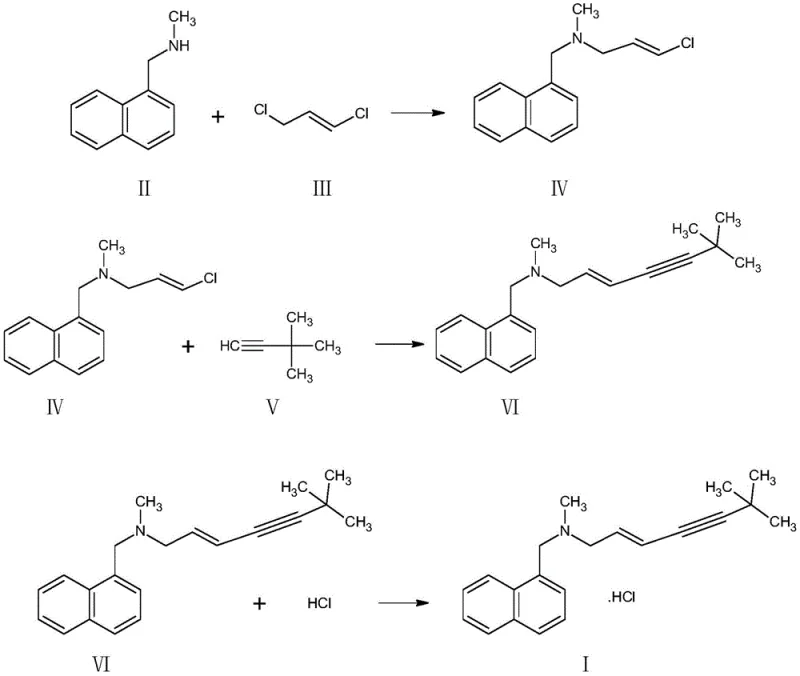
Implementing this solvent-free protocol requires precise adherence to the reaction parameters outlined in the patent to ensure reproducibility and safety. The process begins with the careful addition of (E)-1,3-dichloropropene to a mixture of N-methyl-1-naphthylmethylamine and an acid-binding agent like n-butylamine at controlled low temperatures to manage exothermicity. Once the substitution is complete, the system is cooled, and the catalyst package along with the alkyne precursor is introduced for the coupling phase. The reaction is allowed to proceed for approximately 20 hours to ensure complete conversion, monitored via TLC. Following the reaction, the work-up involves concentration, ammonia washing, and finally, salification and recrystallization from water. This streamlined sequence eliminates the need for complex solvent exchange or drying steps, making it highly amenable to automation and continuous processing. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis protocol below.
- Perform a substitution reaction between N-methyl-1-naphthylmethylamine and (E)-1,3-dichloropropene in an acid-binding agent without organic solvents to form the intermediate amine.
- Directly proceed to a condensation reaction with 3,3-dimethyl-1-butyne using a Pd/Cu composite catalyst system to generate the enyne backbone.
- Treat the reaction mixture with ammonia water for complexing and washing, followed by salification with hydrochloric acid and recrystallization to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this solvent-free synthesis route offers transformative economic and logistical benefits. The primary advantage stems from the drastic reduction in raw material consumption and waste disposal costs associated with organic solvents. By removing solvents like acetonitrile or dichloromethane from the equation, manufacturers eliminate the need for expensive solvent recovery systems and the regulatory burden of handling volatile organic compounds (VOCs). This shift translates directly into a leaner cost structure and a more resilient supply chain, less vulnerable to fluctuations in solvent pricing or availability. Furthermore, the simplified "one-pot" nature of the process reduces the number of unit operations, leading to shorter batch cycles and increased throughput capacity without the need for additional capital investment in reactor hardware.
- Cost Reduction in Manufacturing: The elimination of organic solvents results in substantial cost savings by removing the expense of purchasing, storing, and disposing of large volumes of hazardous chemicals. Additionally, the high molar yield of over 82% compared to roughly 60% in solvent-based comparative examples means that significantly less raw material is required to produce the same amount of finished product. This efficiency gain lowers the cost of goods sold (COGS) and improves overall margin potential. The reduction in waste liquid generation also minimizes the operational expenditure related to wastewater treatment and environmental compliance, further enhancing the economic viability of the process.
- Enhanced Supply Chain Reliability: The reliance on readily available, commodity-grade reagents such as n-butylamine and simple inorganic salts enhances supply chain security. Unlike specialized solvents that may face logistical bottlenecks or regional shortages, the key inputs for this method are widely sourced and stable in price. The robustness of the reaction conditions, which tolerate a broader range of operating parameters without compromising yield, ensures consistent production output. This reliability is crucial for meeting the demanding delivery schedules of global pharmaceutical clients who require uninterrupted supply of critical antifungal intermediates.
- Scalability and Environmental Compliance: The solvent-free design inherently supports safer scale-up, as the risks associated with flammable solvent vapors and large-volume storage are mitigated. This makes the process ideal for expansion from pilot scale to multi-ton commercial production. From an environmental perspective, the drastic reduction in E-factor (mass of waste per mass of product) aligns with green chemistry principles and corporate sustainability goals. The use of water as the primary medium for salification and recrystallization further underscores the eco-friendly nature of the process, facilitating easier permitting and community acceptance for manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis method. These insights are derived directly from the experimental data and technical specifications provided in patent CN111606811B. Understanding these nuances is essential for evaluating the feasibility of integrating this technology into existing production lines. The answers reflect the practical realities of scaling this solvent-free approach and highlight the specific advantages it holds over legacy methods.
Q: Does this synthesis method require organic solvents?
A: No, the patented process (CN111606811B) is distinctively solvent-free, utilizing acid-binding agents as the reaction medium, which significantly reduces waste liquid generation and environmental impact.
Q: What is the purity level achievable with this method?
A: The method consistently achieves high purity levels, with experimental data showing purity exceeding 99.6% after recrystallization, surpassing traditional solvent-based comparative examples.
Q: What catalyst system is used for the coupling step?
A: The process employs a composite catalyst system consisting of palladium chloride, cuprous iodide, and triphenylphosphine, optimized for high conversion rates in the absence of organic solvents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Terbinafine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the one described in CN111606811B for securing a competitive edge in the global pharmaceutical market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and high-quality supply of terbinafine hydrochloride. Our state-of-the-art facilities are equipped to handle solvent-free and green chemistry processes with the utmost precision, adhering to stringent purity specifications and rigorous QC labs to guarantee product integrity. We are committed to delivering not just a chemical product, but a comprehensive solution that optimizes your supply chain and reduces your environmental footprint.
We invite you to collaborate with us to leverage this cutting-edge technology for your antifungal drug development programs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this solvent-free route can enhance your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you navigate the complexities of API intermediate manufacturing with confidence, ensuring timely delivery and uncompromised quality for your critical projects.
