Revolutionizing Tedizolid Phosphate Production: A Novel Intermediate for Scalable Manufacturing
Revolutionizing Tedizolid Phosphate Production: A Novel Intermediate for Scalable Manufacturing
The pharmaceutical landscape for second-generation oxazolidinone antibiotics is constantly evolving, driven by the need for more efficient and robust manufacturing processes. Patent CN116253711A introduces a groundbreaking advancement in the synthesis of Tedizolid Phosphate, a critical antibiotic approved for treating Gram-positive bacterial infections. This patent discloses a novel intermediate, designated as Compound I, which serves as a pivotal precursor in the production of Tedizolid and its phosphate salt. Unlike traditional routes that rely on benzyl-protected intermediates with poor solubility and strict moisture sensitivity, this new approach utilizes versatile chloroformate derivatives, such as allyl or 2-ethylhexyl chloroformates. The innovation lies not just in the chemical structure but in the operational resilience it offers, allowing for higher yields and simplified purification steps that are essential for cost reduction in pharmaceutical intermediates manufacturing. By addressing the inherent limitations of prior art, this technology provides a reliable pathway for commercial scale-up of complex API intermediates, ensuring a stable supply chain for this vital medication.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Tedizolid Phosphate has been plagued by significant technical hurdles that impact both efficiency and cost. As illustrated in earlier patents like CN2009801401444 (Scheme 1), the conventional route involves a boration reaction that yields only 66% of the boronic acid intermediate with a purity of merely 89.8%. A critical bottleneck in these legacy processes is the inability to effectively remove bromine impurities during post-treatment, coupled with high levels of palladium catalyst residue that are difficult to eliminate, with recovery rates as low as 18%. Furthermore, subsequent attempts to improve this, such as the route disclosed in CN2020108676959 (Scheme 2), while achieving better yields of 85%, still rely on an N-benzyl carbamate intermediate. This specific intermediate suffers from notoriously poor solubility and extreme sensitivity to moisture; if the water content in the raw material exceeds 0.1%, the reaction conversion rate drops precipitously. These factors result in prolonged reaction times, lower overall yields of the final Tedizolid amine, and a process durability that is insufficient for robust industrial application, creating substantial risks for reducing lead time for high-purity pharmaceutical intermediates.
The Novel Approach
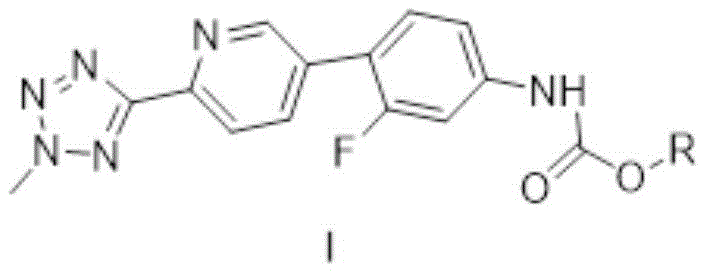
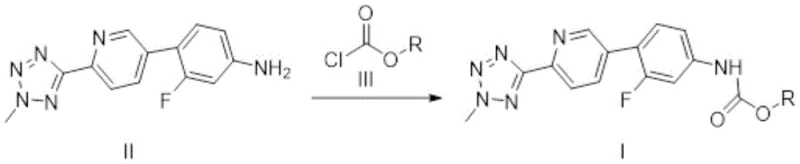
The methodology presented in Patent CN116253711A fundamentally reengineers the synthetic strategy by replacing the problematic benzyl protecting group with more labile and soluble alkyl or substituted alkyl carbamates. This novel approach involves reacting the aniline precursor (Compound II) with various chloroformate compounds to generate Compound I, where the R group can be allyl, 2-ethylhexyl, p-nitrobenzyl, or n-heptyl. This structural modification dramatically enhances the solubility of the intermediate, facilitating smoother reaction kinetics in the subsequent cyclization steps. The process is operationally simple, typically conducted in tetrahydrofuran (THF) with potassium carbonate as a base at mild temperatures ranging from 15°C to 35°C. By avoiding the stringent anhydrous conditions required by the benzyl route, this method significantly improves process tolerance and durability. The resulting intermediate allows for a direct and efficient conversion to Tedizolid, bypassing the complex recrystallization and palladium removal steps that characterize older methods, thereby establishing a new standard for a reliable API intermediate supplier seeking to optimize production workflows.
Mechanistic Insights into Chloroformate-Mediated Carbamation
The core chemical transformation in this patented process is the nucleophilic substitution reaction between the primary amine of Compound II and the electrophilic carbonyl carbon of the chloroformate (Compound III). Mechanistically, the presence of a base like potassium carbonate is crucial to deprotonate the amine, generating a more nucleophilic species that attacks the chloroformate. The choice of the R group on the chloroformate is strategic; for instance, using allyl chloroformate introduces an allyloxycarbonyl (Alloc) protecting group, which is known for its orthogonal stability and ease of removal under mild palladium-catalyzed conditions later in the synthesis, although in this specific route, it participates directly in the oxazolidinone ring formation. The reaction proceeds efficiently with an equivalent ratio of Compound II to chloroformate ranging from 1:1 to 1:2, with a preferred ratio of 1:1.6 ensuring complete conversion while minimizing excess reagent waste. The mild thermal conditions (15°C to 35°C) prevent thermal degradation of the sensitive tetrazole and pyridine moieties, preserving the integrity of the molecular scaffold. This controlled environment ensures that the formation of the carbamate linkage is highly selective, minimizing the generation of urea byproducts or over-acylation, which are common impurities in less optimized carbamation processes.
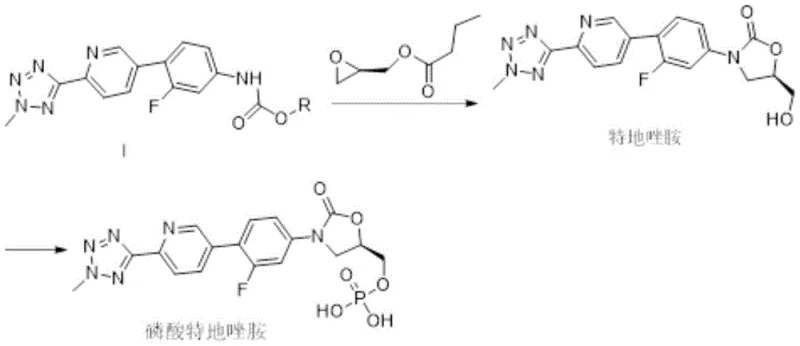
Following the formation of Compound I, the subsequent cyclization to form the oxazolidinone ring is a critical stereoselective step. This involves the use of a strong base, lithium hexamethyldisilazide (LiHMDS), to deprotonate the carbamate nitrogen, followed by nucleophilic attack on R-(-)-glycidyl butyrate. The presence of 1,3-Dimethylpropyleneurea (DMPU) as a co-solvent is mechanistically significant as it enhances the solubility of the lithium cation, increasing the reactivity of the anion and promoting the ring-closing reaction. The stoichiometry is tightly controlled, with a preferred molar ratio of Compound I to LiHMDS of 1:1.2 and to the epoxide of 1:1.4. This precise control over reaction parameters ensures high diastereoselectivity, favoring the formation of the desired (R)-enantiomer of the oxazolidinone ring. The robustness of Compound I in this step is paramount; its superior solubility compared to benzyl analogs ensures homogeneous reaction conditions, which is a key factor in maintaining consistent stereochemical outcomes and high purity profiles in the final high-purity API intermediate product.
How to Synthesize Tedizolid Phosphate Intermediate Efficiently
The synthesis of the novel intermediate Compound I represents a significant optimization in the manufacturing of Tedizolid Phosphate, offering a balance between chemical efficiency and operational simplicity. The process begins with the preparation of the aniline starting material, followed by the key carbamation step using selected chloroformates. This sequence is designed to maximize throughput while minimizing the generation of hazardous waste and difficult-to-remove impurities. The detailed standardized synthesis steps, including specific workup procedures and purification techniques required to achieve pharmaceutical-grade quality, are outlined in the comprehensive guide below. Adhering to these protocols ensures that the intermediate possesses the necessary physicochemical properties, such as enhanced solubility and moisture stability, which are critical for the success of the downstream cyclization and phosphorylation reactions.
- React 3-fluoro-4-(6-(2-methyl-2H-tetrazole-5-yl)pyridin-3-yl)aniline (Compound II) with a selected chloroformate (e.g., allyl chloroformate) in THF.
- Maintain the reaction temperature between 15°C and 35°C using potassium carbonate as the base, ensuring an equivalent ratio of 1: 1.6.
- Isolate the solid product via filtration, washing, and vacuum drying at 40°C to obtain high-purity Compound I suitable for cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers tangible strategic benefits that extend beyond mere chemical novelty. The primary advantage lies in the significant simplification of the manufacturing workflow. By utilizing intermediates with superior solubility and moisture tolerance, the process eliminates the need for rigorous drying of raw materials and complex solvent exchanges that are characteristic of the older benzyl-carbamate routes. This reduction in unit operations translates directly into shorter batch cycle times and lower energy consumption, driving substantial cost savings in utility and labor. Furthermore, the avoidance of difficult-to-remove palladium residues in the intermediate stage reduces the reliance on expensive scavenging resins or multiple recrystallization steps, streamlining the purification train and improving overall material throughput. These efficiencies collectively contribute to a more resilient supply chain capable of meeting fluctuating market demands without compromising on quality or delivery schedules.
- Cost Reduction in Manufacturing: The new process achieves cost optimization through several mechanisms, primarily by enhancing the overall yield and purity of the intermediate. The elimination of low-yielding steps, such as the problematic boration reaction in older routes which previously yielded only 66%, significantly improves the mass balance of the entire synthesis. Additionally, the use of readily available chloroformate reagents like allyl chloroformate, which are often less expensive and easier to handle than specialized benzylating agents, reduces raw material costs. The simplified workup procedure, which often requires only filtration and washing rather than column chromatography or extensive recrystallization, further lowers the cost of goods sold (COGS) by reducing solvent usage and waste disposal fees. This holistic approach to cost reduction ensures that the final API can be produced at a competitive price point, enhancing the commercial viability of the generic drug portfolio.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the robustness of the new chemical route against environmental variables. The tolerance of Compound I to moisture levels greater than 0.1% removes a critical failure point present in previous methods, where slight deviations in humidity could lead to batch failures. This increased process ruggedness reduces the risk of production delays and batch rejections, ensuring a steady flow of materials. Moreover, the reagents used in this synthesis, such as THF, potassium carbonate, and various chloroformates, are commodity chemicals with established global supply chains, mitigating the risk of raw material shortages. This reliability allows manufacturers to maintain leaner inventory levels while still guaranteeing on-time delivery to downstream partners, reinforcing the position of the manufacturer as a dependable source for critical antibiotic intermediates.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this route is inherently greener and more adaptable to large-scale production. The reaction conditions are mild, operating at near-ambient temperatures (15°C to 35°C), which reduces the energy load associated with heating or cryogenic cooling. The high selectivity of the reaction minimizes the formation of byproducts, thereby reducing the volume of organic waste generated per kilogram of product. This aligns with increasingly stringent environmental regulations and corporate sustainability goals. The simplicity of the isolation steps, primarily involving filtration and drying, makes the process easily transferable from pilot plant to multi-ton commercial reactors without the need for specialized equipment. This scalability ensures that the technology can support the growing global demand for Tedizolid Phosphate, providing a sustainable solution for long-term commercial manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel intermediate synthesis. These answers are derived directly from the experimental data and beneficial effects described in Patent CN116253711A, providing clarity on the practical implications of adopting this technology. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing capabilities.
Q: What are the advantages of the new intermediate over the benzyl carbamate route?
A: The novel intermediate (Compound I) utilizes alkyl or substituted alkyl chloroformates (like allyl or 2-ethylhexyl) instead of benzyl chloroformate. This modification significantly improves solubility and moisture tolerance, eliminating the strict water content limits (<0.1%) required by previous methods, thereby enhancing process durability and yield.
Q: How does this process address palladium residue issues found in earlier routes?
A: While the precursor synthesis may involve palladium catalysis, the new intermediate formation step itself avoids heavy metal catalysts. Furthermore, the improved purification profile of Compound I facilitates easier removal of any upstream metallic impurities compared to the complex recrystallization steps needed for the older benzyl-protected intermediates.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process is designed for industrial scalability. It operates under mild temperatures (15-35°C), uses common solvents like THF, and employs a simple workup procedure involving filtration and washing rather than complex chromatographic separations, making it highly viable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tedizolid Phosphate Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes in the modern pharmaceutical industry. Our team of expert chemists has thoroughly analyzed the technological breakthroughs presented in Patent CN116253711A and is fully prepared to leverage this knowledge for our clients. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch of Tedizolid Phosphate intermediate we produce. We are committed to delivering high-quality chemical solutions that meet the exacting standards of the global pharmaceutical market.
We invite potential partners to engage with us to explore how this novel intermediate can optimize your supply chain and reduce manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and requirements. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on concrete technical evidence. By collaborating with NINGBO INNO PHARMCHEM, you gain access to a trusted partner dedicated to advancing the availability of life-saving antibiotics through superior chemical engineering and unwavering commitment to quality.
