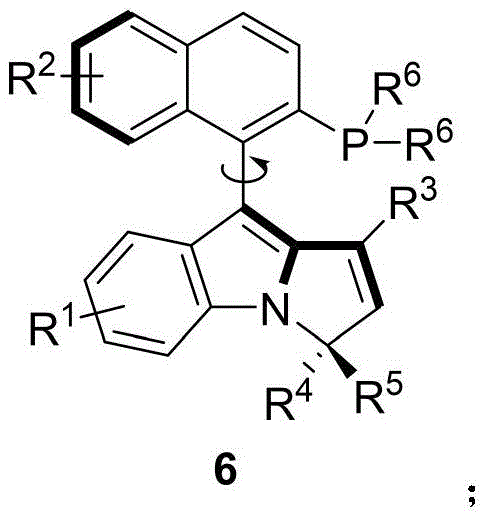
Advanced Synthesis of Axial Chiral Naphthalene-Pyrrole Phosphine Catalysts for Industrial Asymmetric Catalysis
The chemical industry is witnessing a paradigm shift in the development of chiral ligands, moving beyond traditional carbon-centered chirality towards more robust axial chiral systems. Patent CN114029086B discloses a groundbreaking class of axial chiral naphthalene-pyrrole phosphine catalysts, represented by Formula 6, which address the longstanding limitations of biaryl-based ligands. This innovation provides a versatile platform for asymmetric catalysis, offering superior stereocontrol in reactions such as hydrosilylation and allylic alkylation. For R&D directors and procurement specialists, understanding the synthetic accessibility and performance metrics of these novel ligands is critical for optimizing downstream API manufacturing processes.
Traditional methods for synthesizing axial chiral phosphine catalysts often rely on rigid biaryl scaffolds that require harsh resolution conditions or expensive chiral auxiliaries, leading to significant cost inefficiencies and supply chain bottlenecks. These conventional routes frequently suffer from low atom economy and generate substantial waste streams due to the need for multiple recrystallization steps to achieve acceptable optical purity. Furthermore, the structural rigidity of classic BINAP-type ligands can sometimes limit their substrate scope, failing to provide the necessary steric bulk or electronic tuning for challenging asymmetric transformations in complex molecule synthesis.
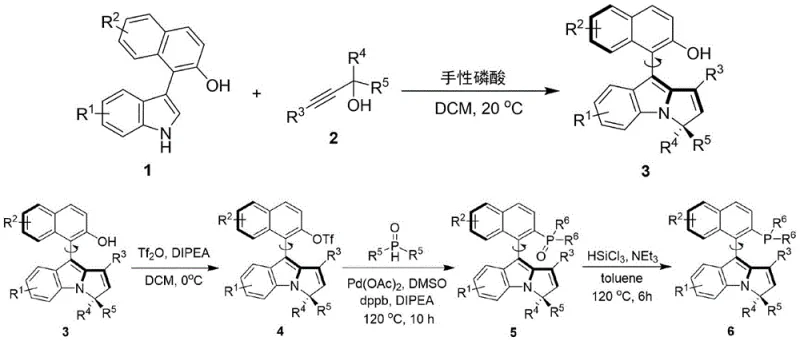
In contrast, the novel approach detailed in the patent utilizes a chiral phosphoric acid-catalyzed construction of the naphthalene-pyrrole axis, enabling the direct installation of chirality under remarkably mild conditions. This strategy bypasses the need for classical resolution, delivering high enantiomeric excess directly from the reaction mixture. The subsequent functionalization steps, including triflation and palladium-catalyzed phosphorylation, are designed for operational simplicity, utilizing common solvents like dichloromethane and toluene. This streamlined pathway not only enhances the overall yield but also significantly reduces the environmental footprint, making it an attractive candidate for green chemistry initiatives in fine chemical manufacturing.
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Axis Formation
The core of this synthetic breakthrough lies in the initial stereoselective coupling between the naphthol-indole derivative (Formula 1) and the propargylic alcohol (Formula 2). The chiral phosphoric acid acts as a bifunctional catalyst, activating the electrophile through hydrogen bonding while simultaneously directing the nucleophilic attack to establish the axial chirality with high fidelity. This mechanism ensures that the steric information from the catalyst backbone is effectively transferred to the product, resulting in enantiomeric excess values reaching up to 96% in optimized examples. The robustness of this induction step is pivotal, as it sets the stereochemical foundation for the entire ligand architecture without requiring cryogenic temperatures.
Following the axis formation, the conversion of the hydroxyl group to a triflate (Formula 4) serves as a crucial activation step for the subsequent carbon-phosphorus bond formation. The palladium-catalyzed coupling with diphenylphosphine oxide proceeds via a standard oxidative addition-reductive elimination cycle, facilitated by the electron-rich nature of the pyrrole ring. Finally, the reduction of the phosphine oxide to the active phosphine (Formula 6) using trichlorosilane is a highly efficient transformation that preserves the axial integrity. This multi-step sequence demonstrates exceptional impurity control, as the mild reaction temperatures prevent racemization and side reactions, ensuring the final catalyst meets stringent purity specifications required for sensitive pharmaceutical applications.
How to Synthesize Axial Chiral Naphthalene-Pyrrole Phosphine Catalyst Efficiently
The synthesis of these high-value ligands follows a logical four-step progression that balances stereochemical control with practical scalability. The process begins with the chiral phosphoric acid mediated coupling, followed by activation, phosphorylation, and final reduction. Each step has been optimized to minimize workup complexity, often requiring only standard extraction and silica gel chromatography. This operational simplicity is a key factor in reducing the cost of goods sold (COGS) for the final catalyst, making it accessible for both academic research and industrial scale-up.
- React naphthol-indole derivative with propargylic alcohol using chiral phosphoric acid catalyst in DCM at 20°C to form the axial chiral intermediate.
- Convert the hydroxyl group of the intermediate to a triflate using trifluoromethanesulfonic anhydride and DIPEA at 0°C.
- Perform Pd-catalyzed C-P bond formation with diphenylphosphine oxide in DMSO at 120°C to install the phosphine oxide moiety.
- Reduce the phosphine oxide to the final phosphine ligand using trichlorosilane and triethylamine in toluene at 120°C.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this synthetic route offers substantial strategic benefits regarding cost stability and supply security. The reliance on economically sourced raw materials, such as substituted naphthols and propargylic alcohols, mitigates the risk of price volatility associated with exotic chiral building blocks. Furthermore, the elimination of cryogenic conditions and the use of ambient temperature for the key stereoselective step drastically reduce energy consumption and infrastructure requirements, translating into significant operational cost savings for contract manufacturing organizations (CMOs) and internal production facilities.
- Cost Reduction in Manufacturing: The synthetic pathway eliminates the need for expensive chiral resolving agents and reduces the number of purification cycles, which directly lowers solvent usage and waste disposal costs. By avoiding extreme reaction conditions, the process minimizes equipment wear and tear and energy expenditure, leading to a more sustainable and cost-effective production model. The high yields reported in the patent examples further contribute to improved material efficiency, ensuring that every kilogram of raw material generates maximum output of the valuable chiral ligand.
- Enhanced Supply Chain Reliability: The use of robust, scalable chemistry ensures that production timelines are predictable and less susceptible to delays caused by difficult reaction controls. Since the reagents and solvents involved are commodity chemicals readily available in the global market, the risk of supply chain disruption is significantly minimized. This reliability is crucial for maintaining continuous production schedules for downstream pharmaceutical intermediates, preventing costly downtime in API manufacturing lines.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, with reaction conditions that are easily transferable from laboratory glassware to industrial reactors. The mild temperatures and standard pressure requirements simplify safety protocols and regulatory compliance, facilitating faster technology transfer. Additionally, the reduced solvent intensity and avoidance of hazardous heavy metal residues in the final product align with increasingly strict environmental regulations, supporting corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalyst technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making.
Q: What are the key advantages of this axial chiral catalyst over traditional biaryl phosphines?
A: Unlike traditional biaryl axial chiral phosphines which are often limited in structural diversity, this naphthalene-pyrrole scaffold offers a unique steric environment. The synthesis utilizes mild conditions (0-40°C) and commercially available raw materials, achieving high enantioselectivity (up to 96% ee) and yields without requiring extreme temperatures or hazardous reagents typically associated with older methods.
Q: Can this catalyst be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the patent explicitly highlights the suitability for industrial production due to simple operations and economic raw materials. The four-step sequence avoids complex purification techniques beyond standard silica gel chromatography, and the reaction conditions (e.g., 20°C for the key stereoselective step) are easily manageable in large-scale reactors, ensuring consistent supply chain reliability.
Q: What specific asymmetric reactions does this catalyst facilitate?
A: This catalyst has been validated for two distinct types of asymmetric transformations: the asymmetric hydrosilylation oxidation of styrene to produce chiral alcohols, and the asymmetric allylic alkylation coupling of allyl acetates with di-tert-butyl malonate. This versatility makes it a valuable tool for constructing chiral centers in complex organic synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axial Chiral Phosphine Catalyst Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, leveraging deep expertise in chiral catalysis to deliver high-performance ligands like the axial chiral naphthalene-pyrrole phosphine catalyst. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global pharmaceutical partners. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of catalyst delivers the expected enantioselectivity and activity, safeguarding the quality of your final drug substances.
We invite you to engage with our technical procurement team to discuss how this advanced catalyst technology can optimize your specific synthetic routes. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this novel ligand system. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to driving innovation and efficiency in your supply chain.
