Advanced Axial Chiral Phosphine Catalysts for Efficient Asymmetric Cyclization Manufacturing
Introduction to Novel Axial Chiral Catalysis Technology
The landscape of asymmetric synthesis is continuously evolving, driven by the demand for more efficient and structurally diverse chiral catalysts. Patent CN111777637B introduces a groundbreaking class of axial chiral oxindole-derived styrene phosphine catalysts, represented generally by Formula 6. This technology addresses a significant gap in the prior art, where axial chiral phosphine catalysts were predominantly limited to binaphthyl skeletons. By leveraging a unique oxindole-styrene framework, this invention provides a robust platform for catalyzing complex organic transformations, specifically asymmetric [4+2] and [3+2] cyclization reactions. The strategic design of these molecules allows for precise control over stereochemistry, making them invaluable assets for the development of high-value pharmaceutical intermediates and fine chemicals.
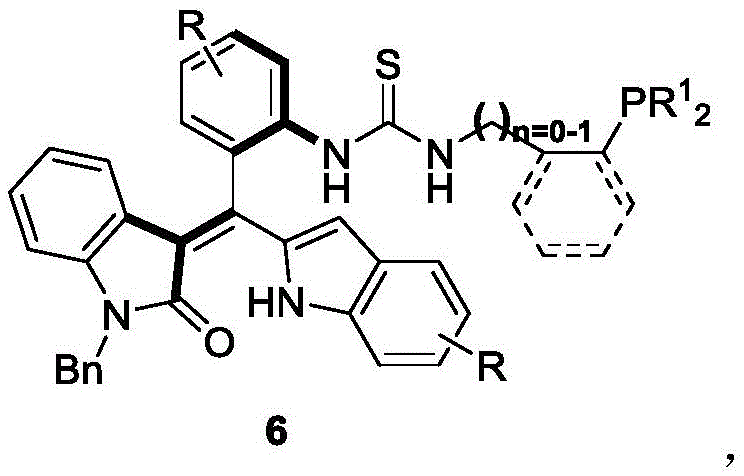
For procurement specialists and supply chain managers, the introduction of this catalyst represents a shift towards more versatile and potentially cost-effective synthetic tools. The ability to access novel chiral spaces often translates to shorter synthetic routes for active pharmaceutical ingredients (APIs), directly impacting the bottom line through reduced material costs and processing time. As a reliable chiral phosphine catalysts supplier, understanding the underlying chemistry is crucial for integrating these advanced materials into existing manufacturing pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the field of axial chiral phosphine catalysis has been dominated by binaphthyl-based structures, such as BINAP and its derivatives. While effective, these traditional scaffolds suffer from limitations in structural diversity and steric tunability. The rigid biaryl axis in binaphthyl systems can sometimes restrict the substrate scope, leading to suboptimal enantioselectivity or reactivity with bulky or electronically distinct substrates. Furthermore, the synthesis of modified binaphthyl ligands often requires harsh conditions or expensive starting materials, which can hinder cost reduction in fine chemical intermediates manufacturing. The reliance on a single structural motif also creates supply chain vulnerabilities, as global demand for these specific precursors can lead to bottlenecks.
The Novel Approach
The technology disclosed in CN111777637B circumvents these issues by introducing an oxindole-derived styrene backbone. This novel scaffold offers a distinct three-dimensional architecture that differs significantly from planar biaryl systems. The synthesis of the key intermediate (Formula 4) utilizes a chiral phosphoric acid-catalyzed reaction, which operates under mild conditions (0°C) and achieves high enantiomeric excess (up to 96% ee). This approach not only expands the library of available chiral catalysts but also provides a modular synthetic route. The subsequent conversion to the final phosphine catalyst (Formula 6) involves straightforward functionalization with thiophosgene and phosphine-containing amines, allowing for rapid optimization of the catalyst's electronic and steric properties to suit specific reaction requirements.
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Assembly
The core of this technology lies in the efficient construction of the axial chirality during the formation of intermediate Formula 4. The process employs a chiral phosphoric acid catalyst, specifically derivatives of binaphthyl or spirocyclic phosphoric acids (Formula 3), to induce asymmetry. In this step, the reaction between the axially chiral precursor (Formula 1) and the azlactone derivative (Formula 2) is meticulously controlled. The chiral phosphoric acid acts as a Brønsted acid activator, organizing the transition state through hydrogen bonding networks. This precise molecular recognition ensures that the new stereogenic axis is formed with high fidelity, as evidenced by the excellent ee values observed in the experimental data.
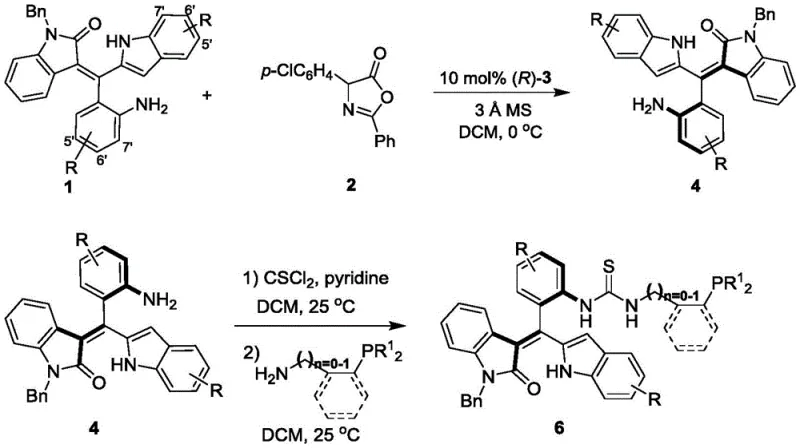
Following the establishment of axial chirality, the molecule undergoes a thiourea linkage formation to attach the phosphine moiety. The intermediate amine (Formula 4) is first converted to an isothiocyanate using thiophosgene (CSCl2) in the presence of pyridine at 25°C. This reactive species is then coupled with various phosphine-containing amines (Formula 5), such as diphenylphosphino-benzylamine or cyclohexylphosphino-methylamine. This two-step functionalization is critical for imparting the Lewis basic character required for the catalyst's activity in downstream cyclization reactions. The modularity of this step allows chemists to fine-tune the nucleophilicity of the phosphorus atom, which is essential for optimizing the catalytic cycle in asymmetric [4+2] and [3+2] cycloadditions.
How to Synthesize Axial Chiral Phosphine Catalysts Efficiently
The preparation of these high-performance catalysts follows a streamlined three-step protocol designed for reproducibility and scalability. The process begins with the enantioselective coupling of the styrene precursor and azlactone, followed by activation with thiophosgene and final coupling with the phosphine amine. Each step utilizes common organic solvents like dichloromethane and operates at near-ambient temperatures, minimizing energy consumption and safety risks associated with cryogenic or high-temperature processes. The detailed standardized synthesis steps are provided in the guide below.
- Condense axially chiral styrene precursors with azlactone derivatives using a chiral phosphoric acid catalyst in dichloromethane at 0°C to form the key intermediate.
- React the intermediate with thiophosgene in the presence of pyridine at 25°C to generate the isothiocyanate species.
- Couple the isothiocyanate with specific phosphine-containing amines in dichloromethane at 25°C to finalize the catalyst structure.
Commercial Advantages for Procurement and Supply Chain Teams
Integrating this novel catalyst technology into industrial workflows offers substantial strategic advantages for procurement and supply chain operations. The synthetic route described in the patent relies on readily available starting materials and avoids the use of scarce precious metals often found in transition metal catalysis. This shift towards organocatalytic or metal-free ligand systems significantly mitigates the risk of supply disruptions caused by geopolitical instability in mining regions. Furthermore, the mild reaction conditions reduce the burden on facility infrastructure, allowing for production in standard glass-lined reactors without the need for specialized high-pressure or extreme temperature equipment.
- Cost Reduction in Manufacturing: The elimination of expensive transition metals and the use of simple purification methods like silica gel chromatography contribute to significant cost savings. The high atom economy of the initial coupling step ensures that raw materials are utilized efficiently, reducing waste disposal costs. Additionally, the ability to achieve high yields (up to 65% for the catalyst itself and up to 91% for downstream applications) means less material is required to produce the same amount of product, directly lowering the cost of goods sold (COGS).
- Enhanced Supply Chain Reliability: The precursors for this catalyst, including isatin derivatives and various anilines, are commodity chemicals with stable global supply chains. By diversifying the catalyst portfolio to include these oxindole-derived structures, manufacturers can reduce dependency on single-source suppliers of traditional binaphthyl ligands. The robustness of the synthesis, which tolerates standard laboratory conditions, ensures consistent batch-to-batch quality, a critical factor for maintaining regulatory compliance in pharmaceutical manufacturing.
- Scalability and Environmental Compliance: The protocol is inherently scalable, having been demonstrated effectively on millimole scales with clear pathways to kilogram and multi-ton production. The use of dichloromethane and toluene, while requiring proper handling, allows for established solvent recovery protocols, aligning with green chemistry principles by minimizing solvent waste. The absence of heavy metal residues in the final catalyst structure simplifies the purification of the final API, reducing the need for costly metal scavenging steps and facilitating easier regulatory approval.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this axial chiral catalyst technology. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a clear understanding of the catalyst's capabilities and limitations in real-world applications.
Q: What distinguishes this oxindole-derived catalyst from traditional binaphthyl phosphines?
A: Unlike traditional binaphthyl scaffolds, this catalyst utilizes a novel oxindole-derived styrene backbone, offering unique steric environments and enhanced tunability for specific asymmetric transformations.
Q: What are the typical reaction conditions for synthesizing this catalyst?
A: The synthesis proceeds under mild conditions, primarily utilizing dichloromethane as a solvent with reaction temperatures ranging from 0°C to 25°C, ensuring operational safety and ease of handling.
Q: Can this catalyst be scaled for industrial production?
A: Yes, the three-step protocol involves standard organic operations such as filtration and column chromatography, making it highly amenable to commercial scale-up from kilogram to multi-ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axial Chiral Phosphine Catalyst Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis and catalyst development, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is well-versed in the nuances of chiral phosphine chemistry and can assist in optimizing the synthesis of these oxindole-derived catalysts to meet your specific purity and throughput requirements. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch of catalyst delivered meets the highest industry standards, guaranteeing consistent performance in your asymmetric synthesis campaigns.
We invite you to collaborate with us to explore the potential of this novel catalyst class in your R&D pipeline. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your project needs. We are ready to provide specific COA data and route feasibility assessments to help you accelerate your path to market with confidence and efficiency.
