Advanced Axial Chiral Phosphine Catalysts for Scalable Asymmetric Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust solutions for asymmetric synthesis, a critical step in producing high-value active pharmaceutical ingredients (APIs). Patent CN111777637B introduces a groundbreaking advancement in this field with the disclosure of a novel axial chiral oxindole-derived styrene-based phosphine catalyst, structurally defined as Formula 6. This innovation addresses the long-standing limitations of traditional carbon-centered chiral phosphine catalysts by leveraging a rigid axially chiral backbone. The structural uniqueness of this catalyst, as depicted below, provides a distinct steric environment that enhances stereocontrol in complex organic transformations. For R&D directors and procurement specialists, this represents a significant opportunity to access high-purity chiral building blocks through a more efficient and scalable synthetic route.
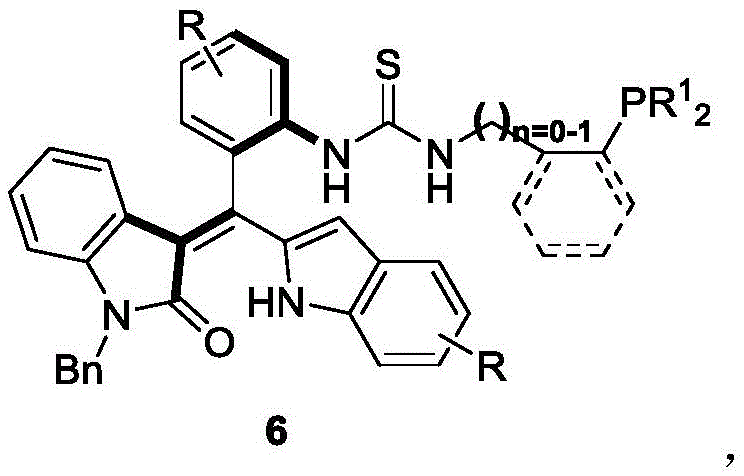
The development of this catalyst fills a critical gap in the existing literature, where axial chiral phosphine catalysts were predominantly restricted to binaphthyl skeletons. By shifting the chiral axis to an oxindole-styrene framework, the inventors have created a versatile platform capable of catalyzing asymmetric [4+2] and [3+2] cyclization reactions with remarkable efficiency. The patent details a streamlined three-step preparation method that avoids harsh conditions, utilizing mild temperatures ranging from 0°C to 25°C. This approach not only simplifies the manufacturing process but also significantly reduces the energy consumption associated with catalyst production. For supply chain managers, the reliance on commercially available starting materials and standard solvents like dichloromethane ensures a stable and continuous supply line, mitigating risks associated with specialized reagent shortages.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the landscape of chiral phosphine catalysis has been dominated by biaryl systems, such as BINAP and its derivatives, which rely on a binaphthyl axial chirality. While effective, these conventional catalysts often suffer from limited structural diversity, restricting their applicability to specific substrate classes. Furthermore, the synthesis of traditional biaryl phosphine ligands frequently involves multi-step sequences requiring expensive transition metal catalysts for coupling reactions, which introduces the risk of heavy metal contamination in the final product. This contamination necessitates additional purification steps, such as scavenging or recrystallization, thereby increasing both the cost and the lead time for API manufacturing. Additionally, the rigidity of the binaphthyl backbone can sometimes be too restrictive, preventing the catalyst from adapting to bulky or electronically diverse substrates, leading to lower yields or poor enantioselectivity in complex cyclization reactions.
The Novel Approach
In contrast, the novel approach described in patent CN111777637B utilizes an oxindole-derived styrene scaffold that offers a unique combination of rigidity and tunability. The synthesis begins with a kinetic resolution or asymmetric construction of the axially chiral styrene intermediate (Formula 4) using a chiral phosphoric acid catalyst, achieving high enantiomeric excess values up to 96%. This intermediate is then functionalized via a thiocarbonylation step followed by coupling with various phosphine-containing amines. The resulting catalyst (Formula 6) possesses a thiourea linkage that can participate in hydrogen bonding, potentially activating substrates through dual activation modes. This structural feature allows for superior control over the stereochemical outcome of reactions like the asymmetric [4+2] cyclization of allenoates. The entire process is conducted under mild conditions, eliminating the need for cryogenic temperatures or inert atmospheres beyond standard protection, which drastically simplifies the operational complexity for commercial scale-up.
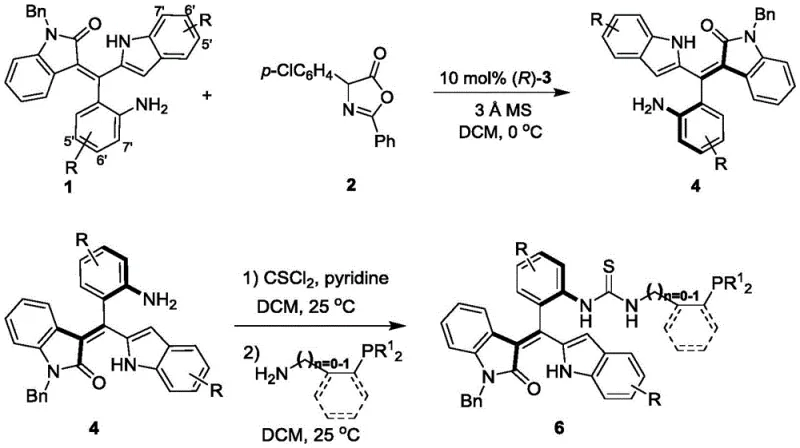
Mechanistic Insights into Axial Chirality Induction and Catalytic Cycle
The core mechanistic advantage of this technology lies in the precise control of axial chirality during the initial synthesis step. The reaction between the oxindole-derived precursor (Formula 1) and the electrophilic partner (Formula 2) is mediated by a chiral phosphoric acid catalyst, such as the binaphthyl derivative (Formula 3). This catalyst creates a chiral pocket that discriminates between the pro-chiral faces of the reacting species, locking the rotational freedom of the emerging styrene bond into a single atropisomer. The presence of molecular sieves in the reaction mixture plays a crucial role in water scavenging, driving the equilibrium towards product formation and preventing hydrolysis of sensitive intermediates. Once the axially chiral backbone is established in Formula 4, the subsequent conversion to the phosphine catalyst preserves this stereochemical integrity. The thiourea moiety introduced in the final steps acts as a hydrogen bond donor, which can coordinate with electron-deficient groups on the substrate, aligning them perfectly for nucleophilic attack by the phosphine center. This dual-activation mechanism ensures that the subsequent cyclization reactions proceed with high regioselectivity and diastereocontrol.
Impurity control is inherently built into this mechanistic design. The mild reaction temperatures (0°C for the chiral induction step and 25°C for the functionalization) minimize side reactions such as polymerization or decomposition of the sensitive allenoate or isatin substrates. Furthermore, the use of dichloromethane as a solvent facilitates easy workup and purification via silica gel column chromatography, allowing for the removal of unreacted starting materials and minor diastereomers. The high atom economy of the coupling steps ensures that waste generation is kept to a minimum, aligning with green chemistry principles. For quality assurance teams, the distinct NMR signatures of the axial chiral backbone provide a reliable analytical handle for verifying batch-to-batch consistency. The robust nature of the oxindole ring system also contributes to the thermal stability of the catalyst, ensuring it remains active throughout the duration of the catalytic cycle without degrading into inactive phosphine oxides.
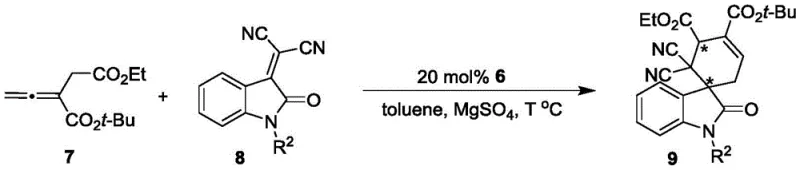
How to Synthesize Axial Chiral Phosphine Catalyst Efficiently
The synthesis of this advanced catalyst is designed for operational simplicity and reproducibility, making it ideal for both laboratory optimization and pilot plant production. The process relies on a convergent strategy where the chiral backbone is constructed first, followed by the installation of the catalytic phosphine headgroup. Detailed standardized synthesis protocols, including specific molar ratios, solvent volumes, and purification parameters, are essential for maintaining the high enantiomeric purity observed in the patent examples. The following guide outlines the critical operational phases required to achieve the reported yields and selectivity.
- React Formula 1 and Formula 2 in dichloromethane with chiral phosphoric acid catalyst at 0°C to obtain axially chiral intermediate Formula 4.
- Treat Formula 4 with thiophosgene and pyridine in dichloromethane at 25°C to form the isothiocyanate intermediate.
- Couple the intermediate with amine Formula 5 in dichloromethane at 25°C to yield the final phosphine catalyst Formula 6.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel catalyst technology offers substantial strategic benefits beyond mere technical performance. The shift away from precious metal catalysts to organocatalysis eliminates the regulatory burden associated with heavy metal residue limits in final drug substances. This transition significantly streamlines the validation process and reduces the cost of goods sold (COGS) by removing expensive metal scavenging resins from the production workflow. Moreover, the synthetic route utilizes commodity chemicals and solvents that are readily available in the global market, insulating the supply chain from volatility associated with rare earth elements or specialized ligands. The mild reaction conditions also translate to lower energy costs, as there is no need for extensive cooling infrastructure or high-pressure equipment, further enhancing the economic viability of large-scale manufacturing.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts from the synthetic sequence removes a major cost driver typically associated with chiral phosphine ligand production. By relying on organocatalytic steps for the initial chirality induction, the process avoids the use of expensive palladium or rhodium complexes, leading to substantial cost savings in raw material procurement. Additionally, the simplified workup procedures, which do not require specialized metal removal steps, reduce the consumption of auxiliary materials and shorten the overall production cycle time. This efficiency gain allows for a more competitive pricing structure for the final chiral intermediates, providing a clear margin advantage in high-volume contracts.
- Enhanced Supply Chain Reliability: The starting materials for this catalyst, including oxindole derivatives and simple amines, are produced by multiple suppliers globally, ensuring a diversified and resilient supply base. Unlike proprietary biaryl ligands that may be sourced from a single vendor, the modular nature of this oxindole-styrene scaffold allows for flexible sourcing strategies. The robustness of the synthesis against minor variations in temperature and moisture content further reduces the risk of batch failures, ensuring consistent delivery schedules. This reliability is critical for maintaining continuous API production lines and avoiding costly downtime due to catalyst shortages or quality deviations.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability potential due to its reliance on homogeneous reaction conditions and standard solvent systems like dichloromethane and toluene. The absence of exothermic hazards associated with metal-hydride reductions or strong bases makes the scale-up from gram to kilogram scale straightforward and safe. From an environmental perspective, the high atom economy and the ability to recycle solvents contribute to a reduced environmental footprint, facilitating compliance with increasingly stringent waste disposal regulations. This sustainability profile enhances the corporate social responsibility (CSR) standing of the manufacturing site and aligns with the green chemistry goals of major pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this axial chiral phosphine catalyst technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a clear understanding of its practical utility and limitations in industrial settings.
Q: What distinguishes this oxindole-derived catalyst from traditional biaryl phosphine catalysts?
A: Unlike traditional biaryl catalysts limited to binaphthyl skeletons, this novel catalyst utilizes an axially chiral oxindole-styrene backbone, offering a unique steric environment and potentially broader substrate scope for asymmetric cyclizations.
Q: How does the synthesis process ensure high enantiomeric excess?
A: The process employs a highly selective chiral phosphoric acid catalyst in the initial resolution step at low temperatures (0°C), achieving enantiomeric excess values up to 96% for the key intermediate before final functionalization.
Q: Is this catalyst suitable for large-scale industrial production?
A: Yes, the synthesis utilizes mild reaction conditions (0°C to 25°C), common solvents like dichloromethane, and avoids expensive transition metals, making it highly scalable and cost-effective for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axial Chiral Phosphine Catalyst Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the axial chiral oxindole-derived phosphine catalysts disclosed in patent CN111777637B for the next generation of asymmetric synthesis. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate these innovative laboratory protocols into robust commercial processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high enantioselectivity and yield reported in the patent are maintained at an industrial scale. We operate stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of catalyst meets the exacting standards required for GMP API manufacturing.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific synthetic challenges. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your project's volume and timeline requirements. By partnering with us, you gain access to specific COA data and comprehensive route feasibility assessments that will accelerate your development timeline. Contact us today to discuss how our supply of high-purity axial chiral phosphine catalysts can optimize your asymmetric synthesis workflow and drive down your overall manufacturing costs.
