Optimizing Gefitinib Production: A Novel Eco-Friendly Synthetic Route for Commercial Scale-Up
Introduction to Advanced Gefitinib Synthesis Technology
The global demand for high-quality epidermal growth factor receptor (EGFR) tyrosine kinase inhibitors continues to surge, driven by the critical role of Gefitinib in treating non-small cell lung cancer. As a leading manufacturer, we recognize that the efficiency of API production hinges on the robustness of the synthetic pathway employed. Patent CN102146060A discloses a groundbreaking preparation method for Gefitinib and its key intermediates that fundamentally rethinks the construction of the quinazoline scaffold. Unlike legacy processes that rely on harsh halogenation conditions, this innovative approach prioritizes environmental sustainability and operational simplicity by strategically delaying the introduction of the morpholine moiety.
This technical insight report analyzes the specific chemical advancements detailed in the patent, offering R&D directors and procurement specialists a clear view of how this methodology translates into tangible supply chain advantages. By connecting the 3-halopropyl side chain prior to the synthesis of the quinazoline parent ring and reserving the morpholine coupling for the final transformation, the process circumvents the tedious pH adjustments that plague conventional routes. This strategic sequencing not only enhances the purity profile of the final active pharmaceutical ingredient but also streamlines the isolation of intermediates, setting a new benchmark for reliable pharma intermediate supplier capabilities in the oncology sector.
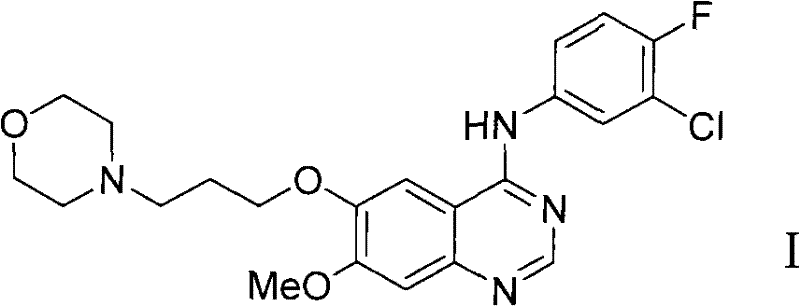
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic pathways for Gefitinib, such as those disclosed in earlier patent applications like CN101348471A, typically involve a linear nine-step sequence starting from isovanillin. A critical bottleneck in these legacy routes is the reliance on highly polluting halogenating agents, specifically thionyl chloride or phosphorus oxychloride, to convert the quinazolinone intermediate into the reactive 4-chloroquinazoline species. This chlorination step generates substantial amounts of acidic waste and requires specialized corrosion-resistant reactor infrastructure, driving up both capital expenditure and operational costs. Furthermore, these conventional methods often introduce the morpholine ring relatively early in the synthesis, creating a basic center that interferes with subsequent purification steps.
The presence of the basic morpholine nitrogen necessitates repeated and precise adjustments of pH values during the isolation of reaction products at multiple stages. This constant oscillation between acidic and basic conditions complicates the workup procedure, leading to significant product loss through emulsion formation or solubility issues in aqueous phases. Consequently, the overall yield of these traditional routes remains suboptimal, often struggling to exceed 35% over the full sequence. For procurement managers focused on cost reduction in API manufacturing, these inefficiencies translate directly into higher raw material consumption and increased waste disposal fees, making the legacy process economically unsustainable for large-scale commercial production.
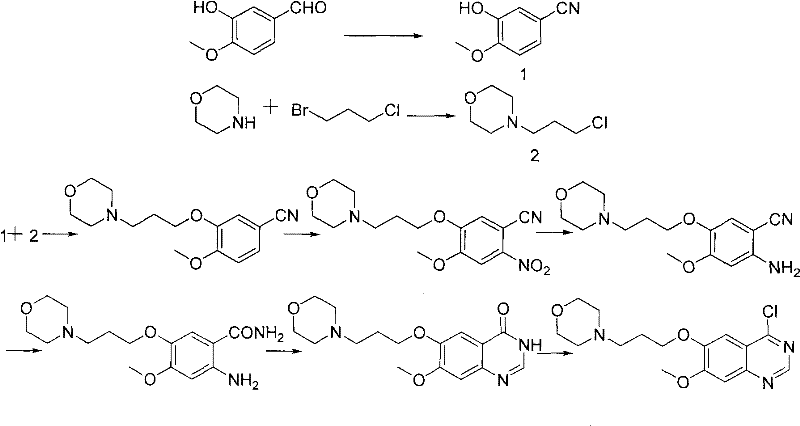
The Novel Approach
The methodology presented in CN102146060A offers a transformative solution by inverting the traditional order of operations. Instead of constructing the quinazoline ring first and then functionalizing it, this novel approach begins with the alkylation of 3-hydroxy-4-methoxybenzonitrile to install the halopropyl side chain immediately. This early installation ensures that the side chain is already in place before the heterocyclic ring is closed, allowing for a more convergent synthesis strategy. Crucially, the cyclization step utilizes N,N-dimethylformamide dimethyl acetal (DMF-DMA) in the presence of 3-chloro-4-fluoroaniline to directly form the 4-anilinoquinazoline structure, thereby completely bypassing the need for a separate 4-chloro intermediate and the associated toxic halogenating reagents.
By deferring the introduction of the morpholine ring until the absolute final step of the synthesis, this route elegantly sidesteps the purification nightmares associated with basic intermediates. The precursors leading up to the final substitution remain neutral or weakly acidic/basic, allowing for straightforward crystallization and filtration without the need for complex acid-base extractions. This simplification of the post-treatment process not only accelerates the production cycle but also significantly boosts the recovery rate of valuable intermediates. The result is a cleaner, greener, and more cost-effective pathway that aligns perfectly with modern green chemistry principles while delivering the high-purity standards required for regulatory approval.
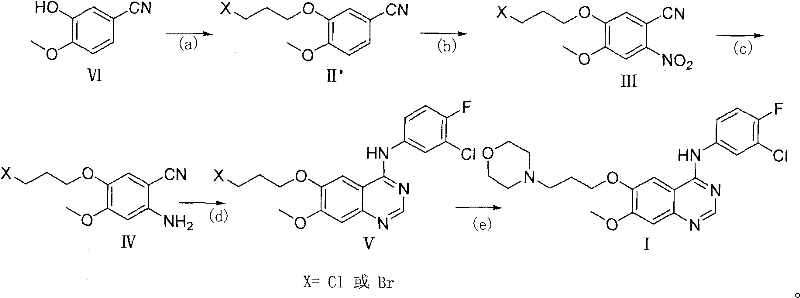
Mechanistic Insights into DMF-DMA Mediated Cyclization
The cornerstone of this improved synthesis lies in the mechanistic elegance of the cyclization step, where the 2-amino-benzonitrile intermediate reacts with DMF-DMA and the aniline derivative. In this transformation, DMF-DMA acts as a one-carbon synthon, reacting with the ortho-amino group to form an amidine intermediate in situ. This reactive species then undergoes intramolecular cyclization with the nitrile group, facilitated by the acidic conditions provided by acetic acid, to close the pyrimidine ring of the quinazoline system. This one-pot procedure effectively merges the formylation and cyclization events, reducing the step count and minimizing the exposure of sensitive intermediates to harsh conditions that could lead to degradation or byproduct formation.
From an impurity control perspective, this mechanism is superior because it avoids the formation of the 4-chloro impurity profile entirely. In traditional routes, incomplete chlorination or hydrolysis of the 4-chloro group can lead to difficult-to-remove quinazolinone contaminants. By directly forming the 4-anilino bond during ring closure, the thermodynamic stability of the product drives the reaction to completion with high selectivity. Furthermore, the subsequent nucleophilic substitution of the terminal chloride on the propyl side chain with morpholine is a clean SN2 reaction. Since this occurs as the final step, any unreacted morpholine or halogenated byproducts can be easily removed during the final recrystallization, ensuring the stringent purity specifications demanded by global pharmacopeias are met with consistency.
How to Synthesize Gefitinib Efficiently
The implementation of this synthetic strategy requires precise control over reaction parameters, particularly during the nitration and reduction phases which set the stage for the final cyclization. The process begins with the O-alkylation of the phenolic starting material using 1,3-dihalopropane under basic conditions, followed by a regioselective nitration that must be carefully temperature-controlled to prevent over-nitration. The subsequent reduction of the nitro group to the amine is preferably conducted using sodium dithionite, a cost-effective and safe reducing agent that avoids the safety hazards associated with catalytic hydrogenation on a large scale. These preparatory steps generate the critical 2-amino-4-methoxy-5-(3-halopropoxy)benzonitrile intermediate, which serves as the direct precursor for the quinazoline assembly.
- Alkylation of 3-hydroxy-4-methoxybenzonitrile with 1,3-dihalopropane to form the halopropoxy intermediate.
- Regioselective nitration followed by reduction to generate the key 2-amino-benzonitrile precursor.
- One-pot cyclization using DMF-DMA and 3-chloro-4-fluoroaniline to construct the quinazoline core.
- Final nucleophilic substitution with morpholine to yield high-purity Gefitinib.
For detailed operational parameters, including specific solvent ratios, temperature gradients, and stoichiometric equivalents for each transformation, please refer to the standardized synthesis guide below. This guide encapsulates the optimized conditions derived from the patent examples, ensuring reproducibility and safety for pilot and commercial scale operations. Adhering to these protocols allows manufacturers to maximize yield while maintaining the environmental benefits inherent to this halogen-free approach.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement officers, the adoption of this synthetic route offers profound logistical and financial benefits that extend beyond simple yield improvements. The elimination of phosphorus oxychloride and thionyl chloride removes a major regulatory burden, as these reagents are subject to strict monitoring and reporting requirements due to their potential use in chemical warfare agents. By sourcing a process that is inherently free of these controlled substances, companies can streamline their compliance documentation and reduce the risk of supply disruptions caused by regulatory audits or shipping restrictions on hazardous materials.
- Cost Reduction in Manufacturing: The economic impact of this route is driven primarily by the simplification of downstream processing. Because the morpholine ring is introduced late, the intermediate purification steps do not require the consumption of large volumes of acids and bases for pH swinging, nor do they require extensive aqueous washing to remove salts. This reduction in auxiliary chemical consumption lowers the variable cost per kilogram significantly. Additionally, the higher overall yield means that less starting material is required to produce the same amount of finished API, directly improving the margin structure for the final drug product.
- Enhanced Supply Chain Reliability: The starting materials for this route, such as 3-hydroxy-4-methoxybenzonitrile and common alkylating agents, are commodity chemicals with robust global supply networks. Unlike specialized catalysts or exotic reagents that might have single-source suppliers, the inputs for this process are widely available, mitigating the risk of raw material shortages. The robustness of the chemistry also means that the process is less sensitive to minor variations in reagent quality, ensuring consistent batch-to-batch performance even when sourcing from different vendors.
- Scalability and Environmental Compliance: From an EHS (Environment, Health, and Safety) perspective, this process is designed for scalability. The absence of highly corrosive halogenating gases simplifies the engineering requirements for the reactor vessels, allowing for the use of standard stainless steel equipment rather than glass-lined or Hastelloy reactors. This lowers the barrier to entry for contract manufacturing organizations (CMOs) and facilitates faster technology transfer. Furthermore, the reduced waste load makes it easier to meet increasingly stringent environmental discharge limits, future-proofing the manufacturing site against tightening regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Gefitinib synthesis pathway. These answers are derived directly from the experimental data and beneficial effects described in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for R&D teams evaluating technology transfer and for quality assurance teams establishing control strategies.
Q: How does this novel route improve environmental compliance compared to traditional methods?
A: This method completely avoids the use of highly polluting halogenating reagents such as thionyl chloride or phosphorus oxychloride, which are typically required to generate the 4-chloroquinazoline intermediate in conventional routes. By eliminating these hazardous chemicals, the process significantly reduces toxic waste generation and simplifies effluent treatment protocols.
Q: Why is the morpholine ring introduced in the final step?
A: Introducing the basic morpholine ring at the very last step prevents the need for repeated pH adjustments during the purification of earlier intermediates. In traditional routes where morpholine is added early, the basic nitrogen complicates extraction and crystallization, requiring extensive acid-base workups that lower overall yield and increase processing time.
Q: What are the key advantages for large-scale manufacturing of this API intermediate?
A: The process features fewer reaction steps and higher overall yields due to simplified purification protocols. The avoidance of corrosive halogenating agents reduces equipment maintenance costs, while the robust crystallization properties of the intermediates ensure consistent quality control suitable for multi-ton commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gefitinib Supplier
At NINGBO INNO PHARMCHEM, we combine deep chemical expertise with industrial-scale capability to deliver high-value pharmaceutical intermediates. Our technical team has extensively analyzed the route described in CN102146060A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We understand that transitioning to a new synthetic route requires rigorous validation; therefore, our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications and characterizing impurity profiles to ensure full regulatory compliance for your filings.
We invite you to collaborate with us to leverage this advanced technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data from our pilot batches and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing process can enhance your competitiveness in the global oncology market.
