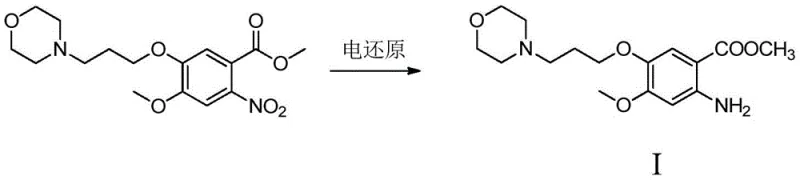
Revolutionizing Gefitinib Intermediate Synthesis via Green Electroreduction for Commercial Scale-Up
Revolutionizing Gefitinib Intermediate Synthesis via Green Electroreduction for Commercial Scale-Up
The pharmaceutical industry is currently witnessing a paradigm shift towards greener, more sustainable manufacturing processes, particularly in the synthesis of complex oncology intermediates like those required for Gefitinib. Patent CN110777391A introduces a groundbreaking electroreduction preparation method that fundamentally alters the production landscape for methyl 2-amino-4-methoxy-5-(3-morpholinopropoxy)benzoate, a critical precursor in the tyrosine kinase inhibitor supply chain. This innovative approach replaces traditional, hazardous chemical reductants and high-pressure catalytic hydrogenation with a precise, electricity-driven reduction mechanism that operates under mild conditions. By leveraging the power of electrons as the primary reducing agent, this technology not only enhances the environmental profile of the synthesis but also significantly improves the control over reaction selectivity and product purity. For global procurement leaders and R&D directors, understanding the implications of this patent is crucial for securing a reliable API intermediate supplier capable of delivering high-quality materials while adhering to increasingly strict regulatory standards regarding heavy metals and process safety.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this key gefitinib intermediate has relied heavily on catalytic hydrogenation using precious metal catalysts such as 10% Pd/C or chemical reduction using inorganic agents like sodium dithionite. While these methods can achieve reasonable yields, they are fraught with significant technical and commercial drawbacks that hinder efficient large-scale manufacturing. Catalytic hydrogenation typically requires high-pressure equipment, often operating at pressures around 50 psi or higher, which imposes severe safety constraints and necessitates expensive, specialized reactor infrastructure. Furthermore, the use of palladium catalysts introduces a persistent risk of heavy metal contamination, as the catalyst can form difficult-to-separate complexes with the intermediate, potentially leading to products that exceed strict pharmacopeial limits for residual metals. Alternatively, chemical reduction with sodium dithionite generates substantial amounts of inorganic waste and sulfur-containing by-products, creating a heavy burden on wastewater treatment facilities and contradicting modern green chemistry principles.
The Novel Approach
In stark contrast to these legacy methods, the electroreduction technique disclosed in the patent offers a streamlined, environmentally benign pathway that directly addresses the痛点 of traditional synthesis. By utilizing a diaphragm electrolytic cell with an acidic aqueous solution as the electrolyte, the process achieves the reduction of the nitro group to an amino group without the need for external chemical reducing agents or organic solvents. The core innovation lies in the precise control of electrode potential, typically maintained between 1.00V and 2.50V relative to a reference electrode, which allows operators to finely tune the conversion rate and selectivity. This level of control ensures that the reaction proceeds efficiently to the desired amine product while minimizing the formation of unwanted side products such as azo or hydrazo compounds. The result is a robust process that operates at atmospheric pressure and moderate temperatures between 25°C and 80°C, drastically simplifying the engineering requirements and enhancing the overall safety profile of the manufacturing facility.
Mechanistic Insights into Electrochemical Nitro Reduction
To fully appreciate the technical superiority of this method, one must delve into the electrochemical mechanisms governing the transformation of the nitro substrate into the target amine. The process occurs at the cathode surface, where the nitro compound accepts electrons in a stepwise manner, progressing through nitroso and hydroxylamine intermediates before finally yielding the primary amine. The patent explicitly details the potential pitfalls of this pathway, noting that improper control of the electrode potential can lead to the condensation of intermediates, resulting in the formation of azo (structure 4) or hydrazo (structure 5) by-products, as illustrated in the reaction scheme. However, by strictly maintaining the cathode current density within the optimal range of 25.0mA/cm² to 250.0mA/cm² and utilizing a strong acid cation exchange membrane to separate the anode and cathode compartments, the process effectively suppresses these side reactions. This precise electrochemical environment ensures that the protonation steps keep pace with the electron transfer, driving the reaction selectively towards the formation of methyl 2-amino-4-methoxy-5-(3-morpholinopropoxy)benzoate with exceptional fidelity.
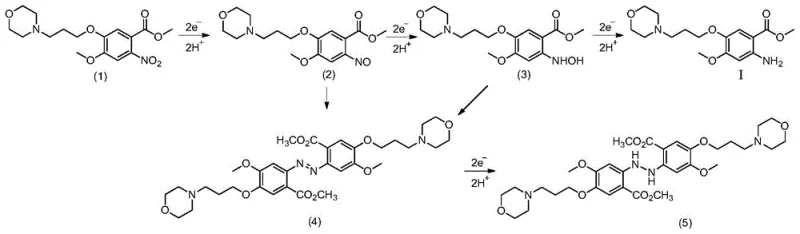
Furthermore, the choice of electrode materials plays a pivotal role in the efficiency and longevity of the system. The patent specifies the use of durable cathode materials such as copper, titanium mesh, nickel, or graphite, paired with Dimensionally Stable Anodes (DSA) composed of metal oxides like ruthenium or iridium on a titanium substrate. This combination provides a stable interface for electron transfer while resisting corrosion in the acidic electrolyte environment, which typically consists of phosphoric, sulfuric, or hydrochloric acid at concentrations between 0.15mol/L and 1.00mol/L. The acidic medium serves a dual purpose: it ensures the solubility of the organic substrate and provides the necessary protons for the reduction half-reaction. At the anode, water oxidation generates oxygen and protons, which migrate through the membrane to maintain charge balance, creating a closed-loop system that minimizes waste generation. This mechanistic elegance translates directly into operational reliability, as the system avoids the deactivation issues common with heterogeneous chemical catalysts.
How to Synthesize Methyl 2-amino-4-methoxy-5-(3-morpholinopropoxy)benzoate Efficiently
Implementing this electroreduction strategy requires a systematic approach to reactor setup and parameter optimization to ensure consistent batch-to-batch quality. The process begins with the preparation of the catholyte, where the nitro precursor is dissolved in the chosen acidic aqueous medium at a concentration optimized for both solubility and conductivity, typically ranging from 4.0g/L to 16.0g/L. Once the electrolytic cell is assembled with the appropriate membrane and electrodes, the potential is applied, and the reaction progress is monitored using thin-layer chromatography (TLC) with a petroleum ether and ethyl acetate mobile phase. The reaction is terminated precisely when the starting material spot disappears, indicating complete conversion, after which the product is isolated through a straightforward workup involving pH adjustment and solvent extraction. For a comprehensive breakdown of the specific operational parameters and equipment configurations required for successful implementation, please refer to the detailed technical guide below.
- Prepare the catholyte by dissolving methyl 4-methoxy-5-(3-morpholinylpropoxy)-2-nitrobenzoate in an acidic aqueous solution (phosphoric, sulfuric, or hydrochloric acid) within a diaphragm electrolytic cell.
- Set the cathode working electrode voltage between 1.00V and 2.50V relative to a reference electrode, maintaining a current density of 25.0mA/cm² to 250.0mA/cm².
- Conduct electrolysis at temperatures between 25°C and 80°C until TLC analysis indicates the disappearance of the nitro starting material, then isolate the amino product via extraction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this electroreduction technology represents a strategic opportunity to optimize costs and mitigate supply risks associated with the production of high-purity pharmaceutical intermediates. The elimination of expensive palladium catalysts removes a significant variable cost component and eradicates the need for complex downstream purification steps designed to remove trace heavy metals, which are often costly and time-consuming. Additionally, the shift away from high-pressure hydrogenation reduces the capital expenditure required for specialized safety equipment and lowers the insurance premiums associated with handling hazardous gases. The use of water as the primary solvent medium further simplifies the waste management workflow, reducing the environmental compliance burden and facilitating smoother regulatory approvals for new manufacturing sites. These factors collectively contribute to a more resilient and cost-effective supply chain capable of meeting the rigorous demands of the global oncology market.
- Cost Reduction in Manufacturing: The transition to an electrochemical process fundamentally alters the cost structure by removing the dependency on precious metal catalysts and toxic chemical reductants. Without the need for palladium on carbon or sodium dithionite, the raw material costs are significantly stabilized, and the expenses related to catalyst recovery and heavy metal scavenging are entirely eliminated. Moreover, the simplified workup procedure, which avoids complex distillation of organic solvents due to the aqueous nature of the reaction, leads to substantial savings in energy consumption and solvent procurement. This streamlined approach allows manufacturers to offer more competitive pricing for the gefitinib intermediate while maintaining healthy margins, providing a distinct economic advantage in a price-sensitive generic pharmaceutical market.
- Enhanced Supply Chain Reliability: Relying on electricity as the primary reagent decouples the production process from the volatility of the chemical reagent market, ensuring a more stable and predictable supply of critical inputs. The mild operating conditions, characterized by atmospheric pressure and moderate temperatures, reduce the likelihood of unplanned shutdowns due to equipment failure or safety incidents, thereby guaranteeing consistent delivery schedules for downstream API manufacturers. Furthermore, the scalability of electrolytic cells allows for flexible production capacity adjustments, enabling suppliers to rapidly respond to fluctuations in market demand without the long lead times associated with constructing high-pressure hydrogenation facilities. This agility is crucial for maintaining continuity of supply in the face of global disruptions.
- Scalability and Environmental Compliance: The inherent green chemistry attributes of this electroreduction method align perfectly with the sustainability goals of modern pharmaceutical companies, facilitating easier regulatory approval and community acceptance of manufacturing sites. The absence of organic solvents and the generation of minimal inorganic waste simplify the effluent treatment process, reducing the environmental footprint and lowering the costs associated with waste disposal and environmental monitoring. As regulatory bodies worldwide tighten restrictions on solvent emissions and heavy metal residues, this technology future-proofs the supply chain against evolving compliance requirements. The ability to scale this process from laboratory benchtop to multi-ton industrial production using standard electrolytic equipment ensures that the technology is ready for immediate commercial deployment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electroreduction synthesis of gefitinib intermediates, drawing directly from the experimental data and process descriptions provided in the patent literature. These insights are intended to clarify the operational feasibility and quality advantages of this novel method for stakeholders evaluating potential technology transfers or supplier partnerships. Understanding these nuances is essential for making informed decisions about integrating this green chemistry approach into existing production workflows.
Q: How does electroreduction improve the purity of gefitinib intermediates compared to catalytic hydrogenation?
A: Electroreduction eliminates the use of palladium catalysts, thereby preventing the formation of difficult-to-remove metal complexes and ensuring the final product meets stringent heavy metal specifications required for oncology APIs.
Q: What are the safety advantages of using electrochemical methods over traditional chemical reduction?
A: This method operates at atmospheric pressure and moderate temperatures (25°C–80°C), removing the explosion risks associated with high-pressure hydrogenation and the environmental hazards of inorganic reducing agents like sodium dithionite.
Q: Can this electroreduction process be scaled for industrial production?
A: Yes, the process utilizes simple acidic aqueous electrolytes and standard diaphragm cells, allowing for straightforward scale-up from laboratory to multi-ton commercial production without complex solvent recovery systems.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Methyl 2-amino-4-methoxy-5-(3-morpholinopropoxy)benzoate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in delivering high-value pharmaceutical intermediates with unmatched purity and consistency. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in an industrial setting. We operate stringent purity specifications and utilize rigorous QC labs to verify that every batch of gefitinib intermediate meets the exacting standards required for oncology drug development, free from the heavy metal contaminants often associated with traditional catalytic methods. Our commitment to technological excellence allows us to serve as a strategic partner for global pharma companies seeking to optimize their supply chains through innovative green chemistry solutions.
We invite you to engage with our technical procurement team to discuss how this electroreduction technology can be tailored to your specific production needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this cleaner, more efficient process. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing you to validate the quality and scalability of our manufacturing capabilities before committing to long-term supply agreements. Let us help you engineer a more sustainable and profitable future for your API production.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
