Revolutionizing Gefitinib Intermediate Production: A Deep Dive into Novel Quinazoline Synthesis and Commercial Scalability
The pharmaceutical landscape for oncology treatments continues to evolve rapidly, with tyrosine kinase inhibitors remaining a cornerstone of therapy for non-small cell lung cancer. At the heart of this therapeutic class lies Gefitinib, a potent EGFR inhibitor whose demand necessitates robust, scalable, and environmentally sustainable manufacturing processes for its key intermediates. Patent CN102153519A introduces a transformative preparation method for quinazoline derivatives that addresses critical bottlenecks in the existing supply chain. Unlike legacy synthetic routes that depend on aggressive halogenation, this innovation leverages advanced peptide coupling technology to achieve high-purity outcomes under remarkably mild conditions. This technical breakthrough not only enhances the chemical integrity of the final active pharmaceutical ingredient but also aligns with modern green chemistry principles essential for long-term regulatory compliance. For global procurement leaders, understanding the mechanistic superiority of this route is vital for securing a reliable quinazoline derivative supplier capable of meeting stringent quality standards. 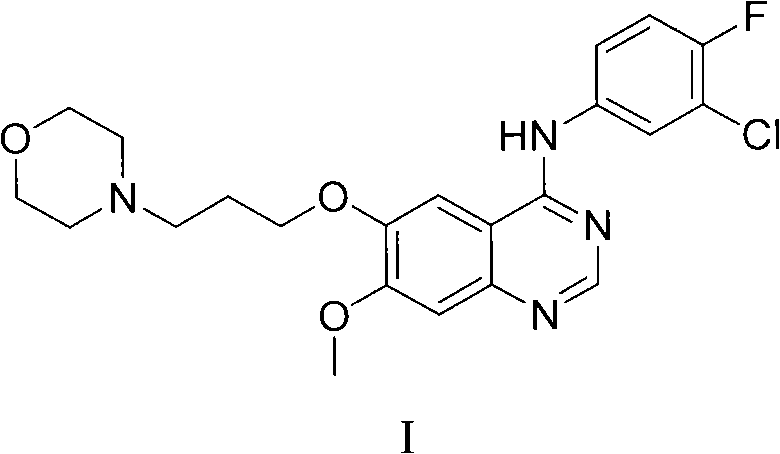
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Gefitinib intermediates has been plagued by reliance on classical chlorination strategies that pose significant operational and environmental risks. Conventional methodologies, as documented in various prior art references, typically involve the conversion of quinazolinone precursors into chloro-quinazolines using reagents such as phosphorus pentachloride, phosphorus oxychloride, or thionyl chloride. These chlorinating agents are inherently corrosive, hazardous to handle, and generate substantial quantities of acidic waste streams that require complex and costly neutralization procedures. Furthermore, the reaction conditions required for these transformations are often severe, involving high temperatures that can promote thermal degradation and the formation of difficult-to-separate byproducts. The subsequent nucleophilic substitution step with 3-chloro-4-fluoroaniline frequently suffers from incomplete conversion and prolonged reaction times, leading to inconsistent yields and compromised purity profiles. 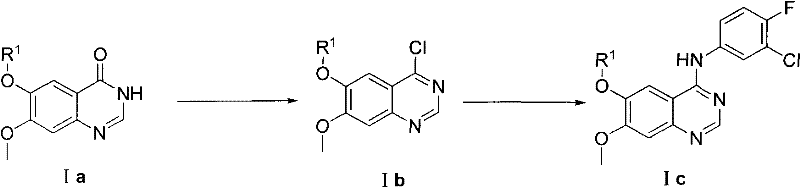
The Novel Approach
In stark contrast to the hazardous legacy pathways, the method disclosed in CN102153519A represents a paradigm shift towards constructive bond formation rather than destructive substitution. This novel approach bypasses the chlorination step entirely by directly coupling a quinazolinone derivative with 3-chloro-4-fluoroaniline using a peptide condensing agent. By activating the carbonyl group of the quinazolinone ring through phosphonium or uronium salt intermediates, the reaction proceeds smoothly under neutral to mildly basic conditions. This eliminates the need for corrosive chlorinating reagents and the associated safety infrastructure, thereby simplifying the reactor setup and reducing capital expenditure on corrosion-resistant equipment. The mildness of the reaction environment preserves the structural integrity of sensitive functional groups, resulting in a cleaner crude product profile that requires less intensive downstream purification. This strategic pivot from substitution to coupling chemistry fundamentally alters the economic and safety calculus of manufacturing high-purity API intermediates.
Mechanistic Insights into Peptide Coupling Catalysis
The core of this technological advancement lies in the precise activation of the amide carbonyl within the quinazolinone scaffold using specialized condensing agents such as benzotriazole-1-base oxygen base three (dimethylamino) phosphorus hexafluorophosphate (BOP). Mechanistically, the condensing agent reacts with the carbonyl oxygen to form a highly reactive O-acylisourea or phosphonium intermediate, which is significantly more electrophilic than the parent amide. This activation lowers the energy barrier for the nucleophilic attack by the amino group of 3-chloro-4-fluoroaniline, facilitating the formation of the new carbon-nitrogen bond at temperatures as low as room temperature to 60°C. The presence of a non-nucleophilic base, such as 1,8-diazabicyclo[5.4.0]-7-undecene (DBU) or triethylamine, serves to scavenge the protons released during the coupling process, driving the equilibrium towards product formation without promoting side reactions. 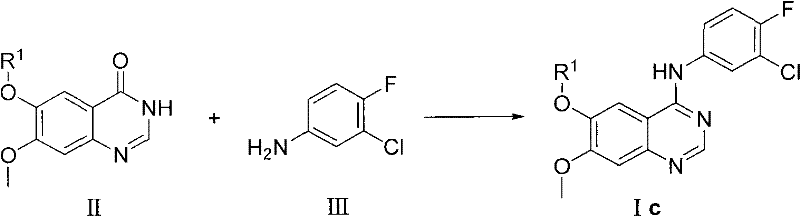
From an impurity control perspective, this mechanism offers distinct advantages over traditional chlorination routes by avoiding the generation of chlorinated organic byproducts and inorganic phosphate sludge. In conventional synthesis, residual phosphorus oxychloride can lead to phosphorylated impurities that are notoriously difficult to remove and can poison downstream catalysts or affect drug stability. The peptide coupling pathway generates urea or phosphine oxide byproducts that are generally more soluble in aqueous washes or organic solvents, allowing for efficient removal during standard workup procedures like ethyl acetate extraction and recrystallization. Furthermore, the specificity of the condensing agent minimizes the risk of over-reaction or polymerization, ensuring a narrow impurity spectrum that simplifies the analytical validation process. This level of chemical precision is critical for R&D directors who must guarantee that the commercial scale-up of complex pharmaceutical intermediates meets the rigorous impurity thresholds set by global health authorities.
How to Synthesize Gefitinib Intermediate Efficiently
The implementation of this synthesis route requires careful attention to reagent stoichiometry and solvent selection to maximize efficiency and yield. The process begins with the suspension of the quinazolinone starting material in an aprotic polar solvent such as acetonitrile, dimethyl sulfoxide, or N,N-dimethylformamide, which ensures adequate solubility of both organic reactants and ionic intermediates. The detailed standardized synthesis steps involve the sequential addition of the condensing agent and base under inert atmosphere to prevent moisture interference, followed by the controlled addition of the aniline nucleophile.
- Prepare the reaction vessel under nitrogen protection and add the quinazolinone derivative (Compound II) along with a suitable aprotic polar solvent such as acetonitrile or DMF.
- Introduce the peptide condensing agent, preferably benzotriazole-1-base oxygen base three (dimethylamino) phosphorus hexafluorophosphate, and the base such as DBU or triethylamine to the mixture.
- Add 3-chloro-4-fluoroaniline (Compound III) to the activated mixture, stir at mild temperatures ranging from room temperature to 60°C, and isolate the product via recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis method translates into tangible strategic benefits that extend beyond simple unit cost calculations. The elimination of hazardous chlorinating agents significantly reduces the regulatory burden associated with the storage, handling, and disposal of toxic chemicals, thereby lowering operational overhead and insurance liabilities. Moreover, the use of readily available peptide coupling reagents and common organic solvents ensures a robust supply chain that is less susceptible to the geopolitical fluctuations often seen with specialized halogenating agents. The mild reaction conditions also imply lower energy consumption for heating and cooling, contributing to a reduced carbon footprint and aligning with corporate sustainability goals.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the removal of expensive and hazardous chlorination reagents, which often require specialized containment and waste treatment facilities. By utilizing standard peptide coupling chemistry, manufacturers can leverage existing infrastructure designed for fine chemical synthesis, avoiding the need for costly retrofitting. The higher selectivity of the reaction reduces the loss of valuable starting materials to byproduct formation, effectively improving the mass balance and overall material efficiency. Additionally, the simplified purification process reduces the consumption of solvents and adsorbents during chromatography or recrystallization, leading to substantial cost savings in downstream processing operations.
- Enhanced Supply Chain Reliability: Securing a reliable quinazoline derivative supplier is contingent upon the availability of raw materials, and this route utilizes commodity chemicals that are widely produced on a global scale. Unlike proprietary catalysts or rare earth metals that may face supply constraints, the reagents required for this peptide coupling method are standard items in the fine chemical inventory. This ubiquity ensures that production schedules are not disrupted by raw material shortages, providing greater predictability for long-term supply contracts. The stability of the reagents also allows for longer shelf life and easier logistics management, reducing the risk of spoilage during transit and storage.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to the plant floor often reveals hidden thermal hazards, but the exothermic profile of this coupling reaction is manageable and well-suited for large batch reactors. The absence of corrosive gases like HCl or SO2 eliminates the need for extensive scrubbing systems, simplifying the engineering requirements for commercial scale-up of complex pharmaceutical intermediates. Furthermore, the reduced generation of hazardous waste streamlines the environmental permitting process and lowers the costs associated with waste disposal vendors. This environmental compatibility ensures continuous operation without the risk of shutdowns due to regulatory non-compliance or community opposition regarding emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this quinazoline synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation to provide clarity for potential partners.
Q: How does this new method improve upon traditional chlorination routes for Gefitinib intermediates?
A: Traditional methods rely on harsh chlorinating agents like phosphorus oxychloride which generate significant hazardous waste and require severe reaction conditions. This novel approach utilizes peptide coupling chemistry under mild conditions, drastically reducing environmental pollution and improving overall yield stability.
Q: What are the primary cost drivers eliminated by this synthesis route?
A: The primary cost reductions come from eliminating the need for expensive and hazardous chlorinating reagents, reducing the complexity of waste treatment protocols, and minimizing energy consumption due to lower reaction temperature requirements.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is specifically designed for industrial suitability. It uses commercially available raw materials, operates under safe pressure and temperature conditions, and avoids the formation of difficult-to-remove impurities common in high-temperature chlorination processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gefitinib Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires more than just chemical knowledge; it demands engineering excellence and unwavering commitment to quality. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of quinazoline derivative meets the exacting standards required for oncology drug manufacturing. We understand the critical nature of API intermediates in the global healthcare supply chain and are dedicated to delivering products that facilitate faster time-to-market for life-saving medications.
We invite you to collaborate with us to optimize your sourcing strategy and leverage the technical advantages of this advanced synthesis route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact us to request specific COA data and route feasibility assessments that demonstrate how our manufacturing capabilities can enhance your project's success. By partnering with us, you gain access to a supply chain that is not only cost-effective but also resilient, compliant, and technically superior.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
