Advanced One-Step Rhodium Catalysis for Scalable Coumarin Intermediate Production
Advanced One-Step Rhodium Catalysis for Scalable Coumarin Intermediate Production
The landscape of fine chemical manufacturing is constantly evolving, driven by the need for more sustainable and efficient synthetic routes for high-value scaffolds. A significant breakthrough in this domain is detailed in patent CN107973768B, which discloses a novel method for the one-step synthesis of coumarin compounds. This technology represents a paradigm shift from traditional multi-step processes, utilizing a robust rhodium-catalyzed carbonylation strategy to transform simple N-phenoxyacetamides into complex 3,4-substituted coumarins. For R&D directors and procurement specialists alike, this innovation offers a compelling value proposition: the ability to access diverse coumarin libraries under remarkably mild conditions (45-70°C) and at normal pressure (1 bar). By eliminating the need for harsh oxidants and extreme thermal inputs, this process not only enhances safety profiles but also drastically reduces the energy footprint associated with producing these critical pharmaceutical and agrochemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of coumarin derivatives has relied heavily on the Pechmann condensation or variations thereof, which typically involve the reaction of salicylaldehydes or phenols with beta-keto esters or acid anhydrides. While these methods are well-established, they suffer from significant inherent drawbacks that hinder modern manufacturing efficiency. The most pressing issue is the requirement for harsh reaction conditions, often necessitating strong acidic catalysts and elevated temperatures that can degrade sensitive functional groups. Furthermore, these traditional routes frequently struggle with regioselectivity, particularly when attempting to introduce specific substituents at the 3- and 4-positions of the coumarin core. This lack of control leads to complex mixtures of isomers, necessitating costly and time-consuming purification steps that erode overall process yield. Additionally, many oxidative coupling methods require stoichiometric amounts of external oxidants, generating substantial quantities of hazardous waste and complicating the environmental compliance profile of the manufacturing site.
The Novel Approach
In stark contrast, the methodology outlined in the patent data introduces a streamlined, atom-economical pathway that directly addresses these legacy inefficiencies. By leveraging transition metal catalysis, specifically a rhodium-based system, the process enables the direct coupling of N-phenoxyacetamides with alkynes and carbon monoxide. This approach bypasses the need for pre-functionalized salicylaldehyde derivatives, allowing manufacturers to start from simpler, more abundant phenol precursors. The reaction proceeds with exceptional efficiency, achieving high yields even with sterically demanding substrates. Crucially, the system exhibits remarkable regioselectivity, ensuring that when asymmetric alkynes are employed, the resulting coumarin products possess a defined substitution pattern at the 3- and 4-positions. This precision eliminates the formation of unwanted isomers, thereby simplifying downstream processing and significantly boosting the purity of the final active pharmaceutical ingredient (API) intermediate.
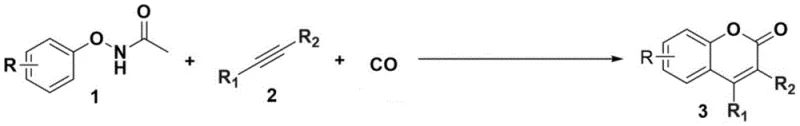
The versatility of this synthetic route is further evidenced by its broad substrate scope, accommodating a wide array of electronic and steric environments on both the phenol and alkyne components. As demonstrated in the experimental data, the protocol successfully tolerates various substituents including methyl, ethyl, and bromo groups on the aromatic ring, as well as different alkyne partners ranging from internal alkynes to terminal variants. This adaptability makes it an invaluable tool for medicinal chemists seeking to rapidly generate analog libraries for structure-activity relationship (SAR) studies. Moreover, the operational simplicity of running the reaction at normal pressure (1 bar) of carbon monoxide removes the need for specialized high-pressure autoclaves, which are often a bottleneck in pilot plant operations. This combination of mild conditions, high selectivity, and operational ease positions this technology as a superior alternative for the cost reduction in fine chemical manufacturing.
Mechanistic Insights into Rh-Catalyzed C-H Activation and Carbonylation
To fully appreciate the technical robustness of this method, one must delve into the underlying catalytic cycle that drives the transformation. The reaction is initiated by the activation of the ortho-C-H bond of the N-phenoxyacetamide substrate by the cationic rhodium(III) species, generated in situ from the dimeric precursor [Cp*RhCl2]2 and a cesium base. This C-H activation step is the cornerstone of the process, forming a stable five-membered rhodacycle intermediate that directs the subsequent chemistry. Once the metallacycle is formed, the alkyne substrate coordinates to the metal center and undergoes migratory insertion into the Rh-C bond. This step is critical for establishing the carbon framework of the coumarin ring and is highly influenced by the steric and electronic properties of the alkyne, which explains the observed regioselectivity. Following alkyne insertion, carbon monoxide inserts into the newly formed Rh-C bond, extending the carbon chain and setting the stage for lactonization.
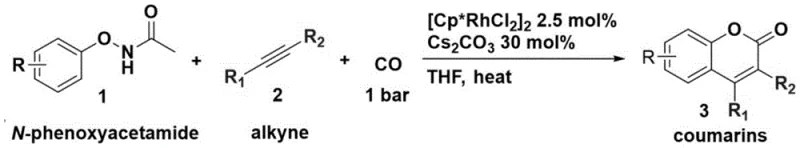
The final stages of the catalytic cycle involve the nucleophilic attack of the amide oxygen onto the acyl-rhodium species, followed by reductive elimination or protonolysis to release the coumarin product and regenerate the active catalyst. The use of a cesium base, such as cesium carbonate or acetate, plays a dual role in this mechanism: it facilitates the initial C-H activation by acting as a proton shuttle and helps to neutralize the acidic byproducts generated during the cycle. This mechanistic pathway is inherently cleaner than oxidative methods because it does not require external oxidants to turn over the catalyst; instead, the redox neutrality is maintained through the coordination and insertion steps. From an impurity control perspective, this mechanism is highly advantageous. The directed nature of the C-H activation ensures that functionalization occurs exclusively at the ortho-position relative to the directing group, preventing random substitution on the aromatic ring. This intrinsic selectivity minimizes the formation of regioisomeric impurities, which are often the most difficult to remove during purification, thereby ensuring a high-purity pharmaceutical intermediate suitable for stringent regulatory standards.
How to Synthesize 3,4-Substituted Coumarins Efficiently
Implementing this advanced synthetic route in a laboratory or pilot plant setting requires careful attention to the specific parameters outlined in the patent to maximize yield and reproducibility. The process is designed to be operationally simple, utilizing common solvents and commercially available reagents, yet it demands precise control over catalyst loading and reaction atmosphere to achieve the reported efficiencies. The following guide summarizes the critical operational steps derived from the proprietary data, serving as a foundational reference for process chemists aiming to replicate or scale this technology. For a comprehensive understanding of the exact stoichiometry and workup procedures, the detailed standardized synthesis steps are provided in the section below.
- Prepare the reaction mixture by combining N-phenoxyacetamide substrate, alkyne, [Cp*RhCl2]2 catalyst (2.5 mol%), and cesium base in THF solvent.
- Introduce carbon monoxide gas to the system at normal pressure (1 bar) and heat the mixture to a temperature range of 45-70°C.
- Maintain the reaction for 15-25 hours to ensure complete conversion, followed by standard workup to isolate the 3,4-substituted coumarin product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this rhodium-catalyzed methodology translates into tangible strategic benefits that extend beyond mere chemical elegance. The primary advantage lies in the significant simplification of the supply chain for raw materials. By utilizing N-phenoxyacetamides derived from simple phenols, manufacturers can bypass the volatile market for specialized salicylaldehyde derivatives, which are often subject to price fluctuations and supply constraints. Furthermore, the ability to run the reaction at normal pressure (1 bar) implies that existing standard reactor infrastructure can be utilized without the need for capital-intensive upgrades to high-pressure equipment. This compatibility with standard hardware drastically reduces the barrier to entry for scaling production, allowing for rapid deployment of manufacturing capacity to meet market demand. The elimination of external oxidants also streamlines the waste management process, reducing the costs associated with hazardous waste disposal and environmental compliance monitoring.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by several key factors that collectively lower the cost of goods sold (COGS). Firstly, the high atom economy of the carbonylation reaction ensures that the majority of the starting material mass is incorporated into the final product, minimizing raw material waste. Secondly, the mild reaction temperatures (45-70°C) result in substantially lower energy consumption compared to traditional high-temperature condensations, leading to direct savings on utility costs. Thirdly, the high regioselectivity reduces the burden on purification teams, as less solvent and chromatography media are required to separate the desired product from isomeric impurities. Finally, the use of a robust catalyst system that functions efficiently at low loadings helps to mitigate the cost impact of the precious metal, especially when coupled with effective recovery protocols.
- Enhanced Supply Chain Reliability: In an era where supply chain resilience is paramount, this method offers a distinct advantage through its reliance on commodity chemicals. Phenols and alkynes are produced on a massive global scale for various industries, ensuring a stable and diversified supply base that is less susceptible to disruption. The simplified synthetic sequence, which achieves the target scaffold in a single step from the amide precursor, shortens the overall lead time for production. This reduction in process steps not only accelerates time-to-market for new drug candidates but also reduces the cumulative yield losses typically associated with multi-step syntheses. Consequently, manufacturers can maintain higher inventory levels of finished intermediates with greater confidence, ensuring continuity of supply for downstream API production.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges regarding heat transfer and safety, but this protocol is inherently designed for scalability. The exothermicity of the reaction is manageable under the described mild conditions, reducing the risk of thermal runaway incidents that can plague large-scale oxidations. From an environmental standpoint, the absence of stoichiometric oxidants and the use of carbon monoxide as a C1 building block align with green chemistry principles by reducing the E-factor (mass of waste per mass of product). The solvent system, typically tetrahydrofuran (THF), is well-understood and can be efficiently recovered and recycled in a closed-loop system. These factors collectively enhance the sustainability profile of the manufacturing site, facilitating easier permitting and fostering stronger relationships with regulatory bodies and community stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this coumarin synthesis technology. These answers are derived directly from the patent specifications and are intended to provide clarity for technical teams evaluating the feasibility of this route for their specific projects. Understanding these nuances is critical for making informed decisions about process adoption and resource allocation.
Q: What are the primary advantages of this Rh-catalyzed method over traditional Pechmann condensation?
A: Unlike traditional methods requiring harsh acids and high temperatures, this protocol operates at mild temperatures (45-70°C) and normal pressure, offering superior regioselectivity for 3,4-substitution without needing external oxidants.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the use of normal pressure carbon monoxide and readily available starting materials like phenols and alkynes significantly simplifies equipment requirements, making it highly scalable for commercial production.
Q: What is the regioselectivity profile for asymmetric alkynes in this reaction?
A: The method demonstrates unique selectivity at the 3- and 4-positions when reacting with asymmetric alkynes, effectively controlling the substitution pattern which is often a challenge in conventional synthesis routes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Coumarin Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in patent CN107973768B for the production of high-value heterocycles. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate such innovative laboratory protocols into robust, commercial-scale manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop to plant floor is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to verify that every batch of coumarin intermediate adheres to the highest quality standards required by the global pharmaceutical industry.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Whether you require custom synthesis of specific 3,4-substituted coumarins or assistance in optimizing an existing route, our technical procurement team is ready to support your needs. Please contact us to request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to reach out today to discuss specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation can drive value for your supply chain.
