Advanced Sulfentrazone Manufacturing: Technical Upgrades and Commercial Scalability Insights
The global demand for high-efficiency herbicides continues to drive innovation in agrochemical intermediate manufacturing, with Sulfentrazone standing out as a critical difluoromethyl triazolinone compound. Recent technological advancements disclosed in patent CN108424395B have introduced a transformative preparation method that fundamentally alters the synthetic landscape for this key active ingredient. By replacing traditional, hazardous reagents with stable methyl sulfonates and employing a specialized halogenated dimethyl sulfonium catalyst, this new approach addresses long-standing challenges regarding safety, yield, and environmental compliance. For R&D directors and procurement specialists, understanding this shift is vital for securing a competitive edge in the supply chain. The structural integrity of the final product is paramount, as shown in the chemical configuration below, which dictates its biological activity and market value.
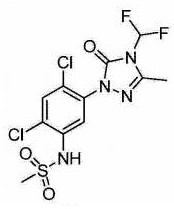
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Sulfentrazone has relied heavily on routes exemplified by US patent US4818275, which utilize methanesulfonyl chloride as the primary sulfonating agent. This conventional pathway is fraught with significant operational hazards and inefficiencies that burden modern manufacturing facilities. The use of methanesulfonyl chloride introduces severe corrosivity risks, requiring specialized equipment and stringent safety protocols to manage the evolution of hydrogen chloride gas during the reaction. Furthermore, to neutralize this acidic byproduct, processes typically demand stoichiometric or excess amounts of organic bases such as triethylamine or pyridine. This neutralization step inevitably generates substantial quantities of salt-containing wastewater, creating a heavy environmental liability and increasing waste treatment costs drastically. Additionally, the atom economy of these traditional methods is poor, often consuming two equivalents of the sulfonylating agent to produce the desired mono-sulfonamide, leading to raw material wastage and yields that stagnate around 66%, which is commercially suboptimal for large-scale production.
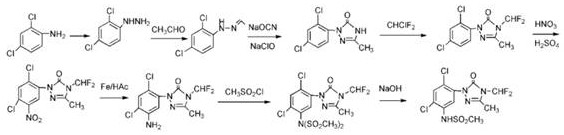
The Novel Approach
In stark contrast, the innovative methodology outlined in CN108424395B circumvents these legacy issues by substituting methanesulfonyl chloride with methyl sulfonate esters, which are inherently more stable and safer to handle. This strategic substitution eliminates the generation of hydrogen chloride gas entirely, thereby removing the necessity for excessive organic bases and the subsequent formation of saline effluent. The core of this breakthrough lies in the application of a halogenated dimethyl sulfonium catalyst, which effectively activates the triazolinone aniline towards nucleophilic attack by the sulfonate ester. This catalytic activation allows the reaction to proceed under much milder conditions, typically between 0°C and 50°C, compared to the harsh reflux or high-pressure conditions required by older ester-amine coupling methods. The result is a streamlined process that not only enhances operator safety but also dramatically improves the overall yield and purity profile, making it a superior choice for cost reduction in herbicide manufacturing and ensuring a more reliable agrochemical intermediate supplier capability.
Mechanistic Insights into Halogenated Dimethyl Sulfonium Catalysis
To fully appreciate the technical superiority of this synthesis, one must delve into the mechanistic role played by the halogenated dimethyl sulfonium catalyst. The catalyst, typically prepared in situ from dimethyl sulfide and a halogen source such as chlorine or bromine, acts as a potent Lewis acid activator. As illustrated in the catalyst preparation reaction below, the interaction between dimethyl sulfide and the halogen creates a highly electrophilic sulfonium species. This species coordinates with the carbonyl oxygen or the nitrogen center of the triazolinone aniline, significantly increasing the nucleophilicity of the amino group. This activation lowers the energy barrier for the subsequent substitution reaction with the methyl sulfonate, allowing the process to occur rapidly at low temperatures without the need for strong external bases. The specificity of this catalytic cycle ensures that side reactions, such as over-sulfonation or degradation of the sensitive triazolinone ring, are minimized, which is critical for maintaining the high-purity sulfentrazone specifications required by regulatory bodies.
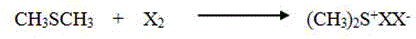
Furthermore, the impurity control mechanism inherent in this catalytic system provides a distinct advantage for quality assurance teams. In traditional base-mediated reactions, the presence of excess amine bases often leads to complex impurity profiles that are difficult to separate during purification. By shifting to a catalytic system that operates without stoichiometric bases, the impurity spectrum is significantly simplified. The reaction primarily produces methanol as a byproduct, which is volatile and easily removed during the workup or recycled for the synthesis of the starting sulfonate ester. This clean reaction profile translates directly to reduced downstream processing requirements, such as fewer recrystallization steps or less intensive chromatographic purification. For commercial scale-up of complex agrochemical intermediates, this simplicity is invaluable, as it reduces batch cycle times and ensures consistent product quality across large production runs, thereby reducing lead time for high-purity herbicides.
How to Synthesize Sulfentrazone Efficiently
Implementing this novel synthesis route requires precise control over reaction parameters to maximize the benefits of the catalytic system. The process begins with the careful preparation of the catalyst, followed by the controlled addition of reagents to manage exotherms and ensure complete conversion. The operational window is relatively broad, accommodating temperatures from 0°C to 50°C, but optimal results are consistently observed in the lower range of 0°C to 20°C. Solvent selection also plays a crucial role, with halogenated aliphatic compounds like dichloromethane proving most effective due to their ability to dissolve reactants while facilitating easy product precipitation upon cooling. The following guide outlines the standardized operational framework derived from the patent examples, serving as a foundational reference for process engineers aiming to adopt this technology.
- Prepare the halogenated dimethyl sulfonium catalyst by reacting dimethyl sulfide with chlorine or bromine in a solvent like dichloromethane at low temperatures (-20°C to 0°C).
- Dissolve triazolinone aniline in a suitable organic solvent such as dichloromethane, cool the mixture to 0-5°C, and add the prepared catalyst (approximately 5% by mass).
- Dropwise add methyl methanesulfonate to the reaction mixture, maintain the temperature between 0°C and 20°C for 3 to 4 hours, then filter and dry the resulting solid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this patented synthesis method represents a strategic opportunity to optimize cost structures and mitigate supply risks. The elimination of corrosive reagents and the reduction in waste treatment requirements translate directly into lower operational expenditures. Moreover, the use of stable, commercially available methyl sulfonates reduces the dependency on hazardous material logistics, simplifying inventory management and storage compliance. The high yield and purity achieved reduce the need for reprocessing, ensuring that production schedules are met with greater predictability. These factors collectively enhance the resilience of the supply chain, making it easier to secure a reliable agrochemical intermediate supplier partnership that can withstand market fluctuations and regulatory pressures.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the drastic simplification of the reaction workflow and the elimination of expensive auxiliary reagents. By avoiding the use of stoichiometric amounts of organic bases like triethylamine and pyridine, manufacturers save significantly on raw material costs. Additionally, the removal of salt-containing wastewater generation reduces the financial burden associated with environmental compliance and waste disposal fees. The high atom economy of the reaction ensures that raw material consumption is minimized, as the methyl sulfonate is utilized efficiently without the need for a twofold excess typically seen in chlorosulfonation routes. These cumulative efficiencies result in substantial cost savings that improve the overall margin profile of the final herbicide product.
- Enhanced Supply Chain Reliability: From a logistics perspective, substituting methanesulfonyl chloride with methyl sulfonate esters significantly de-risks the supply chain. Methanesulfonyl chloride is a hazardous material that requires special handling and transport protocols, often leading to delays and increased shipping costs. Methyl sulfonates, being stable liquids or solids, are easier to source, store, and transport, ensuring a steady flow of materials to the production line. Furthermore, the mild reaction conditions reduce the risk of unplanned shutdowns due to equipment corrosion or safety incidents. This stability allows for more accurate production planning and inventory management, ensuring that customers receive their orders on time without the disruptions common in facilities relying on older, more volatile chemistries.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental challenges, but this novel method is inherently designed for industrial scalability. The absence of corrosive gas evolution means that standard glass-lined or stainless steel reactors can be used without extensive modifications, lowering capital expenditure for scale-up. The ability to recycle the alcohol byproduct back into the synthesis of the methyl sulfonate starting material creates a circular economy within the plant, minimizing fresh raw material intake and waste output. This alignment with green chemistry principles not only satisfies increasingly strict environmental regulations but also enhances the corporate sustainability profile, which is becoming a key criterion for partnerships with major multinational agrochemical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this sulfentrazone synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on performance metrics and operational feasibility. Understanding these details is essential for technical teams evaluating the viability of adopting this new route for commercial production.
Q: Why is methyl sulfonate preferred over methanesulfonyl chloride in this synthesis?
A: Methyl sulfonate is stable and easy to store, whereas methanesulfonyl chloride is highly corrosive and generates hydrogen chloride byproducts. Using methyl sulfonate eliminates the need for excessive organic bases like triethylamine, thereby preventing the formation of large volumes of salt-containing wastewater.
Q: What are the yield improvements associated with the halogenated dimethyl sulfonium catalyst?
A: Traditional methods often achieve yields around 66% due to side reactions and harsh conditions. The novel catalytic system described in patent CN108424395B activates the amino compound effectively, reducing side reactions and achieving yields exceeding 98% with purity levels above 99%.
Q: How does this process impact environmental compliance and waste treatment?
A: The process avoids the generation of hydrochloric acid and organic salt wastes typical of conventional amidation. Additionally, the alcohol byproduct generated can be recovered and recycled to prepare the methyl sulfonate raw material, creating a closed-loop system that significantly reduces environmental burden.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfentrazone Supplier
The technical potential of the CN108424395B patent underscores the importance of partnering with a manufacturer that possesses both the chemical expertise and the infrastructure to execute complex syntheses at scale. NINGBO INNO PHARMCHEM stands at the forefront of this capability, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, ensuring that every batch of Sulfentrazone meets the exacting standards required for global agrochemical registration. We understand that transitioning to a new synthetic route requires confidence in the partner's ability to manage process validation and quality consistency, and our track record demonstrates our commitment to delivering excellence in every kilogram produced.
We invite procurement leaders and technical directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific supply chain needs. By collaborating with our technical procurement team, you can access specific COA data and route feasibility assessments that demonstrate the tangible benefits of switching to this optimized manufacturing process. Whether you are looking to secure long-term supply contracts or explore toll manufacturing opportunities, our team is ready to provide the transparency and technical support necessary to drive your product success forward.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
