Advanced Cytidine Manufacturing: Overcoming Traditional Limitations with Novel Catalytic Systems
Advanced Cytidine Manufacturing: Overcoming Traditional Limitations with Novel Catalytic Systems
The synthesis of cytidine, a critical nucleoside building block for antiviral and antitumor pharmaceuticals such as cytarabine and citicoline, has long been challenged by complex protection-deprotection sequences and harsh reaction conditions. Patent CN108409818B introduces a transformative approach to this chemical challenge, detailing a streamlined two-step methodology that significantly enhances process efficiency and environmental compliance. By utilizing a unique dual-catalyst system comprising trimethylsilyl acetate and tris(pentafluorophenyl)borane, this innovation bypasses the traditionally cumbersome silylation steps required for the cytosine base. For R&D directors and procurement specialists seeking a reliable cytidine supplier, this technology represents a pivotal shift towards more sustainable and cost-effective manufacturing paradigms in the fine chemical industry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of cytidine has relied on methodologies fraught with operational inefficiencies and safety hazards. Early approaches, such as those described by Nishimura, necessitated the use of 1-chlorotriphenylmethyl ribose, a reagent that is not only difficult to source but also yields a complex mixture of alpha and beta anomers, complicating downstream purification. Furthermore, the classic Vorbruggen glycosylation method, while widely used, typically requires the pre-silylation of the nucleobase using hazardous silazanes and the use of tin tetrachloride as a Lewis acid promoter. These conventional routes often suffer from low yields, stringent moisture-free requirements, and the generation of significant heavy metal waste, posing substantial challenges for cost reduction in pharmaceutical intermediates manufacturing and creating bottlenecks in supply chain continuity.
The Novel Approach
In stark contrast to these legacy processes, the method disclosed in CN108409818B employs N4-acylcytosine directly, eliminating the need for a separate silylation step prior to glycosylation. This innovative route leverages the synergistic effect of trimethylsilyl acetate and B(C6F5)3 to activate the sugar donor, tetraacetyl ribose, facilitating a highly regioselective coupling at the N1 position of the pyrimidine ring. By operating under mild thermal conditions ranging from 0°C to 55°C, the process avoids the extreme pressures and temperatures associated with older ammonolysis techniques. This simplification not only reduces the number of unit operations but also drastically lowers the consumption of expensive silylating agents, offering a compelling value proposition for stakeholders focused on the commercial scale-up of complex nucleosides.
Mechanistic Insights into B(C6F5)3-Catalyzed Glycosylation
The core of this technological breakthrough lies in the sophisticated interplay between the Lewis acid catalyst and the silylating agent. Tris(pentafluorophenyl)borane acts as a potent Lewis acid that coordinates with the acetoxy groups of the tetraacetyl ribose, generating a highly reactive oxocarbenium ion intermediate. Simultaneously, trimethylsilyl acetate serves to transiently protect the nucleophilic sites or activate the leaving group, ensuring that the nucleophilic attack by the N4-acylcytosine occurs selectively at the N1 nitrogen rather than the N3 position. This precise control over regioselectivity is critical, as N3-substituted isomers are difficult-to-remove impurities that can compromise the quality of the final API. The absence of water-sensitive tin catalysts further stabilizes the reaction environment, allowing for robust performance even in less-than-ideal laboratory settings.
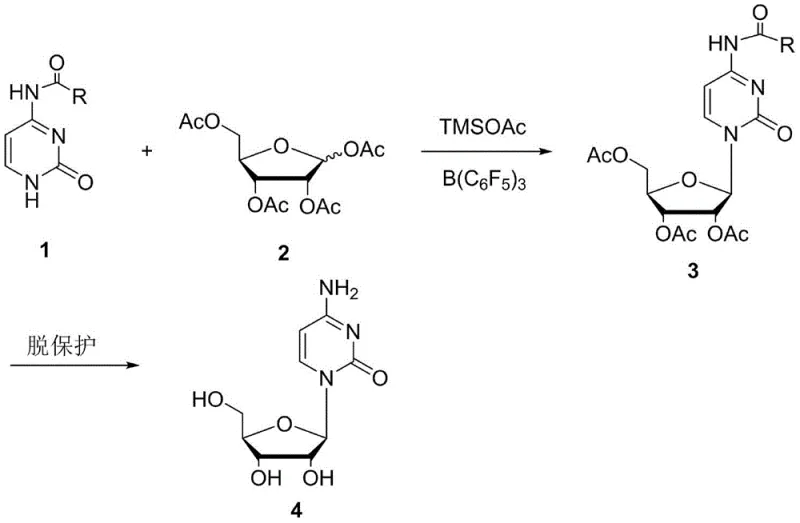
Following the condensation step, the resulting acyl-protected cytidine intermediate undergoes a straightforward deprotection sequence. The patent specifies that this can be achieved under either acidic conditions, using hydrogen chloride in methanol, or basic conditions, using ammonia in methanol. This flexibility is a significant advantage for process chemists, as it allows for the optimization of workup procedures based on available infrastructure and waste treatment capabilities. The mechanism ensures that the acyl protecting group on the N4 position is cleanly removed without affecting the glycosidic bond, yielding cytidine with a reported purity exceeding 99%. Such high purity levels are essential for meeting the rigorous specifications required for high-purity nucleoside intermediates used in oncology and antiviral drug development.
How to Synthesize Cytidine Efficiently
The practical implementation of this synthesis route involves a carefully controlled sequence of mixing, temperature regulation, and workup procedures designed to maximize yield and minimize impurity formation. The process begins with the dissolution of N4-acylcytosine and tetraacetyl ribose in a chlorinated solvent such as dichloroethane, followed by the sequential addition of the catalyst system at low temperatures to manage exothermicity. Detailed standard operating procedures regarding stoichiometry, addition rates, and quenching protocols are essential for reproducibility. For a comprehensive guide on executing this synthesis, please refer to the standardized protocol outlined below.
- Condense N4-acylcytosine and tetraacetyl ribose using trimethylsilyl acetate and B(C6F5)3 catalyst in dichloroethane at 45-55°C.
- Neutralize the reaction mixture with saturated sodium bicarbonate and extract the organic phase to obtain acyl-protected cytidine.
- Perform deprotection using acidic or basic conditions in methanol, followed by crystallization to achieve high-purity cytidine.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this catalytic methodology offers profound benefits that extend beyond mere technical feasibility. By eliminating the requirement for tin tetrachloride, manufacturers can significantly reduce the costs associated with hazardous waste disposal and heavy metal clearance testing, which are often hidden expenses in traditional nucleoside production. Furthermore, the removal of the preliminary silylation step shortens the overall production cycle time, thereby enhancing the responsiveness of the supply chain to fluctuating market demands. This streamlined approach translates into substantial cost savings and improved inventory turnover rates for downstream pharmaceutical clients.
- Cost Reduction in Manufacturing: The elimination of expensive silylating reagents and the reduction in catalyst loading directly lower the bill of materials for each batch. Additionally, the simplified two-step process reduces labor hours and energy consumption associated with multiple isolation and purification stages. This structural efficiency allows for a more competitive pricing model without compromising on the quality of the final active pharmaceutical ingredient precursor.
- Enhanced Supply Chain Reliability: Traditional methods often rely on reagents that are subject to volatile market pricing or supply constraints, such as specialized silyl chlorides. By utilizing more commodity-grade chemicals like trimethylsilyl acetate and avoiding moisture-sensitive intermediates that require special storage, the risk of production delays due to raw material shortages is drastically minimized. This stability ensures a consistent flow of high-quality cytidine to meet the continuous needs of antiviral drug manufacturing lines.
- Scalability and Environmental Compliance: The process operates under mild conditions and generates significantly less toxic waste compared to tin-mediated reactions, aligning perfectly with modern green chemistry initiatives. This environmental compatibility facilitates easier regulatory approval and reduces the burden on wastewater treatment facilities. Consequently, scaling this process from pilot plant to multi-ton commercial production is technically straightforward and environmentally sustainable, securing long-term supply continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel cytidine synthesis route. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the process capabilities and limitations for potential partners.
Q: What are the primary advantages of this cytidine synthesis method over the Vorbruggen method?
A: This method eliminates the need for prior silylation of the cytosine base and avoids the use of toxic tin tetrachloride (SnCl4), resulting in a simpler workflow and reduced environmental waste.
Q: What is the expected purity and yield of the final cytidine product?
A: According to patent data, the process achieves a total yield of approximately 80% with a final product purity exceeding 99% after recrystallization.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes stable reaction conditions (0-55°C) and avoids moisture-sensitive intermediates, making it highly scalable for commercial manufacturing compared to traditional high-pressure or cryogenic methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cytidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the development of life-saving medications. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the B(C6F5)3-catalyzed cytidine synthesis can be seamlessly transferred to industrial manufacturing. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of nucleoside intermediate meets the highest global standards for pharmaceutical applications.
We invite procurement leaders and R&D directors to collaborate with us to leverage this advanced technology for your specific project needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and detailed route feasibility assessments to demonstrate how our optimized manufacturing processes can drive value and efficiency in your supply chain.
