Advanced Solid Acid Catalysis for High-Purity Decitabine Intermediate Manufacturing
Introduction to Next-Generation Decitabine Synthesis
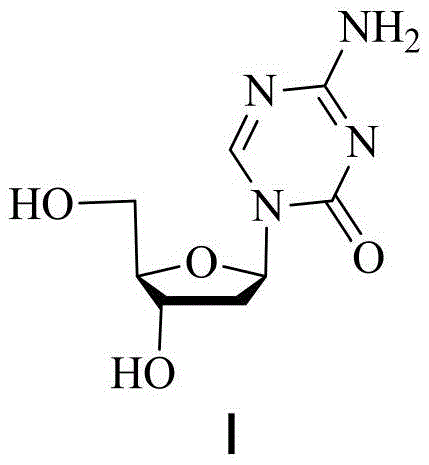
The pharmaceutical landscape for oncology treatments continues to evolve, with Decitabine (Formula I) remaining a cornerstone therapy for myelodysplastic syndromes. As detailed in patent CN111377982B, a significant technological breakthrough has been achieved in the synthesis of its key intermediates. This innovation addresses long-standing challenges in nucleoside analog manufacturing, specifically focusing on the glycosylation step which is critical for establishing the correct stereochemistry. The introduction of a silicon dioxide-supported tin tetrachloride (SnCl4/SiO2) solid acid catalyst represents a paradigm shift from traditional homogeneous catalysis. By transitioning to a heterogeneous system, manufacturers can now access a route that not only enhances the beta-configuration selectivity but also drastically simplifies the downstream purification processes. This development is particularly relevant for reliable pharmaceutical intermediate suppliers aiming to optimize their production lines for high-value antiviral and anticancer agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of decitabine has relied on three primary routes, each fraught with significant industrial drawbacks. The first method, involving the deoxidization of azacitidine, requires multiple protection and deprotection steps that lower overall atom economy. The second method, pioneered by Pliml and Storm, utilizes expensive silver isocyanate for ring closure after glycosylation; this route is plagued by harsh operating conditions and difficult separation of isomers, making it economically unviable for large-scale production. The third and most common industrial method involves direct glycosylation using liquid Lewis acids such as tin tetrachloride (SnCl4) or trimethylsilyl triflate (TMSOTf). While effective, these liquid catalysts create severe post-treatment bottlenecks. The quenching of liquid Lewis acids often leads to stubborn emulsions, necessitating complex extraction procedures or even column chromatography to remove metal residues, which drives up costs and extends lead times. Furthermore, traditional liquid catalysis often struggles to achieve a dominant beta-configuration, typically yielding a beta/alpha ratio of only about 1, which complicates the isolation of the therapeutically active isomer.
The Novel Approach
The methodology disclosed in CN111377982B offers a robust solution by replacing liquid Lewis acids with a solid acid catalyst system. This approach utilizes SnCl4 supported on silica gel, which retains the catalytic activity of tin tetrachloride while introducing the benefits of heterogeneous catalysis. The reaction proceeds smoothly at low temperatures ranging from -10°C to 10°C in common organic solvents like chloroform or dichloromethane. The most striking advantage is the dramatic improvement in stereoselectivity; the process consistently achieves a beta/alpha ratio exceeding 7.7, with some embodiments reaching 8.2. This high selectivity means that the desired beta-anomer is produced in overwhelming abundance, minimizing the formation of the inactive alpha-isomer. Additionally, the solid nature of the catalyst allows for its removal via simple filtration at the end of the reaction, completely bypassing the emulsification issues associated with liquid acid quenching. This results in a cleaner crude product with yields surpassing 80% and purity levels approaching 99%, setting a new standard for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Solid Acid Catalyzed Glycosylation
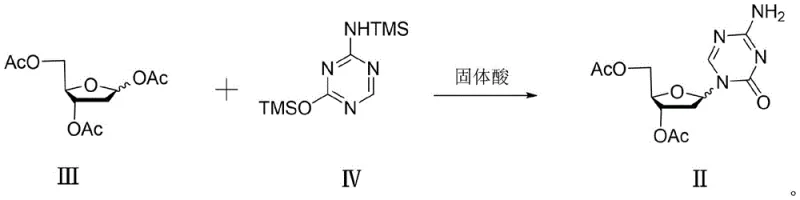
The core of this innovation lies in the interaction between the silylated base and the protected sugar moiety facilitated by the solid acid surface. In this mechanism, the tin tetrachloride species anchored on the silica surface acts as a Lewis acid to activate the anomeric leaving group of the protected ribose (Formula III). Unlike liquid phase reactions where the catalyst is freely diffusing, the supported catalyst provides a unique microenvironment that appears to favor the formation of the beta-glycosidic bond. The silica support likely influences the transition state geometry, stabilizing the oxocarbenium ion intermediate in a conformation that promotes nucleophilic attack from the beta-face by the silylated 5-azacytosine (Formula IV). This steric and electronic modulation is crucial for achieving the high beta/alpha ratios observed. The use of specific particle sizes for the silica support (e.g., 130-80 μm) further optimizes the surface area and accessibility of the active sites, ensuring efficient mass transfer without compromising the selectivity. This mechanistic control is vital for R&D teams focused on high-purity pharmaceutical intermediates, as it reduces the reliance on difficult crystallization or chromatographic separations later in the process.
Impurity control is inherently built into this catalytic system. In traditional liquid acid catalysis, side reactions such as hydrolysis of the protecting groups or degradation of the sensitive triazine ring can occur due to the difficulty in controlling the acidity and water content during the quench phase. With the solid acid method, the reaction is terminated simply by filtering off the catalyst, followed by a mild alkaline wash to neutralize any residual acidity. This gentle workup preserves the integrity of the acetyl protecting groups and the triazine ring, leading to an HPLC purity of the intermediate (α+β) that can reach 99.6%. The reduction in side products directly correlates to a simpler impurity profile, which is a critical parameter for regulatory filings and ensuring the safety of the final API. The ability to tune the catalyst by adjusting the mesh number of the silica gel provides an additional handle for process chemists to fine-tune the reaction for specific impurity profiles, ensuring consistent quality across batches.
How to Synthesize Decitabine Key Intermediate Efficiently
The synthesis protocol outlined in the patent provides a clear pathway for implementing this technology in a pilot or commercial plant. The process begins with the preparation of the necessary precursors: the peracetylated sugar (Formula III) and the bis-silylated base (Formula IV). These are then reacted in the presence of the prepared SnCl4/SiO2 catalyst under controlled low-temperature conditions. The simplicity of the workup—filtration followed by washing—is a major operational advantage. For detailed operational parameters, stoichiometry, and specific temperature ramps, please refer to the standardized synthesis guide below which encapsulates the critical process steps derived from the patent examples.
- Prepare the silylated base (Formula IV) by reacting 5-azacytosine with HMDS and ammonium sulfate under reflux.
- Prepare the solid acid catalyst by supporting tin tetrachloride (SnCl4) on silica gel (SiO2) with specific particle sizes (e.g., 130-80 μm).
- Conduct the glycosylation reaction between the protected sugar (Formula III) and silylated base (Formula IV) at low temperatures (-10 to 10°C) using the solid catalyst, followed by simple filtration and alkaline quenching.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this solid acid catalysis method translates into tangible operational efficiencies and risk mitigation. The elimination of complex post-treatment steps such as column chromatography or extensive aqueous workups to break emulsions significantly reduces the consumption of solvents and consumables. This streamlining of the manufacturing process leads to substantial cost savings by shortening the batch cycle time and reducing labor hours required for purification. Furthermore, the high yield and selectivity mean that less raw material is wasted on producing unwanted isomers or degraded byproducts, optimizing the overall material balance. This efficiency is crucial for maintaining competitive pricing in the global market for commercial scale-up of complex pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The shift from liquid to solid catalysts removes the need for expensive and time-consuming catalyst removal techniques. Traditional methods often require specialized scavengers or multiple extraction cycles to meet heavy metal specifications, which adds significant cost. By simply filtering the solid catalyst, the process avoids these expenses entirely. Additionally, the high beta-selectivity reduces the loss of material during isomer separation, effectively increasing the throughput of the facility without requiring additional capital investment in reactors. The qualitative reduction in waste generation also lowers disposal costs, contributing to a leaner manufacturing budget.
- Enhanced Supply Chain Reliability: The robustness of the solid acid catalyst improves the predictability of the manufacturing schedule. Liquid acid reactions are prone to variability due to moisture sensitivity and quenching difficulties, which can lead to batch failures or extended processing times. The solid acid method offers a more forgiving and reproducible process, ensuring consistent delivery timelines. The use of commercially available and stable reagents like silica gel and tin tetrachloride ensures that the supply chain is not vulnerable to the shortages of exotic or highly specialized catalysts. This reliability is essential for reducing lead time for high-purity pharmaceutical intermediates in a just-in-time manufacturing environment.
- Scalability and Environmental Compliance: Scaling up liquid Lewis acid reactions often presents engineering challenges related to heat transfer during the exothermic quench and the management of large volumes of acidic wastewater. The solid acid approach mitigates these risks by simplifying the quench to a filtration step, generating significantly less acidic waste. This aligns with modern environmental, health, and safety (EHS) standards, making it easier to obtain regulatory approvals for larger production scales. The ability to recycle or safely dispose of the solid catalyst further enhances the sustainability profile of the process, appealing to partners who prioritize green chemistry initiatives in their supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this solid acid catalyzed synthesis. These answers are derived directly from the experimental data and claims within patent CN111377982B, providing a factual basis for evaluating the technology's fit for your specific production needs. Understanding these nuances is key for technical teams assessing the feasibility of technology transfer.
Q: What is the primary advantage of using SnCl4/SiO2 over traditional liquid Lewis acids?
A: The solid acid catalyst eliminates the complex post-treatment associated with liquid acids like SnCl4 or TMSOTf. It prevents emulsification during workup, allowing for simple filtration to remove the catalyst, which significantly reduces processing time and waste.
Q: How does this method improve the stereochemistry of the product?
A: The patented method achieves a beta/alpha ratio greater than 7.7, significantly higher than the approximate 1:1 ratio often seen with traditional tin tetrachloride catalysis. This high selectivity reduces the burden of separating unwanted alpha-isomers.
Q: What are the optimal reaction conditions for this glycosylation?
A: The reaction is best performed at low temperatures between -10°C and 10°C in solvents such as chloroform or dichloromethane. The catalyst particle size (e.g., 130-80 μm) also plays a critical role in maximizing yield and selectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Decitabine Intermediate Supplier
The technological advancements described in CN111377982B highlight the continuous innovation required to produce high-quality oncology intermediates efficiently. At NINGBO INNO PHARMCHEM, we leverage our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring such advanced synthetic routes to life. Our commitment to stringent purity specifications and rigorous QC labs ensures that every batch of decitabine intermediate meets the highest global standards, ready for subsequent conversion into the final API. We understand the critical nature of supply continuity in the pharmaceutical sector and have optimized our operations to deliver consistency and reliability.
We invite you to discuss how this solid acid catalysis technology can be integrated into your supply chain to drive efficiency and quality. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your strategic sourcing goals.
