Advanced Purification Technology for Decitabine Intermediate II to Ensure High Beta-Configuration Yield
Advanced Purification Technology for Decitabine Intermediate II to Ensure High Beta-Configuration Yield
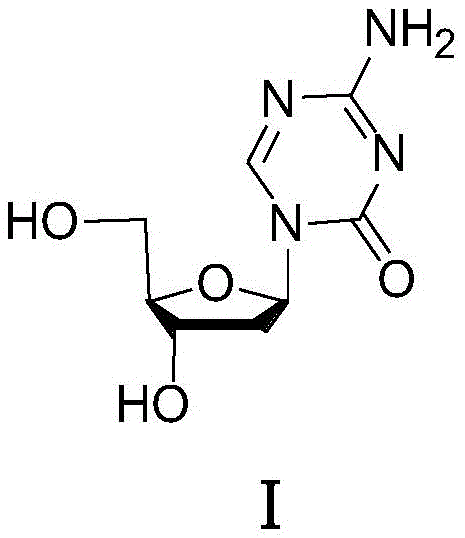
The pharmaceutical landscape for oncology treatments continues to evolve, with nucleoside analogues playing a pivotal role in managing myelodysplastic syndromes (MDS). Central to the production of Decitabine, a potent hypomethylating agent, is the quality of its synthetic intermediates. Patent CN111377986B introduces a transformative purification methodology specifically targeting Decitabine Intermediate II, a critical precursor in the glycosylation step. This innovation addresses a longstanding bottleneck in nucleoside chemistry where trace impurities in the sugar donor drastically skew the stereochemical outcome of the coupling reaction. By implementing a precise solvent-exchange crystallization technique, manufacturers can now achieve intermediate purities exceeding 99%, thereby securing a robust supply chain for high-purity pharmaceutical intermediates. This technical breakthrough not only enhances the beta/alpha anomer ratio but also streamlines the downstream processing required to meet stringent regulatory standards for API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for Decitabine often rely on the direct utilization of crude Intermediate II without rigorous purification, a practice that compromises the stereochemical integrity of the final drug substance. In standard protocols, 2-deoxy-D-ribose is protected and acetylated to form Intermediate II, but this process invariably generates side products such as 3-O-acetyl impurities and residual starting materials. When this crude mixture undergoes glycosylation with silylated 5-azacytosine, these contaminants act as unintended catalysts or steric modifiers that favor the formation of the alpha-anomer. The alpha-configuration is biologically inactive and represents a significant waste of valuable chiral starting materials. Furthermore, the presence of these impurities complicates the final purification of Decitabine, necessitating expensive and time-consuming chromatographic separations that reduce overall process throughput and increase the cost of goods sold (COGS) for the final API.
The Novel Approach
The methodology disclosed in CN111377986B circumvents these issues by introducing a dedicated purification step prior to glycosylation, fundamentally altering the impurity profile of the reaction feedstock. Instead of proceeding with the black oily crude product typically obtained from acetylation, the novel process dissolves the intermediate in a chlorinated solvent and induces precipitation or selective retention of impurities through the controlled addition of an ether-based anti-solvent. This approach effectively scrubs the reaction mixture of the three primary contaminants known to degrade stereoselectivity. By ensuring that only high-purity Intermediate II enters the glycosylation reactor, the process inherently biases the reaction towards the desired beta-configuration. This shift reduces the load on downstream purification units and aligns perfectly with the industry demand for cost reduction in pharmaceutical intermediates manufacturing by minimizing material loss and solvent consumption in later stages.
Mechanistic Insights into Impurity-Induced Stereoselectivity Loss
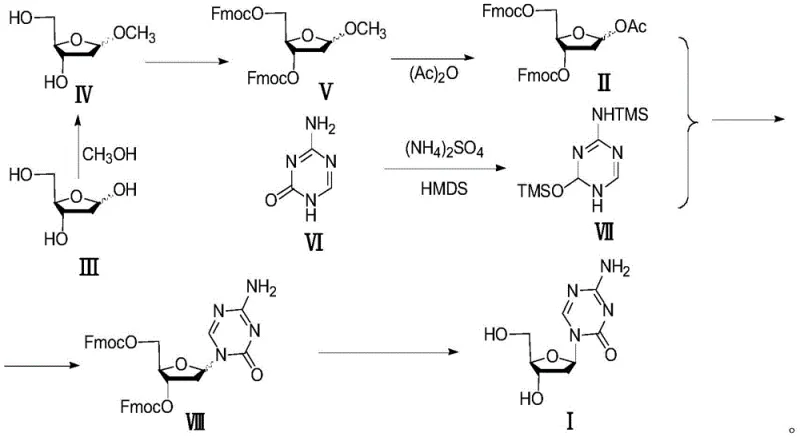
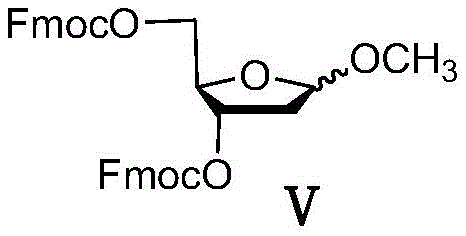
Understanding the mechanistic role of impurities in nucleoside synthesis is crucial for R&D directors aiming to optimize process robustness. The crude Intermediate II typically contains specific structural analogs, notably Impurity 1 (3-O-acetyl derivative), Impurity 2 (partial protection variant), and Impurity 3 (the precursor Intermediate V). These molecules possess different electronic and steric properties compared to the target 3,5-di-Fmoc-protected sugar. During the Lewis acid-catalyzed glycosylation, these impurities can coordinate with the catalyst or the nucleobase, altering the transition state energy landscape. Specifically, the presence of free hydroxyl groups or alternative protecting groups in the impurity mix can stabilize the oxocarbenium ion intermediate in a conformation that favors alpha-attack by the silylated base. This phenomenon explains why unpurified batches consistently yield lower beta/alpha ratios, often hovering near 1:1, which is commercially unacceptable for large-scale production.
The purification mechanism relies on the differential solubility of these impurities in a binary solvent system comprising dichloromethane and methyl tert-butyl ether. At low temperatures ranging from -10°C to 10°C, the target Intermediate II remains soluble or precipitates in a specific crystal habit that excludes the structurally distinct impurities, which remain in the mother liquor or are filtered off depending on the specific embodiment. By removing Impurity 3 (Intermediate V), which has been identified as a potent inducer of alpha-configuration, the process eliminates the primary driver of stereochemical error. This results in a glycosylation product with a significantly improved beta/alpha ratio, shifting from approximately 1.0 in crude processes to 1.2 or higher in purified runs. This enhancement is not merely incremental; it represents a fundamental improvement in the atom economy and chiral efficiency of the entire synthetic sequence.
How to Synthesize Decitabine Intermediate II Efficiently
The implementation of this purification protocol requires precise control over solvent ratios and thermal conditions to maximize recovery while ensuring impurity removal. The process is designed to be scalable, utilizing common industrial solvents that are easily recovered and recycled. Operators must adhere to strict temperature profiles during the anti-solvent addition to prevent oiling out or co-precipitation of contaminants. The following guide outlines the standardized operational parameters derived from the patent examples, serving as a baseline for process engineers looking to integrate this technology into existing production lines. For the complete standard operating procedure and safety data, please refer to the detailed technical documentation provided below.
- Dissolve the crude decitabine intermediate II in a first solvent such as dichloromethane or trichloromethane under reflux conditions to ensure complete solubilization.
- Cool the solution to a temperature range of -10°C to 10°C and slowly dropwise add a second anti-solvent such as methyl tert-butyl ether over a period of 2 to 4 hours.
- Maintain the low temperature and stir continuously for 3 to 5 hours to allow impurities to remain in solution while the purified product precipitates or concentrates in the filtrate upon evaporation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this purification technology translates directly into enhanced operational stability and reduced risk exposure. The ability to source or manufacture a high-purity intermediate mitigates the variability often seen in multi-step nucleoside synthesis, where batch-to-batch fluctuations in impurity profiles can lead to failed quality control tests and delayed shipments. By stabilizing the input quality for the glycosylation step, manufacturers can predict output yields with greater accuracy, facilitating better inventory planning and reducing the need for safety stock buffers. This reliability is paramount for maintaining continuous supply contracts with global pharmaceutical partners who demand consistent quality attributes for their regulatory filings.
- Cost Reduction in Manufacturing: The elimination of critical impurities prior to the coupling reaction significantly lowers the burden on downstream purification processes. In traditional workflows, separating the alpha-anomer from the beta-anomer requires extensive chromatography, which consumes vast quantities of silica gel and organic solvents. By shifting the reaction selectivity towards the beta-form through substrate purification, the volume of waste solvent generated is drastically reduced, and the lifespan of chromatography columns is extended. Furthermore, the high recovery yield of the purification step itself, reported to be over 90%, ensures that valuable protected sugar intermediates are not lost to waste streams, optimizing the overall material balance and reducing the effective cost per kilogram of the final API.
- Enhanced Supply Chain Reliability: The solvents employed in this purification method, specifically dichloromethane and methyl tert-butyl ether, are commodity chemicals with robust global supply chains. Unlike specialized reagents that may face geopolitical or logistical bottlenecks, these solvents are readily available from multiple reliable decitabine intermediate supplier networks. This accessibility ensures that production schedules are not disrupted by raw material shortages. Additionally, the simplicity of the operation—essentially a dissolve-cool-filter sequence—reduces the dependency on highly specialized operator skills, allowing for flexible staffing and easier technology transfer between manufacturing sites, thereby strengthening the resilience of the supply network against local disruptions.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this method offers substantial advantages over alternative purification techniques that might involve complex derivatization or hazardous reagents. The process operates at mild temperatures and does not generate heavy metal waste or toxic byproducts, aligning with modern green chemistry principles and stringent environmental regulations. The ease of solvent recovery in this binary system further minimizes the environmental footprint of the manufacturing process. As production scales from pilot kilograms to commercial tonnage, the thermodynamic consistency of this crystallization-based purification ensures that the process remains stable and controllable, avoiding the non-linear scale-up issues often encountered in complex organic syntheses.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and impact of this purification strategy. These insights are derived directly from the experimental data and mechanistic explanations provided in the patent literature, offering clarity on how this method resolves specific synthesis challenges. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for commercial production.
Q: Why is the purification of Intermediate II critical for Decitabine synthesis?
A: Crude Intermediate II contains specific impurities (such as 3-O-acetyl derivatives and unreacted precursors) that induce the formation of the inactive alpha-anomer during glycosylation. Removing these impurities significantly increases the beta/alpha configuration ratio, which is vital for drug efficacy.
Q: What solvents are used in this purification process?
A: The process utilizes a binary solvent system. A first solvent like dichloromethane or trichloromethane dissolves the crude material, while a second anti-solvent like methyl tert-butyl ether or petroleum ether is added at low temperatures to facilitate the separation of impurities.
Q: Does this method improve the overall yield of the final API?
A: Yes, by increasing the purity of the glycosylation substrate (Intermediate II) to over 99%, the subsequent coupling reaction produces a higher proportion of the desired beta-configured nucleoside, reducing the burden on downstream chromatographic purification and improving overall process mass intensity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Decitabine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the quality of the final therapeutic agent is inextricably linked to the precision of its intermediate synthesis. Our technical team has extensively analyzed the purification protocols described in CN111377986B and integrated similar high-efficiency crystallization technologies into our own manufacturing platforms. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this advanced purification method are realized at an industrial scale. Our facilities are equipped with rigorous QC labs and stringent purity specifications that guarantee every batch of intermediate meets the exacting standards required for oncology drug development, providing our partners with a secure foundation for their regulatory submissions.
We invite global pharmaceutical companies and contract research organizations to collaborate with us on optimizing their Decitabine supply chains. By leveraging our expertise in nucleoside chemistry and process optimization, we can help you navigate the complexities of stereochemical control and impurity management. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific production volumes. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can drive efficiency and quality in your project.
