Advanced Fragment Condensation Strategy for Commercial Goserelin Manufacturing
Advanced Fragment Condensation Strategy for Commercial Goserelin Manufacturing
The pharmaceutical industry continuously seeks robust manufacturing pathways for complex polypeptides like Goserelin, a potent GnRH analog used in treating hormone-sensitive cancers. Patent CN111233980A introduces a sophisticated hybrid synthesis methodology that strategically combines solid-phase peptide synthesis (SPPS) with liquid-phase coupling to overcome traditional bottlenecks. This technical breakthrough addresses the critical challenges of impurity control, racemization, and low yields that have historically plagued the commercial production of decapeptides. By segmenting the ten-amino-acid sequence into manageable fragments—specifically a pentapeptide, a linker, and a tripeptide—the process enables parallel synthesis streams that drastically enhance overall throughput. For R&D directors and procurement specialists, this patent represents a pivotal shift towards more economically viable and chemically pure manufacturing protocols, ensuring a stable supply of this high-value therapeutic agent.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Goserelin has been fraught with significant technical hurdles when relying exclusively on either full liquid-phase or stepwise solid-phase methodologies. Traditional liquid-phase synthesis often involves cumbersome post-treatment operations for each coupling step, leading to extended production cycles and substantial solvent waste, which negatively impacts both cost and environmental compliance. Conversely, while stepwise solid-phase synthesis simplifies purification, it suffers from cumulative yield losses over ten sequential coupling cycles, particularly when dealing with sterically hindered residues. Furthermore, prior art methods frequently struggled with the active guanidyl group on the Arginine residue, which acts as a nucleophile and induces competitive side reactions, resulting in difficult-to-remove deletion peptides and lowering the final assay purity below acceptable pharmaceutical standards.
The Novel Approach
The innovative strategy outlined in the patent data circumvents these issues by adopting a modular fragment condensation approach. Instead of building the entire chain linearly on a single resin or in solution, the process synthesizes three distinct segments: Fragment I (Pyr-His-Trp-Ser-Tyr), Fragment II (D-Ser linker), and Fragment III (Leu-Arg-Pro-hydrazide). This segmentation allows for the optimization of reaction conditions for each specific sequence; for instance, the use of Wang resin for the N-terminal fragment and MBHA resin for the C-terminal fragment ensures optimal loading and cleavage characteristics. Crucially, the final assembly is performed in the liquid phase, which facilitates better mixing and heat transfer for the critical macro-coupling step, thereby achieving superior conversion rates and simplifying the isolation of the crude peptide precursor compared to resin-bound final couplings.
Mechanistic Insights into Hybrid Fragment Condensation
A cornerstone of this synthesis route is the meticulous management of protecting groups to ensure stereochemical integrity and minimize side reactions. The patent highlights the strategic use of the nitro group (NO2) to protect the guanidyl functionality of the Arginine residue within Fragment III. Unlike traditional Pbf or Mtr groups which might require harsh acidic conditions for removal or participate in side reactions during activation, the nitro group offers a stable yet effective mask that prevents the Arginine side chain from interfering with the activated carboxyl species during coupling. This specific chemical modification is vital for maintaining the high purity profile required for injectable APIs, as it effectively suppresses the formation of branched impurities that are notoriously difficult to separate chromatographically.
Furthermore, the cleavage strategy employs a specialized trifluoroethanol (TFE) and dichloromethane (DCM) mixture for releasing the intermediate Fragment II from the 2-Chlorotrityl chloride (2-CTC) resin. This mild acidic condition is specifically chosen to prevent the racemization of the sensitive D-Serine residue, a common pitfall in peptide synthesis where basic or strongly acidic conditions can invert the chiral center. The preservation of chirality is non-negotiable for biological activity, and this mechanistic nuance demonstrates a deep understanding of peptide stability. The subsequent liquid-phase coupling utilizes modern uranium-based reagents like HATU or HBTU in the presence of HOBt and DIEA, which generate highly reactive O-acylisourea intermediates that facilitate rapid amide bond formation with minimal epimerization.
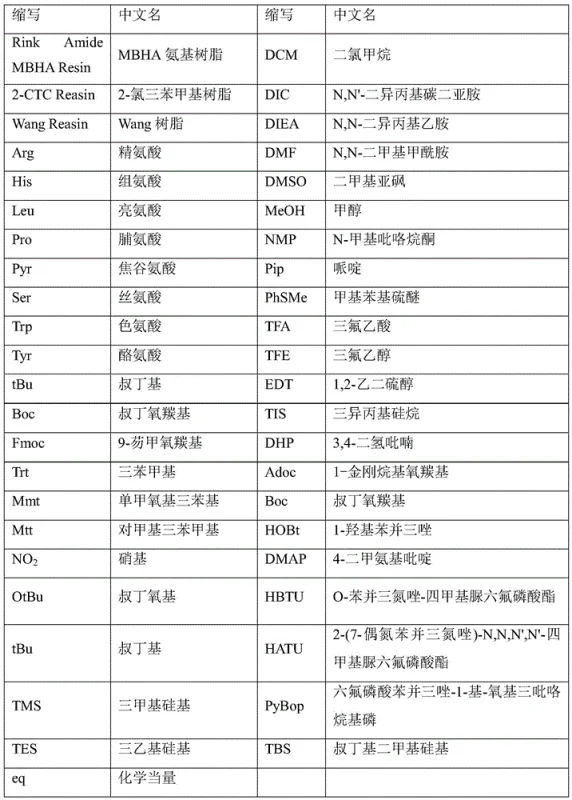
How to Synthesize Goserelin Efficiently
The operational workflow for this synthesis involves precise sequential steps starting with the swelling of specific resins such as Wang and MBHA in dichloromethane, followed by iterative cycles of Fmoc deprotection using piperidine and amino acid coupling. The process requires rigorous monitoring, typically via the Kaiser test, to ensure complete reaction at each step before proceeding to the next amino acid addition. Once the individual fragments are synthesized and cleaved from their respective resins, they undergo purification and characterization before the final liquid-phase condensation. For detailed standard operating procedures and specific stoichiometric ratios required to replicate this high-yield pathway, please refer to the standardized synthesis guide below.
- Synthesize Fragment [5-9] on Wang Resin using Fmoc chemistry and cleave with TFA mixture.
- Synthesize Fragment [1-3] on MBHA Resin utilizing Arg(NO2) protection to prevent side reactions.
- Couple fragments in liquid phase using HATU/HOBt/DIEA system and purify via preparative HPLC.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain and procurement perspective, the adoption of this hybrid fragment synthesis method offers substantial strategic benefits over legacy manufacturing routes. The ability to synthesize multiple fragments simultaneously rather than sequentially creates a parallel processing capability that significantly compresses the overall lead time for batch production. This efficiency gain translates directly into improved responsiveness to market demand fluctuations, allowing suppliers to maintain healthier inventory levels of critical intermediates without tying up excessive working capital in long-cycle work-in-progress goods. Moreover, the simplified purification profile resulting from the Arg(NO2) protection strategy reduces the burden on downstream processing units, lowering the consumption of expensive chromatography media and solvents.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the reduction in resin consumption due to the shorter solid-phase sequences contribute to a leaner cost structure. By shifting the final, most critical coupling step to the liquid phase, the process avoids the diminishing returns associated with coupling on heavily loaded long-chain resins, thereby maximizing the utility of every gram of starting material. This structural efficiency ensures that the cost of goods sold (COGS) is optimized, providing a competitive pricing advantage in the global marketplace for gonadotropin-releasing hormone analogs.
- Enhanced Supply Chain Reliability: The modularity of the fragment approach mitigates the risk of total batch failure; if one fragment synthesis encounters an issue, it does not necessarily compromise the entire ten-step sequence, allowing for quicker recovery and replacement of the specific module. Additionally, the reliance on commercially available and stable reagents such as Fmoc-protected amino acids and standard coupling agents ensures that raw material sourcing remains robust and unaffected by niche supply constraints. This resilience is critical for maintaining uninterrupted supply to pharmaceutical partners who require strict adherence to delivery schedules for regulatory filings and commercial launches.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, as liquid-phase reactions are generally easier to manage in large-scale reactors compared to the swelling and filtration limitations of industrial-scale solid-phase vessels. The reduction in solvent usage per unit of product, driven by higher yields and fewer purification cycles, aligns with increasingly stringent environmental regulations regarding volatile organic compound (VOC) emissions. This green chemistry aspect not only reduces waste disposal costs but also future-proofs the manufacturing site against tightening ecological compliance standards, ensuring long-term operational viability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Goserelin using this advanced fragment condensation technology. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages and chemical rationale behind the process. Understanding these details is essential for technical evaluators assessing the feasibility of technology transfer or long-term supply agreements.
Q: Why is the Arg(NO2) protection group critical in this synthesis?
A: The nitro group protection on the arginine residue effectively masks the active guanidyl group, preventing competitive side reactions during condensation and significantly improving product purity.
Q: How does the hybrid solid-liquid phase method improve scalability?
A: By synthesizing shorter fragments on solid support and performing the final coupling in liquid phase, the method reduces resin consumption and allows for easier handling of larger reaction volumes compared to full solid-phase synthesis.
Q: What is the advantage of using TFE/DCM for cleavage?
A: Using a trifluoroethanol (TFE) mixed solution for cleavage provides milder conditions than strong bases, effectively minimizing amino acid racemization and preserving the stereochemical integrity of the peptide.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Goserelin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of purity and process robustness in the production of life-saving oncology therapeutics like Goserelin. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated fragment condensation strategies described in recent patents can be seamlessly translated into industrial reality. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the absence of racemization and deletion sequences, guaranteeing that every batch meets the highest international pharmacopeial standards.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the economic efficiencies achievable through this method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project timelines, ensuring a secure and high-quality supply of this essential pharmaceutical intermediate.
