Advanced Solid-Phase Synthesis of Goserelin Using CDI Activation for Commercial Scale-Up
The pharmaceutical landscape for oncology and endocrine disorder treatments relies heavily on the consistent supply of high-purity peptide hormones, with goserelin standing out as a critical LHRH agonist. Patent CN102746383A introduces a transformative solid-phase synthesis methodology that addresses longstanding bottlenecks in constructing the unique Azgly (NH2-NH-COOH) moiety found at the C-terminus of the peptide chain. This innovation shifts the paradigm from traditional active ester couplings to a more robust resin activation strategy using N,N'-carbonyldiimidazole (CDI), thereby enhancing both reaction kinetics and final product quality. The structural complexity of goserelin, characterized by its specific sequence Pyro-Glu-His-Trp-Ser-Tyr-D-Ser(But)-Leu-Arg-Pro-Azgly-NH2, demands precise chemical control to avoid racemization and deletion sequences. 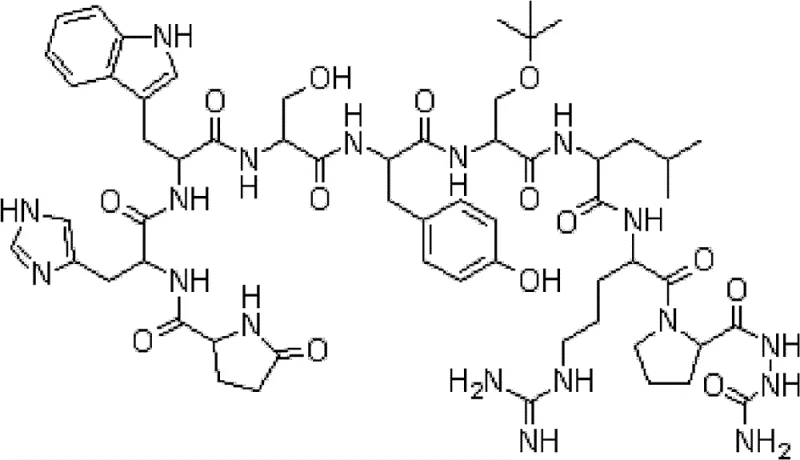 By optimizing the initial resin loading step, this patented process ensures that the subsequent elongation of the nonapeptide chain proceeds with maximal efficiency, offering a viable pathway for reliable goserelin supplier networks to meet global demand.
By optimizing the initial resin loading step, this patented process ensures that the subsequent elongation of the nonapeptide chain proceeds with maximal efficiency, offering a viable pathway for reliable goserelin supplier networks to meet global demand.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of goserelin has been plagued by the inefficiencies associated with preparing the Fmoc-Azgly-OSu active ester, a key building block required for attaching the terminal hydrazine group. As documented in prior art such as US5602231, the reliance on this active ester results in sluggish reaction kinetics, necessitating prolonged reaction times and excessive equivalents of reagents to drive the coupling to completion. Furthermore, the inherent instability of Fmoc-Azgly-OSu often leads to the formation of side products, including residual N,N'-disuccinimidyl carbonate (DSC), which complicates downstream purification and compromises the overall purity profile of the crude peptide. These chemical inefficiencies translate directly into operational burdens, as manufacturers must contend with lower yields, increased solvent waste, and the need for rigorous analytical monitoring to detect trace impurities that could affect biological efficacy. The cumulative effect of these drawbacks is a manufacturing process that is both cost-prohibitive and difficult to scale without sacrificing quality standards.
The Novel Approach
The methodology outlined in CN102746383A circumvents these challenges by introducing a direct activation strategy where the Rink Amide MBHA resin is first reacted with CDI to generate a highly reactive imidazolide intermediate. This intermediate exhibits superior electrophilicity compared to traditional active esters, allowing for rapid and quantitative coupling with Fmoc-NH-NH2 to form the stable Fmoc-NH-NH-CO-NH-Resin scaffold. This strategic shift eliminates the need to isolate and handle the unstable Fmoc-Azgly-OSu species, thereby removing a significant source of variability and impurity from the synthesis workflow. The result is a streamlined process where the critical C-terminal linkage is established quickly and cleanly, setting a solid foundation for the sequential addition of the remaining amino acids. By resolving the connectivity problem of the Azgly unit at the outset, the novel approach effectively boosts the overall throughput and reliability of the synthesis, making it an attractive option for cost reduction in peptide manufacturing.
Mechanistic Insights into CDI-Mediated Resin Activation
The core mechanistic advantage of this synthesis lies in the high reactivity of N,N'-carbonyldiimidazole (CDI) towards nucleophilic attack by the amine groups on the solid support. Upon mixing CDI with the swollen Rink Amide MBHA resin, the carbonyl carbon of the CDI is attacked by the resin-bound amine, displacing one imidazole molecule and forming a reactive urea-like intermediate that is primed for further substitution. 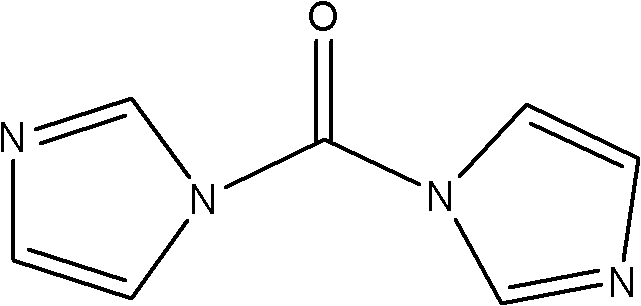 When Fmoc-protected hydrazine (Fmoc-NH-NH2) is introduced, it attacks this activated carbonyl center, displacing the second imidazole group and locking the hydrazine functionality onto the resin via a stable semicarbazide linkage. This two-step activation and coupling sequence occurs under mild conditions, typically at room temperature, which minimizes the risk of thermal degradation or unwanted side reactions that are common in more aggressive coupling protocols. The use of CDI ensures that the activation energy barrier is sufficiently lowered to allow the reaction to proceed to near-completion within a short timeframe, typically ranging from 0.5 to 3 hours depending on the specific batch conditions.
When Fmoc-protected hydrazine (Fmoc-NH-NH2) is introduced, it attacks this activated carbonyl center, displacing the second imidazole group and locking the hydrazine functionality onto the resin via a stable semicarbazide linkage. This two-step activation and coupling sequence occurs under mild conditions, typically at room temperature, which minimizes the risk of thermal degradation or unwanted side reactions that are common in more aggressive coupling protocols. The use of CDI ensures that the activation energy barrier is sufficiently lowered to allow the reaction to proceed to near-completion within a short timeframe, typically ranging from 0.5 to 3 hours depending on the specific batch conditions.
From an impurity control perspective, this mechanism offers distinct advantages by avoiding the introduction of succinimide-based leaving groups that are difficult to remove completely. In conventional methods, residual DSC can react with amino groups on the growing peptide chain, leading to capped sequences that are structurally similar to the target product and challenging to separate via chromatography. The CDI-mediated pathway generates imidazole as the sole byproduct, which is highly soluble in the wash solvents like DMF and dichloromethane, allowing it to be easily removed during the standard washing cycles between coupling steps. This cleanliness in the reaction profile translates to a cleaner crude peptide mixture after cleavage, reducing the load on the preparative C18 columns and extending the lifecycle of the purification media. Consequently, the final goserelin acetate obtained through ion exchange exhibits a superior purity profile, often exceeding 98.5% as confirmed by HPLC analysis, which is critical for meeting stringent regulatory requirements for injectable pharmaceutical products.
How to Synthesize Goserelin Efficiently
The synthesis of goserelin via this patented route involves a systematic sequence of deprotection, coupling, and washing steps that leverage the high reactivity of the CDI-activated resin to ensure robust chain elongation. The process begins with the preparation of the functionalized resin, followed by the iterative addition of protected amino acids including Proline, Arginine, Leucine, Serine, Tyrosine, Tryptophan, Histidine, and Pyroglutamic acid in the correct C-to-N order. Detailed standardized synthetic steps see the guide below.
- Activate Rink Amide MBHA Resin using N,N'-carbonyldiimidazole (CDI) to form a highly reactive intermediate.
- React the activated intermediate with Fmoc-NH-NH2 to establish the critical Fmoc-NH-NH-CO-NH-Resin linkage.
- Perform sequential amino acid condensation, followed by low-concentration TFA cleavage and C18 column purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this CDI-based synthesis route presents a compelling value proposition centered around operational efficiency and resource optimization. By eliminating the requirement for pre-synthesized and potentially unstable active esters, the process simplifies the raw material inventory, reducing the need for specialized cold storage and minimizing the risk of reagent spoilage before use. The accelerated reaction kinetics mean that reactor occupancy times are significantly reduced, allowing for higher batch turnover rates within existing facility footprints without the need for capital-intensive equipment upgrades. Furthermore, the improved yield and purity profiles reduce the volume of crude material that must be processed during the purification stage, leading to substantial savings in chromatography resins and solvent consumption. These factors combine to create a more resilient and cost-effective supply chain capable of responding swiftly to market fluctuations in demand for this critical oncology therapeutic.
- Cost Reduction in Manufacturing: The elimination of the Fmoc-Azgly-OSu synthesis step removes a costly and low-yielding sub-process from the overall workflow, directly lowering the bill of materials for each production batch. Since CDI is a commodity reagent with stable pricing and high availability, substituting it for custom-synthesized active esters drives down the variable costs associated with raw material procurement. Additionally, the reduction in reaction times and solvent usage per kilogram of product contributes to lower utility and waste disposal expenses, enhancing the overall economic viability of the manufacturing process. These cumulative efficiencies allow for a more competitive pricing structure while maintaining healthy margins, which is essential for long-term contracts with generic drug manufacturers.
- Enhanced Supply Chain Reliability: Relying on widely available reagents like CDI and standard Fmoc-amino acids mitigates the risk of supply disruptions that can occur with niche or custom-synthesized intermediates. The robustness of the chemistry ensures consistent batch-to-batch performance, reducing the likelihood of failed runs that could delay shipment schedules and impact customer commitments. Moreover, the simplified process flow reduces the dependency on highly specialized operator skills, making it easier to train personnel and maintain continuous production lines even during periods of workforce fluctuation. This stability is crucial for ensuring the uninterrupted availability of goserelin for patients who rely on it for chronic hormone-sensitive conditions.
- Scalability and Environmental Compliance: The solid-phase nature of this synthesis is inherently scalable, allowing for seamless transition from pilot-scale validation to multi-kilogram commercial production using standard peptide synthesizers. The use of low-concentration trifluoroacetic acid for cleavage minimizes the generation of hazardous acidic waste, aligning with increasingly strict environmental regulations and corporate sustainability goals. Efficient solvent recovery systems can be easily integrated into the workflow due to the standardized solvent palette (DMF, DCM, Methanol), further reducing the environmental footprint of the operation. This alignment with green chemistry principles not only ensures regulatory compliance but also enhances the brand reputation of the manufacturer as a responsible partner in the pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis technology, drawing directly from the experimental data and benefits outlined in the patent documentation. These insights are designed to clarify the operational advantages and quality assurances provided by the CDI activation method for stakeholders evaluating potential manufacturing partners. Understanding these details is key to making informed decisions about sourcing strategies and technology transfer initiatives.
Q: How does the CDI activation method improve upon traditional Fmoc-Azgly-OSu synthesis?
A: The CDI activation method eliminates the need for synthesizing unstable Fmoc-Azgly-OSu active esters, which often suffer from poor reactivity and residual DSC impurities. By reacting CDI directly with the resin, the process achieves faster coupling rates and significantly higher purity in the final goserelin product.
Q: What are the primary purity specifications achievable with this synthesis route?
A: Utilizing the optimized cleavage conditions with low-concentration trifluoroacetic acid and subsequent C18 column chromatography, this method consistently delivers goserelin acetate with purity levels exceeding 95%, often reaching up to 98.5% as verified by HPLC analysis.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the protocol utilizes standard solid-phase peptide synthesis equipment and common reagents like CDI and DMF, making it highly scalable. The simplified workflow reduces reaction times and solvent consumption, facilitating efficient transition from laboratory benchtop to multi-kilogram manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Goserelin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthesis technologies like the CDI-mediated route to ensure the highest standards of quality and efficiency in peptide manufacturing. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a GMP-compliant environment. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art HPLC and mass spectrometry instruments to verify that every batch of goserelin meets or exceeds pharmacopeial standards. Our commitment to technical excellence allows us to navigate the complexities of peptide synthesis, delivering a product that is both chemically pure and biologically potent for your downstream formulation needs.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can be tailored to your specific volume requirements and cost targets. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this more efficient manufacturing protocol. We encourage potential partners to contact us directly to obtain specific COA data from recent batches and to receive comprehensive route feasibility assessments that demonstrate our capability to be your trusted long-term supplier for high-value peptide intermediates.
