Advanced Solid-Phase Synthesis of Semaglutide: Technical Breakthroughs for Commercial Scale-Up
Advanced Solid-Phase Synthesis of Semaglutide: Technical Breakthroughs for Commercial Scale-Up
The pharmaceutical landscape for Type 2 diabetes treatment has been revolutionized by GLP-1 analogs, with Semaglutide standing out as a premier therapeutic agent due to its superior efficacy and half-life profile. Patent CN109180801B introduces a transformative solid-phase chemical synthesis method designed specifically to address the longstanding challenges associated with producing this complex peptide. By strategically employing a specialized lysine monomer, Fmoc-Lys(N-epsilon-ADO-ADO-(gamma-Glu(N-alpha-X)-OtBu)-OH, wherein the glutamic acid alpha-amino protecting group X can be Alloc, Mmt, Mtt, Dde, or ivDde, the invention fundamentally alters the impurity profile of the crude product. This technical advancement is not merely an incremental improvement but a significant leap forward that allows manufacturers to bypass the generation of stubborn impurity peptides that typically plague traditional synthesis routes. The result is a substantial elevation in the purity of crude peptides, which directly translates to streamlined downstream processing and a marked reduction in overall production costs for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Semaglutide has been fraught with complexities arising from the need to introduce a long-acting fatty acid modification at the Lys26 position while maintaining the integrity of the 31-amino acid backbone. Conventional gene recombination technologies often require subsequent chemical conjugation steps that leave multiple active sites unprotected, leading to a proliferation of side reactions and significant material loss. Furthermore, traditional solid-phase peptide synthesis (SPPS) methods that rely on sequential coupling of single amino acids frequently suffer from the accumulation of deletion sequences, particularly the troublesome +/- Gly impurities and 1-Ala variants. These impurities possess physicochemical properties strikingly similar to the target molecule, rendering them exceptionally difficult to remove during the purification phase. Consequently, manufacturers face exorbitant costs related to extensive chromatographic separation, low overall yields, and the inability to consistently meet the stringent purity specifications required for regulatory approval in the competitive API market.
The Novel Approach
The methodology disclosed in CN109180801B dismantles these barriers through a sophisticated orchestration of monomer selection and coupling strategy. By introducing four specific dipeptides, including Fmoc-Glu(OtBu)-Gly-OH and Fmoc-Arg(Pbf)-Gly-OH, directly into the solid-phase synthesis cycle, the process effectively eliminates the risk of generating +/- Gly process impurities at their source. Moreover, the strategic use of Fmoc-Ala-OH at positions 18 and 19 specifically mitigates the formation of the 1-Ala impurity peptide, a common byproduct that complicates purification. Perhaps most critically, the use of the pre-functionalized lysine monomer allows for the side chain modification to be built into the sequence early, with the protecting group X removed selectively on-resin before fatty acidification. This approach ensures that the fatty acid chain is coupled under controlled conditions, avoiding the chaotic side reactions seen in post-synthetic modification, thereby drastically simplifying the process flow and enhancing the robustness of cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Solid-Phase Peptide Assembly and Side-Chain Modification
The core of this synthetic breakthrough lies in the precise management of orthogonality between the various protecting groups utilized throughout the peptide chain assembly. The process initiates with the loading of Fmoc-Arg(Pbf)-Gly-OH onto a Wang or CTC resin, establishing a stable anchor for the growing chain. As the synthesis proceeds towards the critical Lys26 residue, the incorporation of the bulky Fmoc-Lys(N-epsilon-ADO-ADO-(gamma-Glu(N-alpha-X)-OtBu)-OH monomer requires careful optimization of coupling reagents, such as HATU/HOAT/DIPEA or DIC/HOBT, to ensure complete reaction despite steric hindrance. The choice of the protecting group X is pivotal; for instance, if Alloc is selected, it can be removed under mild palladium-catalyzed conditions using Pd(PPh3)4 and phenylsilane, leaving the rest of the acid-labile protecting groups intact. Alternatively, acid-labile groups like Mmt or Mtt can be cleaved with dilute TFA, while hydrazine-labile groups like Dde or ivDde offer another layer of orthogonality. This mechanistic flexibility allows process chemists to tailor the synthesis to their specific equipment and safety constraints while maintaining high fidelity in the peptide sequence.
Following the selective deprotection of the glutamic acid alpha-amino group on the side chain, the fatty acidification step is executed directly on the solid support. This on-resin acylation with octadecanedioic acid is facilitated by the spatial separation of reactive sites, ensuring that the fatty acid couples exclusively to the intended gamma-glutamic acid nitrogen without attacking other nucleophilic sites on the peptide backbone. The use of the ADO-ADO linker serves a dual purpose: it acts as a hydrophilic spacer that improves the solvation of the growing peptide chain on the resin, reducing aggregation, and it positions the fatty acid tail optimally for biological activity. 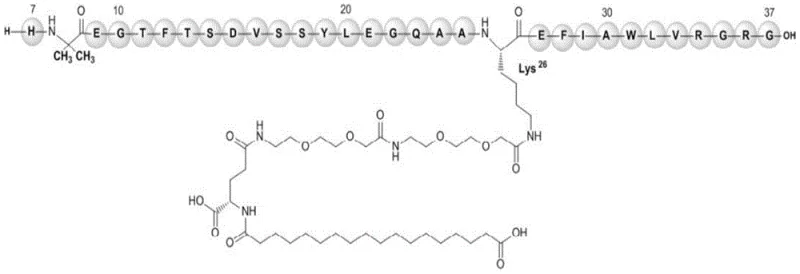 As illustrated in the molecular structure, the precise attachment at Lys26 is crucial for the drug's albumin binding affinity and prolonged half-life. By controlling this modification at the molecular level during synthesis rather than as a post-process step, the method ensures a homogeneous product distribution, significantly lowering the burden on final purification columns and enabling the commercial scale-up of complex pharmaceutical intermediates with consistent quality.
As illustrated in the molecular structure, the precise attachment at Lys26 is crucial for the drug's albumin binding affinity and prolonged half-life. By controlling this modification at the molecular level during synthesis rather than as a post-process step, the method ensures a homogeneous product distribution, significantly lowering the burden on final purification columns and enabling the commercial scale-up of complex pharmaceutical intermediates with consistent quality.
How to Synthesize Semaglutide Efficiently
Implementing this advanced synthesis route requires a disciplined adherence to the optimized coupling cycles and deprotection protocols detailed in the patent documentation. The process is designed to be compatible with standard automated peptide synthesizers, allowing for seamless translation from laboratory benchtop to pilot plant scales. Operators must pay particular attention to the monitoring of reaction endpoints using Kaiser reagents to prevent incomplete couplings, which are the primary source of deletion impurities. The following guide outlines the critical operational phases necessary to achieve the high crude purity and yields reported in the patent examples, serving as a foundational reference for process development teams aiming to adopt this technology.
- Load Fmoc-Arg(Pbf)-Gly-OH onto Wang or CTC resin to initiate the peptide chain assembly.
- Sequentially couple protected amino acids, utilizing dipeptides like Fmoc-Glu(OtBu)-Gly-OH to prevent deletion sequences.
- Incorporate the key monomer Fmoc-Lys(N-epsilon-ADO-ADO-(gamma-Glu(N-alpha-X)-OtBu)-OH at position 26.
- Remove the alpha-amino protecting group X on the side chain glutamic acid using specific deprotection reagents.
- Perform on-resin fatty acidification with octadecanedioic acid followed by cleavage and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the synthesis method described in CN109180801B offers a compelling value proposition centered on risk mitigation and cost efficiency. The traditional bottlenecks in Semaglutide production—namely the low yield of crude peptide and the expensive, time-consuming purification required to remove structurally similar impurities—are directly addressed by this innovation. By elevating the starting purity of the crude material, the demand for preparative HPLC capacity is significantly reduced, which is often the most capital-intensive part of peptide manufacturing. This reduction in downstream processing complexity translates into shorter batch cycle times and lower consumption of expensive chromatography resins and solvents. Furthermore, the use of readily available dipeptide building blocks stabilizes the raw material supply chain, reducing dependency on hard-to-source custom amino acids and insulating production schedules from raw material volatility.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven primarily by the drastic simplification of the purification workflow. In conventional processes, a significant portion of the manufactured batch is lost during the rigorous cleaning steps required to separate the target peptide from +/- Gly and 1-Ala impurities. By preventing the formation of these specific byproducts through the use of dipeptides and optimized monomers, the process preserves a much higher percentage of the theoretical yield. This efficiency gain means that less raw material is needed to produce the same amount of finished API, directly lowering the cost of goods sold (COGS). Additionally, the elimination of transition metal catalysts in certain deprotection variants (such as those using hydrazine or dilute acid instead of palladium) removes the need for costly heavy metal scavenging steps, further streamlining the operational expenditure without compromising product quality.
- Enhanced Supply Chain Reliability: Supply continuity is paramount for high-demand medications like Semaglutide, and this synthesis route enhances reliability by robustifying the manufacturing process. The method's tolerance for different protecting group strategies (Alloc, Mmt, Dde, etc.) provides supply chain managers with flexibility; if a specific reagent faces a shortage, the process can be adapted to use an alternative orthogonal protecting group without redesigning the entire synthetic route. Moreover, the improved crude purity reduces the likelihood of batch failures during quality control testing, ensuring a more predictable output of saleable product. This stability allows for more accurate forecasting and inventory planning, reducing the need for excessive safety stock and freeing up working capital for other strategic investments in the supply chain network.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, the process aligns well with modern green chemistry principles and large-scale production requirements. The reduction in solvent usage associated with fewer purification cycles directly lowers the facility's environmental footprint and waste disposal costs. The solid-phase nature of the synthesis is inherently scalable, as demonstrated by the successful preparation of multi-gram quantities in the patent examples using standard resin loading capacities. The ability to scale from 100 kgs to 100 MT annual commercial production is facilitated by the linear nature of the coupling steps and the avoidance of complex solution-phase convergent syntheses that often encounter mixing and heat transfer issues at large volumes. This scalability ensures that manufacturers can rapidly ramp up production to meet surging global demand while maintaining strict adherence to environmental regulations regarding solvent emissions and chemical waste.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Semaglutide synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a clear understanding of the method's capabilities and limitations for potential licensees and manufacturing partners.
Q: How does this patent method improve the purity of crude Semaglutide?
A: The method utilizes specific dipeptides (e.g., Fmoc-Glu-Gly-OH) and a pre-modified Lysine monomer to prevent the formation of difficult-to-remove impurities like +/- Gly sequences and 1-Ala deletions, resulting in crude peptide purities exceeding 70%.
Q: What are the advantages of using the ADO-ADO linker in this synthesis?
A: The ADO-ADO (8-amino-3,6-dioxaoctanoic acid) linker provides optimal spacing and solubility characteristics during solid-phase synthesis, facilitating efficient coupling of the long-chain fatty acid moiety at the Lys26 position.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process simplifies purification steps by improving crude quality and uses standard Fmoc chemistry compatible with existing peptide synthesizers, making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Semaglutide Supplier
The technical sophistication required to execute the synthesis of Semaglutide according to CN109180801B demands a partner with deep expertise in peptide chemistry and process optimization. NINGBO INNO PHARMCHEM stands at the forefront of this field, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the stringent purity specifications required for GLP-1 analogs, supported by rigorous QC labs that ensure every batch meets international pharmacopoeia standards. We understand that the transition from patent theory to commercial reality involves nuanced adjustments in reaction parameters, and our team of seasoned chemists is dedicated to refining these processes to maximize yield and minimize impurity profiles for our clients.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to leverage this advanced synthesis technology for their supply chains. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. We encourage you to contact our technical procurement team today to request specific COA data from our recent pilot runs and to discuss detailed route feasibility assessments. Let us help you secure a stable, cost-effective, and high-quality supply of Semaglutide intermediates that will drive your commercial success in the competitive diabetes care market.
