Advanced Liquid-Phase Convergent Synthesis of Semaglutide Side Chain for Commercial Scale-Up
The pharmaceutical industry is currently witnessing an unprecedented surge in demand for Glucagon-like peptide-1 (GLP-1) receptor agonists, driven largely by the blockbuster success of Semaglutide in treating type 2 diabetes and obesity. As a critical component of this molecule, the lipophilic side chain plays a pivotal role in extending the drug's half-life by promoting albumin binding. Patent CN111253287A introduces a groundbreaking liquid-phase convergent synthesis method for this specific side chain, addressing the longstanding limitations of traditional solid-phase peptide synthesis (SPPS). This technical breakthrough offers a robust pathway for cost reduction in pharmaceutical intermediates manufacturing, shifting the paradigm from resin-bound limitations to scalable solution-phase chemistry. By leveraging orthogonal protection strategies and one-pot reaction sequences, this method ensures high purity and yield, making it an ideal candidate for industrial adoption by any reliable pharmaceutical intermediate supplier aiming to secure the global supply chain.
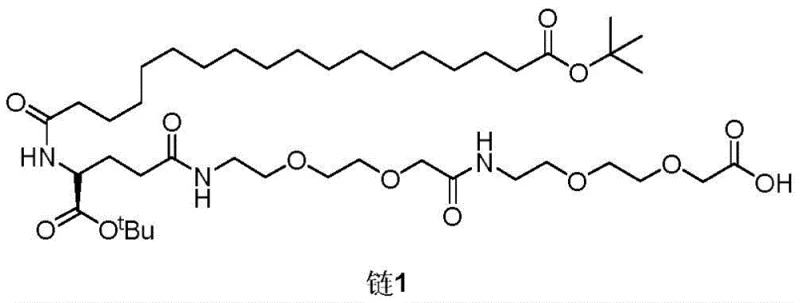
The structural complexity of the Semaglutide side chain, composed of two molecules of 8-amino-3,6-dioxa-octanoic acid, one molecule of glutamic acid, and one molecule of octadecanedioic acid mono-tert-butyl ester, presents significant synthetic challenges. The presence of multiple amide bonds, ether linkages, and chiral centers requires precise control over reaction conditions to prevent racemization and ensure regioselectivity. The patent outlines a meticulous route where the final product can be directly coupled to the Lys residue of the Semaglutide main chain, highlighting its immediate applicability in API production. For procurement managers and supply chain heads, understanding this molecular architecture is crucial, as it dictates the choice of raw materials and the complexity of the purification processes required to meet stringent regulatory standards for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of complex peptide side chains has relied heavily on solid-phase peptide synthesis (SPPS), a method that, while effective for small batches, suffers from poor atom economy and significant scalability issues. In SPPS, the growing peptide chain is anchored to an insoluble resin, requiring large excesses of reagents to drive reactions to completion due to heterogeneous kinetics. This results in substantial waste generation, particularly of hazardous organic solvents used for washing the resin between coupling and deprotection steps. Furthermore, the final cleavage from the resin often requires harsh acidic conditions that can degrade sensitive functional groups or lead to difficult-to-remove impurities. For large-scale commercial scale-up of complex polymer additives or peptide intermediates, the cost of the resin itself and the inability to easily monitor reaction progress in real-time become major bottlenecks, limiting the ability to produce metric-ton quantities efficiently.
The Novel Approach
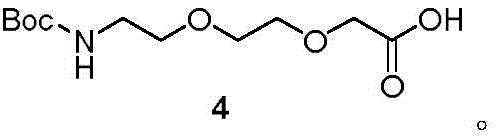
In stark contrast, the method described in Patent CN111253287A employs a liquid-phase convergent synthesis strategy that fundamentally alters the economic and operational landscape of production. By utilizing solution-phase chemistry, the process allows for homogeneous reaction conditions, which significantly improves reaction kinetics and reduces the need for excessive reagent equivalents. A key innovation is the "one-pot" synthesis of key intermediates, such as Compound 4, where protection, nucleophilic substitution, and hydrolysis occur in a streamlined sequence without isolating unstable intermediates. This approach not only minimizes solvent usage and waste disposal costs but also drastically shortens the overall production cycle time. The convergent nature of the synthesis, where separate fragments are assembled late in the sequence, ensures that the longest linear step count is minimized, thereby maximizing the overall yield and facilitating the commercial scale-up of complex pharmaceutical intermediates with greater predictability and control.
Mechanistic Insights into Liquid-Phase Convergent Assembly
The core of this synthesis lies in the strategic manipulation of protecting groups to achieve orthogonality, allowing specific functional groups to be modified without disturbing others. The process begins with the protection of the amino group of 2-(2-aminoethoxy)ethanol using groups like Boc, Fmoc, or Trt, followed by nucleophilic substitution with ethyl bromoacetate. A critical mechanistic advantage is observed in the subsequent ester hydrolysis step, which is performed in the same pot using inorganic bases such as sodium hydroxide or lithium hydroxide. This one-pot transformation eliminates the need for intermediate isolation, reducing material loss and exposure to potential contaminants. The selection of solvents, such as a tetrahydrofuran and methanol mixture (1:1 v:v), is optimized to maintain solubility of both organic substrates and inorganic bases, ensuring efficient mass transfer and reaction homogeneity throughout the process.
As the synthesis progresses to the assembly of the full side chain, the mechanism relies on precise amide bond formation using carbodiimide coupling agents like EDCI in the presence of additives like HOBT or DMAP. These additives suppress racemization of the chiral glutamic acid center, a critical quality attribute for the final API. The removal of protecting groups is equally sophisticated; for instance, benzyl (Bn) groups are removed via catalytic hydrogenation using palladium on carbon, a clean method that produces toluene as the only byproduct, whereas tert-butyl groups are cleaved under mild acidic conditions. This orthogonal deprotection strategy ensures that the final deprotection step to yield Target Product 1 can be performed selectively on the terminal carboxyl group without affecting the internal amide bonds or the fatty acid chain, guaranteeing the structural integrity required for biological activity.
How to Synthesize Semaglutide Side Chain Efficiently
The synthesis of the Semaglutide side chain via this liquid-phase convergent route represents a significant advancement in process chemistry, offering a clear roadmap for manufacturers seeking to optimize their production capabilities. The protocol detailed in the patent provides specific parameters for temperature, stoichiometry, and solvent systems that have been validated to ensure reproducibility and high yield. For R&D teams looking to implement this technology, the following guide outlines the critical operational phases, emphasizing the importance of strict adherence to reaction conditions to maintain product quality. Detailed standardized synthesis steps are provided in the guide below to facilitate technology transfer and process validation.
- Protect 2-(2-aminoethoxy)ethanol with R1 (e.g., Boc), react with ethyl bromoacetate, and perform one-pot ester hydrolysis to yield Compound 4.
- Protect the carboxyl group of Compound 4 with R2 (e.g., Bn), remove R1 to obtain Compound 6, and condense with Fmoc-Glu(OtBu)-OH to form Compound 8.
- Deprotect R2 from Compound 8, couple with another unit of Compound 6 to form Compound 10, remove Fmoc, couple with the fatty acid chain, and finally deprotect R2 to yield the target Side Chain 1.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from solid-phase to this liquid-phase convergent synthesis offers tangible benefits that directly impact the bottom line and operational resilience. The elimination of expensive solid supports (resins) and the reduction in solvent volumes translate into a leaner manufacturing process with a smaller physical footprint. Moreover, the ability to purify intermediates via crystallization or extraction rather than preparative HPLC significantly lowers capital expenditure on equipment and consumables. This process intensification leads to substantial cost savings in raw material procurement and waste management, making the supply of this critical intermediate more stable and less susceptible to market fluctuations in resin availability.
- Cost Reduction in Manufacturing: The implementation of one-pot reaction sequences significantly reduces the number of unit operations, thereby lowering energy consumption and labor costs associated with intermediate isolation and drying. By avoiding the use of stoichiometric excesses of amino acids typically required in solid-phase synthesis to overcome diffusion limitations, the process achieves higher atom economy. The use of common, inexpensive reagents like sodium hydroxide for hydrolysis and standard coupling agents further drives down the cost of goods sold (COGS), enabling competitive pricing for the final API without compromising quality.
- Enhanced Supply Chain Reliability: Liquid-phase synthesis relies on commodity chemicals and solvents that are readily available from multiple global suppliers, reducing the risk of supply chain disruptions associated with specialized resins or proprietary reagents. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent grades, ensures consistent production output even when facing minor variations in raw material quality. This reliability is crucial for maintaining continuous manufacturing schedules and meeting the aggressive delivery timelines demanded by top-tier pharmaceutical companies.
- Scalability and Environmental Compliance: The homogeneous nature of the liquid-phase reactions facilitates straightforward scale-up from kilogram to multi-ton production scales without the engineering challenges posed by swelling resins or filtration bottlenecks. Furthermore, the reduced solvent usage and the ability to recycle mother liquors contribute to a greener manufacturing profile, aligning with increasingly stringent environmental regulations and corporate sustainability goals. The simplified waste stream, devoid of solid resin waste, simplifies disposal and lowers the environmental compliance burden on the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this liquid-phase synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in Patent CN111253287A, providing clarity on process feasibility and quality control measures. Understanding these aspects is essential for stakeholders evaluating the adoption of this method for large-scale production.
Q: What are the primary advantages of liquid-phase synthesis over solid-phase synthesis for Semaglutide side chains?
A: Liquid-phase synthesis offers superior atom economy compared to solid-phase methods, significantly reducing raw material waste and solvent consumption. It allows for easier monitoring of reaction progress via TLC or HPLC and facilitates large-scale purification through crystallization or extraction rather than expensive preparative HPLC, leading to substantial cost reductions in manufacturing.
Q: How does the convergent strategy improve production efficiency?
A: The convergent strategy involves synthesizing distinct fragments (such as the PEG linker and the fatty acid chain) separately and coupling them at a later stage. This approach minimizes the number of sequential steps required for the longest linear sequence, thereby improving overall yield and reducing the accumulation of impurities that often occur in long linear syntheses.
Q: What specific protecting group strategies are employed in this patent?
A: The patent utilizes an orthogonal protecting group strategy, employing groups such as Boc, Fmoc, Trt, and Benzyl (Bn). This allows for selective deprotection at specific stages without affecting other functional groups, ensuring high purity and structural integrity of the complex side chain molecule throughout the multi-step synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Semaglutide Side Chain Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the successful commercialization of life-saving medications like Semaglutide. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify identity and potency at every stage of the manufacturing process. Our state-of-the-art facilities are equipped to handle the specific solvent systems and reaction conditions required for this liquid-phase convergent synthesis, guaranteeing a consistent and reliable supply for our global partners.
We invite you to collaborate with us to leverage this advanced synthesis technology for your supply chain needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your project's timeline and budget. Let us be your strategic partner in navigating the complexities of peptide intermediate production and securing your position in the rapidly evolving GLP-1 market.
