Advanced Manufacturing of Palbociclib Intermediates via Optimized Bromination Technology
Advanced Manufacturing of Palbociclib Intermediates via Optimized Bromination Technology
The global demand for CDK4/6 inhibitors, particularly palbociclib, has surged following its approval as a first-line treatment for HR-positive, HER2-negative advanced breast cancer. At the heart of this supply chain lies the critical intermediate, 6-bromo-2-chloro-8-cyclopentyl-5-methylpyrido[2,3-d]pyrimidin-7(8H)-one, often referred to as Compound (1). Recent intellectual property developments, specifically patent CN112661753A, have unveiled a transformative preparation method that addresses long-standing bottlenecks in yield, cost, and environmental compliance. This technical insight report analyzes the proprietary synthesis route disclosed in the patent, highlighting its potential to redefine the manufacturing landscape for reliable palbociclib intermediate suppliers. By shifting away from transition-metal catalysis toward a highly efficient electrophilic substitution strategy, this technology offers a robust pathway for cost reduction in pharmaceutical intermediates manufacturing while ensuring the stringent purity profiles required for oncology drug substances.
![Chemical structure of 6-bromo-2-chloro-8-cyclopentyl-5-methylpyrido[2,3-d]pyrimidin-7(8H)-one (Compound 1)](/insights/img/palbociclib-intermediate-synthesis-pharma-supplier-20260314061634-01.png)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the pyrido[2,3-d]pyrimidine core has relied heavily on palladium-catalyzed cross-coupling reactions, such as the Heck reaction described in prior art like CN105008357A. In these conventional routes, 5-bromo-2,4-dichloropyrimidine is reacted with cyclopentylamine, followed by a cyclization step utilizing palladium acetate (Pd(OAc)2) as a catalyst. While chemically feasible, this approach presents severe commercial disadvantages for large-scale operations. The reliance on precious metal catalysts introduces significant cost volatility, as palladium prices are subject to global market fluctuations. Furthermore, the removal of trace palladium residues to meet ICH Q3D guidelines for elemental impurities requires additional purification steps, such as scavenging or recrystallization, which inevitably erode overall yield. Additionally, the use of ethanol and triethylamine in earlier steps generates wastewater laden with organic salts, complicating effluent treatment and increasing the environmental footprint of the facility.
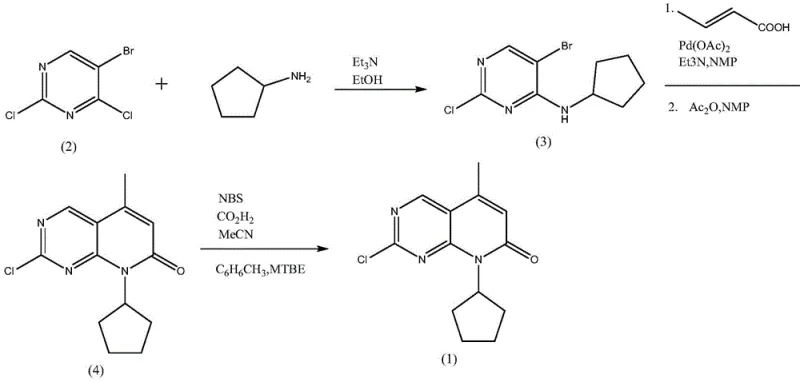
The Novel Approach
In stark contrast, the methodology outlined in CN112661753A bypasses the need for transition metals entirely by employing a direct bromination strategy on a pre-formed bicyclic ketone scaffold. The process utilizes 2-chloro-8-cyclopentyl-5-methylpyrido[2,3-d]pyrimidin-7(8H)-one as the starting material, subjecting it to N-bromosuccinimide (NBS) bromination in the presence of oxalic acid and acetic anhydride. This shift represents a paradigm change in process chemistry, moving from a catalytic cycle dependent on expensive ligands and metals to a stoichiometric reagent-based transformation that is inherently more predictable and scalable. The elimination of the palladium catalyst not only removes a major cost driver but also simplifies the regulatory dossier by avoiding heavy metal contamination risks. Moreover, the solvent system is optimized for recovery, allowing the mother liquor to be distilled and reused, which aligns perfectly with the principles of green chemistry and sustainable manufacturing practices demanded by modern regulatory bodies.
Mechanistic Insights into NBS Bromination with Acid Catalysis
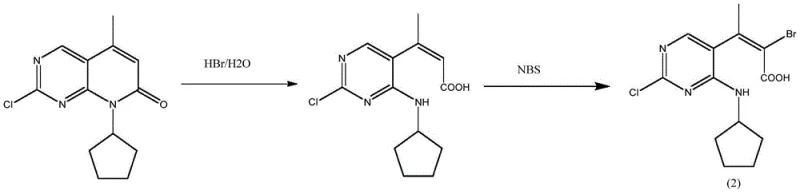
The core innovation of this patent lies in the specific combination of NBS with oxalic acid and acetic anhydride to achieve high regioselectivity at the C6 position of the pyridopyrimidine ring. Mechanistically, the reaction proceeds via an electrophilic aromatic substitution where the succinimidyl radical or cationic bromine species attacks the electron-rich position adjacent to the nitrogen. The presence of oxalic acid likely serves to activate the NBS or stabilize the transition state, facilitating the bromination under relatively mild thermal conditions of 50-60°C. Crucially, the addition of acetic anhydride plays a dual role: it acts as a dehydrating agent to maintain anhydrous conditions and, more importantly, it acetylates potential nucleophilic sites that could otherwise lead to side reactions. This careful modulation of the reaction environment ensures that the bromine atom is installed exclusively at the desired position without affecting the chloro-substituent or the cyclopentyl ring, preserving the structural integrity of the complex heterocycle.
One of the most significant technical achievements of this route is the effective suppression of impurity formation, specifically the hydrolysis byproduct designated as Formula (2) in the patent literature. In standard aqueous or protic workups, the reactive imine or enamine functionalities within the pyrimidine ring are prone to hydrolysis, leading to ring-opening or oxidation products that are difficult to separate. The patent data indicates that the inclusion of acetic anhydride effectively caps these reactive intermediates or maintains a non-aqueous environment that prevents hydrolytic degradation. As illustrated in the structural analysis, avoiding the formation of Formula (2) is critical because this impurity shares similar polarity and solubility characteristics with the target product, making chromatographic separation costly and inefficient. By preventing its formation at the source, the process achieves an impressive HPLC purity of over 98.5% directly after crystallization, drastically reducing the burden on downstream purification units.
How to Synthesize 6-bromo-2-chloro-8-cyclopentyl-5-methylpyrido[2,3-d]pyrimidin-7(8H)-one Efficiently
The operational protocol for this synthesis is designed for seamless integration into existing multipurpose reactor trains commonly found in fine chemical facilities. The process begins with the suspension of the ketone precursor in acetonitrile, followed by the sequential addition of the catalytic system and the brominating agent. The reaction is then heated to a controlled temperature range of 50-60°C and maintained for 10-12 hours to ensure complete conversion. Upon completion, the mixture is cooled to 0-10°C to induce crystallization, leveraging the differential solubility of the product in cold acetonitrile. The solid is isolated via filtration or centrifugation and washed to remove residual succinimide and unreacted starting materials. For a comprehensive, step-by-step breakdown of the exact mass ratios, stirring speeds, and safety parameters required for GMP-compliant production, please refer to the standardized operating procedure detailed below.
- Dissolve 2-chloro-8-cyclopentyl-5-methylpyrido[2,3-d]pyrimidin-7(8H)-one in acetonitrile with catalytic oxalic acid and acetic anhydride.
- Add N-bromosuccinimide (NBS) and heat the mixture to 50-60°C for 10-12 hours to effect bromination.
- Cool the reaction to 0-10°C to crystallize the product, filter, and wash with acetonitrile to obtain high-purity intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits that extend beyond simple unit price negotiations. The primary advantage is the drastic simplification of the bill of materials (BOM). By eliminating palladium acetate and specialized phosphine ligands, the process relies on commodity chemicals like NBS, oxalic acid, and acetic anhydride, which are widely available from multiple global suppliers. This diversification of the supply base mitigates the risk of single-source bottlenecks and protects the project from raw material shortages. Furthermore, the ability to recover and reuse acetonitrile with high purity (over 99.9% GC detection) means that solvent consumption costs are significantly reduced over the lifecycle of the campaign. The recovery of succinimide as a saleable byproduct further offsets the cost of goods sold (COGS), turning a waste stream into a revenue-generating asset.
- Cost Reduction in Manufacturing: The economic impact of removing the palladium catalyst cannot be overstated. In traditional routes, the catalyst cost can account for a substantial portion of the total variable cost, especially when considering the loss of metal during workup and the expense of metal scavengers. This new method completely eradicates that line item. Additionally, the high yield of approximately 97% minimizes the amount of starting material required per kilogram of finished good, directly improving material efficiency. The qualitative reduction in waste treatment costs, due to the recycling of solvents and the recovery of solid byproducts, further enhances the overall margin profile, making the final API more competitive in tender situations.
- Enhanced Supply Chain Reliability: Supply continuity is paramount for life-saving oncology medications. The robustness of this bromination process, demonstrated successfully at the 100 kg scale in the patent examples, suggests a high degree of scalability to multi-ton production without significant re-engineering. The reaction conditions are mild (50-60°C) and do not require cryogenic cooling or high-pressure equipment, reducing the risk of batch failures due to equipment malfunction. Moreover, the simplified purification workflow—essentially a cool-down and filter operation—shortens the cycle time per batch. This increased throughput capacity allows manufacturers to respond more agilely to demand spikes, ensuring a steady flow of high-purity pharmaceutical intermediates to downstream API producers.
- Scalability and Environmental Compliance: Modern chemical manufacturing is increasingly constrained by environmental regulations regarding volatile organic compound (VOC) emissions and hazardous waste disposal. This process excels in sustainability metrics by implementing a closed-loop solvent recovery system. The distillation of mother liquor allows for the reuse of acetonitrile, drastically cutting down on fresh solvent purchases and hazardous waste generation. The recovery of succinimide from the residue prevents solid waste accumulation. These features not only lower the environmental tax burden but also future-proof the supply chain against tightening ecological regulations. The absence of heavy metals simplifies the wastewater treatment profile, making it easier for facilities to maintain their operating permits and avoid regulatory fines.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. They are derived from a deep analysis of the experimental data and comparative advantages presented in the patent documentation. Understanding these nuances is essential for R&D teams evaluating technology transfer and for quality assurance teams assessing the risk profile of the new supply chain. The answers reflect the specific improvements in yield, purity, and waste management that distinguish this method from legacy processes.
Q: How does this method improve upon traditional palladium-catalyzed routes?
A: This method eliminates the need for expensive palladium catalysts (like Pd(OAc)2) used in Heck reactions, significantly reducing raw material costs and removing the complex step of heavy metal residue clearance required for API compliance.
Q: What is the role of acetic anhydride in this bromination process?
A: Acetic anhydride acts as a critical additive that suppresses the formation of specific hydrolysis impurities (Formula 2), thereby boosting the reaction yield to approximately 97% and simplifying downstream purification.
Q: Is the solvent system sustainable for large-scale production?
A: Yes, the process utilizes acetonitrile which can be distilled and recovered with over 99.9% purity from the mother liquor, allowing for closed-loop recycling that drastically reduces liquid waste discharge and operational expenditure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-bromo-2-chloro-8-cyclopentyl-5-methylpyrido[2,3-d]pyrimidin-7(8H)-one Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory discovery to commercial reality requires a partner with deep technical expertise and unwavering commitment to quality. Our team has extensively analyzed the route disclosed in CN112661753A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production necessary to bring this efficient bromination process to life. We understand that consistency is key in the pharmaceutical sector; therefore, our facilities are equipped with rigorous QC labs and advanced analytical instrumentation to ensure that every batch meets stringent purity specifications, including the critical control of the Formula (2) impurity. Our engineering team is ready to optimize the solvent recovery loops and crystallization parameters to maximize your yield and minimize your environmental footprint.
We invite you to collaborate with us to secure a stable and cost-effective supply of this vital oncology intermediate. Whether you are looking to optimize an existing supply chain or qualify a new vendor for an upcoming IND filing, our technical procurement team is prepared to support your needs. Please contact us to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are eager to provide specific COA data from our pilot runs and conduct detailed route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your project timelines and enhance your bottom line.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
