Revolutionizing Palbociclib Parent Nucleus Production via Cytosine-Based Catalytic Routes
The pharmaceutical landscape for oncology treatments continues to evolve rapidly, with CDK4/6 inhibitors like Palbociclib standing as cornerstone therapies for advanced breast cancer. However, the economic viability of these life-saving drugs often hinges on the efficiency of their intermediate supply chains. Patent CN111362939A introduces a transformative preparation method for the Palbociclib parent nucleus structure compound, specifically targeting the critical bottleneck of raw material availability and process selectivity. By shifting the synthetic starting point from scarce, expensive trisubstituted pyrimidines to widely available Cytosine, this technology fundamentally alters the cost structure and scalability of high-purity Pharmaceutical Intermediates manufacturing. The disclosed route not only simplifies the operational complexity but also drastically improves the impurity profile, offering a robust solution for reliable API intermediate supplier networks seeking to secure long-term production stability.
This technical breakthrough addresses the urgent need for cost reduction in Pharmaceutical Intermediates manufacturing by leveraging a five-step sequence that maximizes atom economy and minimizes waste. The strategic use of Cytosine (CAS 71-30-7) as the foundational building block allows for precise control over halogenation patterns, a challenge that has plagued previous generations of synthesis routes. For R&D directors and procurement strategists, understanding this shift is crucial, as it represents a move away from supply-constrained specialty chemicals toward commodity-grade starting materials without sacrificing the stringent quality standards required for oncology drug substances.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the Palbociclib core has relied heavily on routes described in patents such as WO2014128588, which utilize 5-bromo-2,4-dichloropyrimidine as the primary starting material. This conventional approach suffers from inherent chemical limitations, primarily due to the presence of multiple halogen atoms with similar reactivity profiles on the pyrimidine ring. The similarity in activity between the halogens at the 2, 4, and 5 positions leads to significant challenges in regioselectivity during nucleophilic substitution reactions. Consequently, manufacturers often face a proliferation of isomeric impurities that are structurally similar to the desired product, making purification extremely difficult and costly. Furthermore, the starting material itself, 5-bromo-2,4-dichloropyrimidine, is a specialized chemical with limited global suppliers, creating a fragile supply chain vulnerable to price volatility and availability disruptions. The cumulative effect of these factors is a process with a relatively low overall yield, often hovering around 50%, and a high environmental footprint due to the extensive solvent usage and purification steps required to meet pharmaceutical grade specifications.
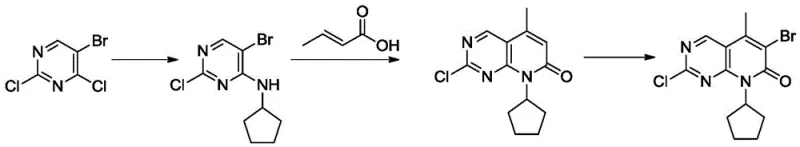
The Novel Approach
In stark contrast, the novel methodology disclosed in CN111362939A reconstructs the synthetic pathway from the ground up, utilizing Cytosine or its immediate derivatives (Intermediate 1 or 2) as the entry point. This strategy effectively bypasses the regioselectivity nightmare of the trisubstituted pyrimidine route by introducing functional groups in a sequential, controlled manner. The process begins with the chlorination of Cytosine to form 2-chloro-4-aminopyrimidine, followed by a highly selective bromination to install the bromine atom specifically at the 5-position. This stepwise construction ensures that each reaction occurs at a distinct chemical environment, virtually eliminating the formation of difficult-to-remove isomers. Moreover, the subsequent introduction of the cyclopentyl group and the construction of the pyridone ring via Heck coupling are optimized for high conversion rates. By replacing the expensive and problematic starting material with Cytosine, the new route not only enhances the chemical robustness of the process but also aligns perfectly with the goals of commercial scale-up of complex Pharmaceutical Intermediates, offering a pathway that is both economically superior and operationally simpler.
Mechanistic Insights into Pd-Catalyzed Cyclization and Regioselective Halogenation
The heart of this synthetic innovation lies in the sophisticated orchestration of transition metal catalysis and electrophilic aromatic substitution. A critical transformation in this route is the conversion of Intermediate 3 to Intermediate 4, which involves a Palladium-catalyzed reaction with crotonic acid. This step effectively constructs the fused pyrido[2,3-d]pyrimidine skeleton through a tandem Heck coupling and intramolecular amidation cyclization mechanism. The choice of palladium catalyst, such as PdCl2 or Pd(OAc)2, alongside a suitable base like triethylamine, facilitates the oxidative addition into the carbon-halogen bond of the pyrimidine ring. This is followed by migratory insertion of the alkene from crotonic acid and subsequent beta-hydride elimination, which sets the stage for the ring closure. The precision of this catalytic cycle is paramount, as it dictates the formation of the methyl group at the 5-position and the integrity of the lactam ring, ensuring that the core scaffold is formed with minimal side reactions.
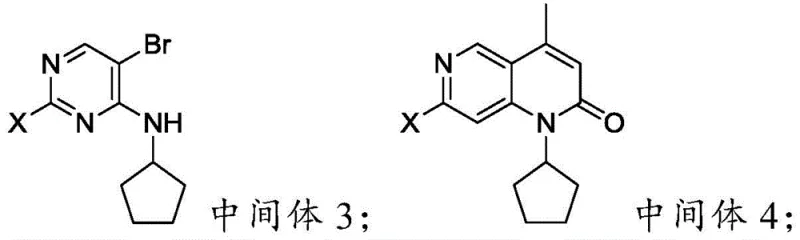
Following the skeleton construction, the final bromination step to yield Formula (I) demonstrates exceptional regiocontrol. Unlike the indiscriminate halogenation seen in older methods, this process utilizes specific catalyst-solvent systems, such as KBr in glacial acetic acid or NBS in acetonitrile, to target the 6-position of the pyrido-pyrimidinone system exclusively. The electron density distribution of the Intermediate 4 molecule, influenced by the electron-withdrawing carbonyl and the electron-donating cyclopentyl amino group, directs the electrophilic bromine species to the desired position. This mechanistic understanding allows for the suppression of poly-brominated byproducts, which are common impurities in less optimized processes. The result is a final product with a purity profile exceeding 99.3%, significantly reducing the burden on downstream purification units and ensuring that the high-purity Pharmaceutical Intermediates produced are ready for the subsequent coupling reactions required to complete the Palbociclib API synthesis.
How to Synthesize Palbociclib Parent Nucleus Efficiently
The implementation of this synthesis route requires careful attention to reaction conditions, particularly temperature control and stoichiometry, to maximize the benefits of the novel pathway. The process is designed to be linear and convergent, allowing for the isolation of stable intermediates that can be quality-controlled at each stage. From the initial chlorination of Cytosine in toluene to the final recrystallization of the parent nucleus, each step has been engineered to balance reaction kinetics with ease of workup. For process chemists looking to adopt this technology, the detailed procedural breakdown below outlines the critical parameters for achieving optimal yields and purity.
- Perform chlorination of Cytosine using phosphorus pentachloride in toluene to generate 2-chloro-4-aminopyrimidine (Intermediate 1).
- Execute selective bromination of Intermediate 1 with bromine and KBr catalyst in glacial acetic acid to form 5-bromo-2-chloro-4-aminopyrimidine (Intermediate 2).
- Conduct nucleophilic substitution with halogenated cyclopentane to attach the cyclopentyl group, followed by Palladium-catalyzed Heck cyclization with crotonic acid.
- Finalize the synthesis via regioselective bromination of the pyrido-pyrimidinone core to obtain the target Formula (I) compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this Cytosine-based route translates directly into enhanced operational resilience and financial efficiency. The most immediate impact is the decoupling of production from the volatile market of specialized halogenated pyrimidines. By anchoring the synthesis on Cytosine, a commodity chemical with a stable and diverse global supply base, manufacturers can mitigate the risks associated with single-source dependencies. This shift not only stabilizes the cost of goods sold (COGS) but also provides the flexibility to scale production volumes rapidly in response to market demand without being constrained by the lead times of exotic starting materials. Furthermore, the simplified purification protocols reduce the consumption of solvents and energy, contributing to a leaner, more sustainable manufacturing operation that aligns with modern green chemistry initiatives.
- Cost Reduction in Manufacturing: The elimination of expensive 5-bromo-2,4-dichloropyrimidine as a starting material results in substantial raw material cost savings. Additionally, the improved regioselectivity minimizes the formation of isomeric impurities, which drastically reduces the need for complex and yield-losing purification steps like preparative chromatography. The overall process yield improvement, potentially exceeding 65% compared to the ~50% of legacy routes, means that less raw material is wasted per kilogram of final product, driving down the effective unit cost significantly.
- Enhanced Supply Chain Reliability: Sourcing Cytosine and basic halogenating agents like phosphorus pentachloride and bromine is far more reliable than sourcing specialized trisubstituted heterocycles. This abundance ensures that production schedules are not disrupted by upstream supply shortages. The robustness of the intermediates also allows for safer storage and transportation, reducing the logistical complexities often associated with unstable or highly reactive chemical precursors in the global supply chain.
- Scalability and Environmental Compliance: The process utilizes common industrial solvents such as toluene, methanol, and acetic acid, which are easily recovered and recycled in standard distillation units. The avoidance of heavy metal contamination issues, beyond the standard palladium catalyst which is efficiently removed, simplifies the waste treatment process. This makes the route highly scalable from pilot batches to multi-ton commercial production, ensuring that reducing lead time for high-purity Pharmaceutical Intermediates is achievable without compromising environmental compliance or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering a clear perspective on the practical benefits for industrial application.
Q: Why is the Cytosine-based route superior to traditional 2,4,5-trisubstituted pyrimidine methods?
A: Traditional methods rely on expensive 5-bromo-2,4-dichloropyrimidine where similar halogen activities lead to poor regioselectivity and difficult purification. The Cytosine route offers abundant raw materials, distinct reactivity steps for higher selectivity, and significantly improved overall yield exceeding 65%.
Q: How does this process ensure high purity for pharmaceutical applications?
A: The process utilizes specific crystallization steps, such as ice-water bath crystallization for intermediates and mixed solvent recrystallization (toluene/n-hexane) for the final product. This rigorous purification protocol ensures the final Palbociclib parent nucleus achieves purity levels greater than 99.3%, minimizing downstream processing burdens.
Q: Is this synthesis method scalable for commercial API production?
A: Yes, the method is designed for industrial scalability. It avoids complex chromatographic separations, uses common solvents like toluene and acetic acid, and employs robust catalytic systems (Pd-catalyzed cyclization) that are well-suited for large-scale reactor operations ranging from pilot plants to multi-ton commercial facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Palbociclib Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to more efficient synthetic routes is critical for maintaining competitiveness in the global oncology market. Our team of expert process chemists has extensively evaluated the Cytosine-based pathway described in CN111362939A and confirmed its viability for large-scale manufacturing. We possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a GMP-compliant environment. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of Palbociclib intermediate we deliver supports the highest standards of patient safety and drug efficacy.
We invite pharmaceutical partners to collaborate with us to leverage this technological advantage for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to discuss specific COA data and route feasibility assessments, ensuring that your production of Palbociclib and related CDK4/6 inhibitors remains cost-effective, reliable, and ready for the demands of the future market.
