Advanced Catalytic Strategy for High-Purity L-Thyroxine Sodium Commercial Manufacturing
Introduction to Advanced L-Thyroxine Sodium Synthesis Technology
The pharmaceutical industry continuously seeks robust manufacturing pathways for critical hormonal therapies, and L-thyroxine sodium remains a cornerstone treatment for hypothyroidism and related metabolic disorders. A significant technological breakthrough in this domain is documented in patent CN102199103B, which outlines an improved method for synthesizing L-thyroxine sodium with enhanced efficiency and purity profiles. This innovation specifically targets the oxidative coupling reaction, historically the bottleneck step limiting overall process yield and economic viability. By transitioning from traditional manganese sulfate catalysts to more soluble manganese chloride or nitrate variants, the process achieves a dramatic improvement in reaction homogeneity. This technical refinement not only elevates the yield of the key intermediate, N-acetyl-L-thyroxine ethyl ester, but also ensures the final active pharmaceutical ingredient meets rigorous international quality specifications. For global procurement teams and R&D directors, understanding this catalytic evolution is essential for securing a reliable pharmaceutical intermediates supplier capable of delivering consistent, high-volume production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the chemical synthesis of L-thyroxine sodium has been plagued by inefficiencies in the oxidative coupling stage, where two iodinated tyrosine derivatives are joined to form the thyroxine backbone. Conventional methodologies predominantly utilized manganese sulfate as the catalyst for this transformation in an ethanol solvent system. However, manganese sulfate suffers from inherently poor solubility in ethanol, leading to a heterogeneous reaction mixture that severely restricts mass transfer and catalytic contact. Literature and prior art indicate that under these suboptimal conditions, the yield of the oxidative coupling reaction stagnates at approximately 42%, resulting in a dismal overall total yield of merely 11.3% for the entire synthetic sequence. This low conversion rate necessitates the processing of excessive raw materials to achieve target output volumes, thereby inflating production costs and generating substantial chemical waste. Furthermore, the heterogeneity of the reaction often leads to inconsistent batch quality, complicating the purification process and risking the presence of difficult-to-remove impurities that could compromise the safety profile of the final drug substance.
The Novel Approach
The innovative strategy presented in the patent data fundamentally re-engineers the catalytic environment to overcome these solubility barriers. By substituting manganese sulfate with manganese chloride or manganese nitrate, the process leverages the superior solubility characteristics of these salts in ethanol to create a truly homogeneous catalytic system. This shift ensures that the catalyst is molecularly dispersed throughout the reaction medium, maximizing the interaction between the catalytic metal centers and the organic substrate. As demonstrated in the experimental data, this modification propels the yield of the oxidative coupling reaction from the historical baseline of 42% to an impressive range of 61.5% to 62.5%. 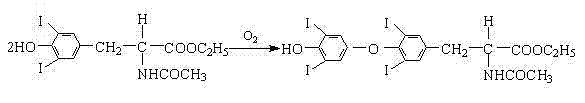 This substantial gain in efficiency is further optimized by a split-addition protocol for the catalyst, where at least 60% is introduced initially to drive the reaction onset, followed by the remainder to maintain activity over the extended reaction time. This novel approach not only enhances cost reduction in pharmaceutical intermediates manufacturing by minimizing raw material waste but also significantly streamlines the downstream purification workload, offering a compelling value proposition for large-scale commercial production.
This substantial gain in efficiency is further optimized by a split-addition protocol for the catalyst, where at least 60% is introduced initially to drive the reaction onset, followed by the remainder to maintain activity over the extended reaction time. This novel approach not only enhances cost reduction in pharmaceutical intermediates manufacturing by minimizing raw material waste but also significantly streamlines the downstream purification workload, offering a compelling value proposition for large-scale commercial production.
Mechanistic Insights into MnCl2-Catalyzed Oxidative Coupling
The core mechanistic advantage of this improved synthesis lies in the thermodynamics of catalyst solvation within the alcoholic solvent matrix. In the conventional manganese sulfate system, the limited solubility creates a suspension where a significant portion of the catalyst remains inactive in the solid phase, unavailable for the redox cycle required to facilitate the oxidative dimerization of the phenolic rings. In contrast, manganese chloride dissociates readily in ethanol, providing a high concentration of active manganese ions in the solution phase. This homogeneous environment facilitates a more efficient electron transfer process between the oxygen gas, the catalyst, and the diiodotyrosine substrate. The reaction proceeds under mild conditions, typically maintained at a temperature of 40-50°C and a pH of 8-9, which preserves the stereochemical integrity of the L-tyrosine chiral center while promoting the formation of the ether linkage. The use of boric acid as a cocatalyst further stabilizes the reaction environment, likely by coordinating with the phenolic hydroxyl groups to enhance their nucleophilicity or by buffering the local pH near the active sites.
Impurity control is another critical aspect governed by this mechanistic refinement. The higher conversion rate achieved with the manganese chloride system means that less unreacted starting material, specifically N-acetyl-3,5-diiodo-L-tyrosine ethyl ester, remains in the crude product mixture. Residual starting materials are often structurally similar to the product, making them notoriously difficult to separate via crystallization or chromatography. By driving the reaction to higher completion, the load on the purification units is drastically reduced.  Furthermore, the milder reaction conditions and the specific selectivity of the manganese chloride catalyst minimize the formation of side products such as over-oxidized species or racemized byproducts. This results in a crude intermediate with a purity profile that is far superior to that obtained via the sulfate route, ultimately ensuring that the final L-thyroxine sodium API complies with strict limits on related substances, such as keeping triiodothyronine levels below 1% as required by USP standards.
Furthermore, the milder reaction conditions and the specific selectivity of the manganese chloride catalyst minimize the formation of side products such as over-oxidized species or racemized byproducts. This results in a crude intermediate with a purity profile that is far superior to that obtained via the sulfate route, ultimately ensuring that the final L-thyroxine sodium API complies with strict limits on related substances, such as keeping triiodothyronine levels below 1% as required by USP standards.
How to Synthesize N-Acetyl-L-Thyroxine Ethyl Ester Efficiently
Executing this improved synthetic route requires precise adherence to the optimized reaction parameters to fully realize the benefits of the manganese chloride catalyst system. The process begins with the preparation of the reaction vessel, where the esterified precursor, N-acetyl-3,5-diiodo-L-tyrosine ethyl ester, is dissolved in ethanol along with the boric acid cocatalyst. The pH of the solution must be carefully adjusted to the 8-9 range using sodium hydroxide before the introduction of oxygen and the catalyst. The detailed standardized synthesis steps, including specific mass ratios and timing for the split catalyst addition, are outlined in the technical guide below to ensure reproducibility and safety during scale-up operations.
- Prepare the reaction vessel with N-acetyl-3,5-diiodo-L-tyrosine ethyl ester, boric acid cocatalyst, and ethanol solvent, adjusting pH to 8-9.
- Add at least 60% of the total manganese chloride catalyst mass and heat to 40-50°C while passing oxygen for at least 40 hours.
- Add the remaining catalyst and continue the oxidative coupling reaction under oxygen flow for another 40 hours to maximize yield.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis method offers tangible strategic advantages beyond mere technical elegance. The primary driver of value is the substantial improvement in process yield, which directly correlates to a reduction in the cost of goods sold (COGS). By increasing the yield of the rate-limiting oxidative coupling step by nearly 20 percentage points, the consumption of expensive iodinated precursors is significantly lowered per kilogram of final product. This efficiency gain mitigates the impact of raw material price volatility and enhances the overall margin structure of the manufacturing operation. Additionally, the robustness of the homogeneous catalytic system reduces the risk of batch failures, ensuring a more predictable and reliable supply of critical thyroid medication intermediates to the global market.
- Cost Reduction in Manufacturing: The transition to a high-solubility catalyst system eliminates the inefficiencies associated with heterogeneous reactions, leading to a drastic reduction in raw material waste. Since the yield of the key coupling step increases from roughly 42% to over 61%, the amount of starting material required to produce a fixed quantity of L-thyroxine is substantially decreased. This reduction in material intensity translates directly into lower variable costs for every production batch. Furthermore, the improved purity of the crude intermediate reduces the consumption of solvents and energy required for downstream purification and recrystallization steps, compounding the cost savings throughout the entire production workflow without compromising on quality standards.
- Enhanced Supply Chain Reliability: A manufacturing process with higher yields and fewer side reactions is inherently more stable and easier to control at a commercial scale. The use of readily available and highly soluble manganese chloride ensures that the reaction kinetics are consistent from batch to batch, minimizing the variability that often leads to supply disruptions. This reliability is crucial for maintaining continuous inventory levels of L-thyroxine sodium, a life-saving medication for patients with hypothyroidism. By partnering with a manufacturer utilizing this optimized technology, supply chain heads can secure a more resilient source of high-purity pharmaceutical intermediates that is less susceptible to the delays caused by re-processing off-spec batches or sourcing additional raw materials to compensate for low yields.
- Scalability and Environmental Compliance: The reaction conditions specified in this improved method are mild, operating at atmospheric pressure and moderate temperatures between 40-50°C, which simplifies the engineering requirements for large-scale reactors. This ease of scale-up facilitates the commercial scale-up of complex pharmaceutical intermediates from pilot plants to multi-ton annual production capacities without the need for specialized high-pressure equipment. Moreover, the higher atom economy and reduced waste generation align with modern green chemistry principles, lowering the environmental footprint of the manufacturing process. This compliance with environmental standards reduces the regulatory burden and potential liabilities associated with waste disposal, making the supply chain more sustainable and future-proof against tightening environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this improved L-thyroxine sodium synthesis method. These insights are derived directly from the experimental data and technical specifications outlined in patent CN102199103B, providing clarity on the operational benefits and quality outcomes of this catalytic innovation. Understanding these details helps stakeholders evaluate the feasibility and value of integrating this technology into their existing supply networks.
Q: Why is manganese chloride preferred over manganese sulfate for this synthesis?
A: Manganese chloride exhibits significantly higher solubility in ethanol compared to manganese sulfate. This enhanced solubility ensures a more homogeneous catalytic system, which directly improves the reaction rate and overall yield of the oxidative coupling step from approximately 42% to over 61%.
Q: What is the critical parameter for catalyst addition in this process?
A: The catalyst must be added in two stages. The initial addition should comprise at least 60% of the total catalyst mass to initiate the reaction effectively, followed by the remainder to sustain catalytic activity throughout the extended 80-hour reaction period.
Q: Does this method meet international pharmacopoeia standards?
A: Yes, the L-thyroxine sodium produced via this improved method meets the stringent standards of the United States Pharmacopoeia (USP32-NF27), specifically achieving a triiodothyronine impurity level of less than 1%, which is superior to previous standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Thyroxine Sodium Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and compliant manufacturing processes for essential hormonal therapies like L-thyroxine sodium. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of advanced patents like CN102199103B are fully realized in practical, industrial settings. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of our pharmaceutical intermediates meets or exceeds global pharmacopoeia standards, including USP and EP requirements. Our facility is equipped to handle the specific nuances of oxidative coupling chemistries, ensuring that the high yields and purity profiles demonstrated in the lab are consistently delivered at scale.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our expertise in this improved synthesis route, we can help you optimize your supply chain costs while securing a dependable source of high-quality materials. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can support your long-term strategic goals in the pharmaceutical sector.
