Advanced Synthetic Route for High-Purity L-Thyroxine Sodium via Optimized Oxidative Coupling
The pharmaceutical industry continuously seeks robust synthetic pathways for critical hormonal therapies, and the production of L-thyroxine sodium stands as a prime example of process evolution. As detailed in patent CN102199103A, a significant technological breakthrough has been achieved in the synthesis of this vital thyroid hormone, moving away from inefficient animal extraction methods toward a highly controlled chemical manufacturing process. This patent specifically addresses the historical bottlenecks associated with the oxidative coupling reaction, which has traditionally been the limiting step in terms of both yield and optical purity. By re-engineering the catalytic system and optimizing reaction parameters, this methodology offers a compelling solution for producing high-purity active pharmaceutical ingredients (APIs) that meet stringent pharmacopoeial standards such as USP32-NF27. For R&D directors and procurement specialists, understanding these mechanistic improvements is essential for securing a reliable supply chain of high-quality thyroid therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the chemical synthesis of L-thyroxine sodium relied heavily on manganese sulfate as the primary catalyst for the critical oxidative coupling step. However, this conventional approach suffered from inherent physicochemical limitations that severely impacted overall process efficiency. The primary issue lies in the poor solubility of manganese sulfate in ethanol, the standard solvent for this reaction, which leads to a heterogeneous catalytic environment. This heterogeneity results in inconsistent catalyst activity, causing the oxidative coupling yield to stagnate at approximately 42%, as documented in prior literature. Furthermore, the low conversion rates necessitate extensive downstream purification to remove unreacted starting materials and byproducts, thereby inflating production costs and extending lead times. For a reliable pharmaceutical intermediate supplier, these inefficiencies represent a significant barrier to delivering cost-effective solutions to the global market.
The Novel Approach
The innovative methodology described in the patent data fundamentally alters the catalytic landscape by substituting manganese sulfate with manganese chloride or manganese nitrate. This strategic substitution leverages the superior solubility profile of manganese chloride in ethanol, ensuring a truly homogeneous catalytic system that maximizes interaction between the catalyst and the substrate. Additionally, the process introduces a split-addition protocol where the catalyst is introduced in two stages, with the majority added at the onset of the reaction. This technique, combined with the use of boric acid as a cocatalyst and precise pH control between 8 and 9, drives the oxidative coupling yield up to 61.5%. Such improvements not only enhance the economic viability of the process but also significantly reduce the impurity profile, aligning perfectly with the demands for high-purity pharmaceutical intermediates in modern drug manufacturing.
Mechanistic Insights into MnCl2-Catalyzed Oxidative Coupling
The core of this technological advancement lies in the thermodynamic and kinetic advantages provided by the manganese chloride catalytic system. In a homogeneous phase, the diffusion limitations associated with solid catalyst particles are eliminated, allowing for uniform activation of the molecular oxygen required for the coupling reaction. The presence of boric acid further stabilizes the reaction intermediate, facilitating the formation of the ether linkage between the two tyrosine derivatives. This mechanistic refinement is crucial because the oxidative coupling step determines the final stereochemistry and purity of the L-thyroxine molecule. By maintaining the reaction temperature within a narrow window of 40-50°C and ensuring a continuous flow of oxygen, the process minimizes side reactions such as over-oxidation or racemization. This level of control is paramount for R&D teams focused on cost reduction in API manufacturing, as it reduces the burden on downstream chromatographic separation units.
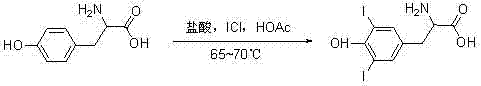
Furthermore, the precursor synthesis plays a vital role in setting the stage for successful coupling. The initial iodination of L-tyrosine, as depicted in the reaction scheme above, must be executed with high precision to ensure the correct positioning of iodine atoms at the 3 and 5 positions of the phenolic ring. Any deviation in this early stage can propagate through the synthesis, leading to difficult-to-remove impurities in the final API. The patent outlines a rigorous protocol involving iodine chloride in acetic acid, followed by careful neutralization and purification. This attention to detail in the early synthetic steps underscores the importance of a holistic approach to process chemistry, where every unit operation contributes to the overall quality and yield of the final commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize L-Thyroxine Sodium Efficiently
Implementing this optimized synthetic route requires strict adherence to the sequential reaction steps outlined in the patent documentation. The process begins with the iodination of L-tyrosine, followed by N-acylation to protect the amine functionality, and subsequently esterification to improve solubility for the coupling step. The critical oxidative coupling is then performed under atmospheric pressure with oxygen sparging, followed by hydrolysis and final salt formation. Each step is designed to maximize material throughput while minimizing waste generation. For technical teams looking to replicate or license this technology, the following guide provides a structural overview of the standardized synthesis protocol required to achieve the reported yields and purity specifications.
- Perform iodination of L-Tyrosine using iodine chloride in acetic acid to form 3,5-diiodo-L-tyrosine.
- Conduct N-acylation with acetic anhydride to protect the amine group, forming N-acetyl-3,5-diiodo-L-tyrosine.
- Execute esterification using ethanol and p-toluenesulfonic acid to produce the ethyl ester substrate.
- Perform the key oxidative coupling using manganese chloride catalyst and boric acid cocatalyst in ethanol under oxygen flow.
- Hydrolyze the coupled ester using hydrochloric and acetic acid to obtain L-Thyroxine.
- Form the final sodium salt using sodium carbonate solution followed by recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this improved synthesis method offers tangible benefits that extend beyond simple yield metrics. For procurement managers and supply chain heads, the stability and predictability of the manufacturing process are key drivers for vendor selection. The shift to a more efficient catalytic system reduces the consumption of raw materials per kilogram of finished product, directly impacting the cost of goods sold. Moreover, the use of readily available reagents like manganese chloride and ethanol simplifies the sourcing strategy, mitigating risks associated with specialized or scarce catalyst availability. This robustness ensures reducing lead time for high-purity pharmaceutical intermediates and guarantees a steady flow of materials to downstream formulation facilities.
- Cost Reduction in Manufacturing: The transition from manganese sulfate to manganese chloride eliminates the inefficiencies caused by poor catalyst solubility, leading to a substantial increase in reaction yield without requiring expensive equipment upgrades. By achieving higher conversion rates in the oxidative coupling step, the process significantly reduces the volume of unreacted starting material that must be recovered or disposed of, thereby lowering waste treatment costs. Additionally, the improved purity of the crude product minimizes the need for multiple recrystallization cycles, saving both solvent and energy resources. These cumulative efficiencies translate into a more competitive pricing structure for the final API, offering significant value to partners focused on cost reduction in pharmaceutical manufacturing.
- Enhanced Supply Chain Reliability: The reliance on common industrial chemicals such as ethanol, hydrochloric acid, and manganese salts ensures that the supply chain is resilient against market fluctuations for exotic reagents. The mild reaction conditions, operating at atmospheric pressure and moderate temperatures, reduce the safety risks and maintenance requirements associated with high-pressure reactors. This operational simplicity allows for greater flexibility in production scheduling and facilitates faster turnaround times between batches. Consequently, suppliers utilizing this technology can offer more reliable delivery schedules, which is critical for pharmaceutical companies managing tight inventory levels and regulatory compliance timelines.
- Scalability and Environmental Compliance: The patent data includes successful scale-up examples, demonstrating that the process is viable for large-scale commercial production without loss of efficiency. The use of ethanol as a primary solvent aligns with green chemistry principles, as it is less toxic and easier to recover compared to chlorinated solvents often used in older methodologies. Furthermore, the reduction in byproduct formation decreases the environmental load of the manufacturing process, simplifying wastewater treatment and regulatory reporting. This alignment with environmental, social, and governance (ESG) goals makes the technology attractive for multinational corporations seeking sustainable supply chain partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this technology into their existing manufacturing portfolios. The answers reflect the specific advantages of the manganese chloride catalytic system and the operational parameters required for success.
Q: Why is manganese chloride preferred over manganese sulfate in this synthesis?
A: Manganese chloride exhibits significantly higher solubility in ethanol compared to manganese sulfate. This enhanced solubility ensures a more homogeneous catalytic system, directly improving catalyst activity and increasing the oxidative coupling yield from approximately 42% to over 61%.
Q: What is the impact of the split-addition catalyst strategy?
A: Adding the manganese chloride catalyst in two portions, with at least 60% added initially, maintains optimal catalytic activity throughout the extended 80-hour reaction period. This prevents catalyst deactivation and ensures consistent conversion rates, minimizing residual starting material.
Q: Is this synthetic route suitable for large-scale commercial production?
A: Yes, the patent demonstrates successful scale-up in Example 10, where 16kg of substrate was processed to yield 8.5kg of product with 62.5% efficiency. The mild reaction conditions (40-50°C) and atmospheric pressure operation facilitate safe and scalable manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Thyroxine Sodium Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes for life-saving medications like L-thyroxine sodium. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are seamlessly translated into industrial reality. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the highest international standards. Our commitment to process excellence allows us to deliver high-quality intermediates and APIs that support the global healthcare infrastructure.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of adopting this technology. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project requirements, ensuring a partnership built on transparency, quality, and mutual growth.
