Advanced Manufacturing of L-Thyroxine Sodium via Optimized Iodination Technology
Advanced Manufacturing of L-Thyroxine Sodium via Optimized Iodination Technology
The pharmaceutical industry continuously seeks robust synthetic pathways that balance high purity with operational safety, particularly for critical endocrine therapeutics like L-thyroxine sodium. Patent CN109810009B introduces a significant technological breakthrough in the synthesis of this vital hormone, specifically addressing the bottlenecks found in the final iodination stages of production. By shifting from hazardous organic amine solvents to a benign inorganic base system, this methodology not only enhances the safety profile of the manufacturing process but also delivers substantial improvements in yield and cost efficiency. This report analyzes the technical merits of this five-step sequence, starting from 3,5-diiodo-L-tyrosine, and evaluates its potential for commercial adoption by forward-thinking pharmaceutical enterprises seeking reliable supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of L-thyroxine has been plagued by the reliance on methylamine alcohol solutions during the critical iodination phase. These organic amine solvents present severe logistical and safety challenges, as they are classified as easy-to-detonate controlled chemicals in many jurisdictions, making their procurement difficult and subject to strict regulatory oversight. Furthermore, the pungent odor and high volatility of these amines create unpleasant working environments and necessitate expensive containment infrastructure to prevent atmospheric release. From a process chemistry perspective, the use of organic solvents often complicates downstream purification, requiring energy-intensive distillation or extraction steps to remove residual amines that could otherwise contaminate the final active pharmaceutical ingredient. These factors collectively drive up the cost of goods sold and introduce significant supply chain fragility, as any disruption in the availability of controlled precursors can halt production lines entirely.
The Novel Approach
The innovative strategy outlined in the patent data fundamentally re-engineers the iodination step by substituting the problematic organic amines with an aqueous sodium hydroxide system. This transition allows the reaction to proceed under alkaline conditions where the substrate is fully soluble, facilitating a more homogeneous and controllable reaction environment. By utilizing water as the primary solvent, the process eliminates the fire and explosion risks associated with volatile organics, thereby simplifying facility safety requirements and reducing insurance and compliance costs. Moreover, the use of inorganic bases enables precise pH control during the reaction quench, which is critical for precipitating the product in high purity without the need for complex chromatographic separations. This methodological shift represents a paradigm change in thyroid hormone manufacturing, prioritizing green chemistry principles while simultaneously enhancing the economic viability of large-scale production campaigns.
Mechanistic Insights into Copper-Mediated Coupling and Electrophilic Iodination
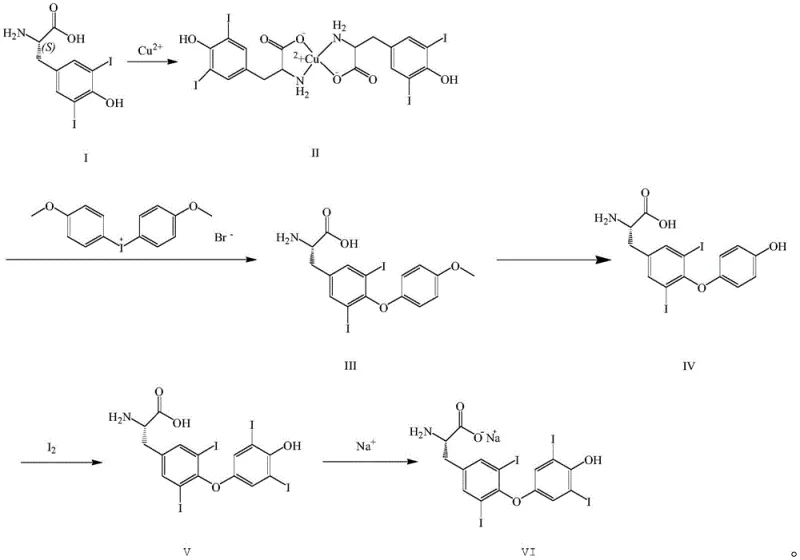
The synthetic pathway begins with the formation of a copper complex, where 3,5-diiodo-L-tyrosine coordinates with copper ions in an alkaline medium to form a stable intermediate. This copper chelation serves a dual purpose: it protects the amino group from unwanted side reactions during the subsequent coupling phase and activates the aromatic ring for nucleophilic attack. The coupling reaction with dianisole iodonium bromide proceeds through a nucleophilic aromatic substitution mechanism, where the phenoxide oxygen attacks the electron-deficient aromatic ring of the iodonium salt. Maintaining the copper complex integrity during this step is crucial, as premature decomplexation could lead to racemization or polymerization, compromising the optical purity of the final drug substance. The stability of this intermediate ensures that the diphenyl ether linkage is formed with high regioselectivity, setting the stage for the subsequent structural modifications.
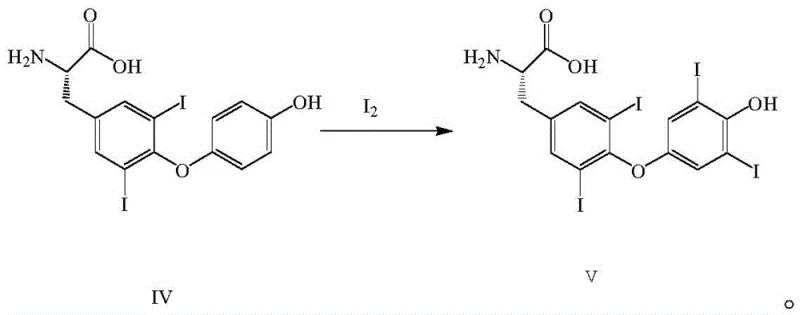
The cornerstone of this patent is the optimized electrophilic aromatic substitution during the iodination of the thyronine precursor. In this step, elemental iodine acts as the electrophile, attacking the activated positions on the phenolic ring of the substrate. The use of sodium hydroxide creates a phenoxide anion, which is significantly more reactive towards electrophilic attack than the neutral phenol, allowing the reaction to proceed efficiently at low temperatures ranging from -10 to 10°C. Controlling the stoichiometry is paramount; the patent specifies a molar ratio of iodine to substrate of approximately 3:1 to ensure complete di-iodination without excessive waste or the formation of poly-iodinated impurities. The reaction is quenched by adjusting the pH to 4-5, which protonates the carboxylate and phenoxide groups, causing the L-thyroxine to precipitate out of the aqueous solution. This precise control over reaction parameters minimizes the formation of regio-isomers and ensures that the final product meets the stringent purity specifications required for endocrine therapies.
How to Synthesize L-Thyroxine Sodium Efficiently
The execution of this synthetic route requires careful attention to reaction conditions, particularly temperature control during the exothermic iodination phase and pH management during workup. The process is designed to be linear and scalable, moving from the initial copper complexation through to the final salt formation without the need for intermediate isolation of unstable species. Operators must ensure that the copper complex is thoroughly washed to remove excess metal ions before coupling, as residual copper can catalyze decomposition in later steps. The detailed standardized synthesis steps below outline the specific operational parameters derived from the patent examples, providing a clear roadmap for process chemists aiming to replicate this high-yielding pathway in a pilot or production setting.
- Perform copper complexation of 3,5-diiodo-L-tyrosine using copper sulfate and sodium hydroxide to form the stable copper complex intermediate.
- Execute the coupling reaction with dianisole iodonium bromide at elevated temperatures (80-120°C) to establish the diphenyl ether linkage.
- Conduct acid hydrolysis using a mixture of acetic acid and hydroiodic acid to remove protecting groups and yield the thyronine precursor.
- Perform the critical iodination step using elemental iodine in an aqueous sodium hydroxide solution at low temperatures (-10 to 10°C).
- Complete the synthesis by reacting the free acid with boiling sodium carbonate solution to form the final L-thyroxine sodium salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this improved synthesis method offers tangible strategic benefits that extend beyond simple yield metrics. By eliminating the dependency on controlled organic amines, manufacturers can drastically simplify their raw material sourcing strategies, reducing the administrative burden associated with handling hazardous chemicals. The shift to aqueous systems also implies a significant reduction in solvent recovery costs and waste disposal fees, as water is inherently cheaper and easier to treat than organic waste streams. These operational efficiencies translate directly into a more resilient supply chain, where production continuity is less likely to be disrupted by regulatory changes or supplier shortages of specialized reagents. Furthermore, the enhanced safety profile of the process lowers the barrier for contract manufacturing organizations to accept production orders, expanding the pool of potential partners for capacity expansion.
- Cost Reduction in Manufacturing: The replacement of expensive and difficult-to-source methylamine alcohol solutions with commodity-grade sodium hydroxide and water results in a direct reduction in raw material expenditure. Additionally, the simplified workup procedure, which relies on precipitation and filtration rather than complex extractions or distillations, reduces energy consumption and labor hours per batch. The elimination of hazardous waste streams further decreases the environmental compliance costs associated with solvent incineration or treatment, contributing to a leaner overall cost structure for the final API.
- Enhanced Supply Chain Reliability: Sourcing inorganic bases like sodium hydroxide and elemental iodine is far more stable and predictable than procuring controlled organic amines, which are subject to fluctuating availability and strict government quotas. This reliability ensures that production schedules can be maintained without the risk of sudden stoppages due to raw material shortages. The robustness of the aqueous chemistry also means that the process is less sensitive to minor variations in reagent quality, providing a buffer against supply chain variability and ensuring consistent batch-to-batch quality for downstream customers.
- Scalability and Environmental Compliance: The use of water as a primary solvent inherently improves the scalability of the process, as heat transfer and mixing are more efficient in aqueous media compared to viscous organic solutions. This facilitates the transition from laboratory scale to multi-ton commercial production with minimal engineering modifications. From an environmental standpoint, the reduction in volatile organic compound (VOC) emissions aligns with increasingly stringent global environmental regulations, positioning the manufacturer as a responsible partner committed to sustainable chemistry practices and long-term regulatory compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic route. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on how this technology resolves historical manufacturing pain points. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this process into their existing production portfolios.
Q: Why is the new iodination method safer than traditional routes?
A: Traditional methods rely on methylamine alcohol solutions, which are classified as easy-to-detonate controlled chemicals with strict purchasing regulations. The improved method utilizes inorganic bases like sodium hydroxide and water, eliminating the safety hazards and procurement difficulties associated with volatile organic amines.
Q: How does this process impact the purity profile of the final API?
A: By optimizing reaction conditions such as temperature (0°C) and stoichiometry (3:1 iodine ratio) during the iodination step, the process minimizes over-iodination and side reactions. This results in a cleaner crude product that requires less aggressive purification, thereby maintaining high optical purity and reducing impurity loads.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the replacement of hazardous organic solvents with aqueous systems significantly improves process safety and heat transfer capabilities. The use of readily available inorganic reagents and simplified workup procedures makes the route highly scalable and economically viable for multi-ton manufacturing campaigns.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Thyroxine Sodium Supplier
The technical advancements detailed in patent CN109810009B underscore the immense potential for optimizing the production of critical thyroid hormones, yet translating laboratory success into commercial reality requires a partner with deep process engineering expertise. NINGBO INNO PHARMCHEM stands at the forefront of this capability, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our commitment to quality is unwavering, supported by stringent purity specifications and rigorous QC labs that ensure every batch of L-thyroxine sodium meets the highest international pharmacopoeia standards. We understand that consistency is key in the pharmaceutical supply chain, and our state-of-the-art facilities are designed to deliver that reliability regardless of order volume.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized synthetic routes, we can help you reduce total landed costs while securing a stable supply of high-quality intermediates. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments that demonstrate how our manufacturing capabilities can support your long-term strategic goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
