Industrial Scale-Up of Alpha-Amino-Gamma-Butyrolactone via Aqueous Cyclization Technology
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable pathways for critical intermediates, and the production of alpha-amino-gamma-butyrolactone represents a significant area of innovation. As detailed in patent CN103282355A, a novel manufacturing method has been developed that fundamentally shifts the solvent paradigm from organic-heavy mixtures to water-dominant systems. This technological breakthrough addresses long-standing inefficiencies in the cyclization of methionine derivatives, offering a pathway that is not only chemically superior but also economically advantageous for large-scale operations. By leveraging a solvent composition containing 60% or more water by weight, this method achieves remarkable improvements in reaction conversion rates and final product isolation yields. For R&D directors and process chemists, this represents a viable alternative to legacy protocols that often struggle with mass balance issues and excessive solvent waste. The ability to produce high-purity alpha-amino-gamma-butyrolactone or its salts with enhanced efficiency positions this technology as a cornerstone for modern API intermediate manufacturing, ensuring a reliable supply chain for downstream drug synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alpha-amino-gamma-butyrolactone has relied on methodologies described in literature such as Tetrahedron Letters, which utilize complex mixed solvent systems. These conventional approaches typically employ a ternary mixture of water, 2-propanol, and acetic acid in specific volumetric ratios to facilitate the reaction between methionine and haloacetic acids. While functional on a laboratory scale, these methods present significant drawbacks when translated to industrial manufacturing. The reliance on substantial quantities of organic solvents like acetic acid and 2-propanol increases the overall cost of goods sold (COGS) and complicates the environmental footprint of the process due to the need for extensive solvent recovery and waste treatment. Furthermore, the reported yields for these traditional methods hover around 58.2%, which is suboptimal for high-value pharmaceutical intermediates where material throughput is critical. The presence of multiple organic components also necessitates rigorous distillation steps to concentrate the reaction mixture prior to crystallization, adding time, energy costs, and potential thermal degradation risks to the heat-sensitive lactone product.
The Novel Approach
In stark contrast, the innovative approach outlined in the patent data revolutionizes the process by prioritizing water as the primary reaction medium. By ensuring the solvent contains at least 60% by weight of water, and preferably upwards of 80% to 95%, the reaction environment becomes significantly more conducive to the nucleophilic substitution and subsequent lactonization steps. This shift eliminates the need for large volumes of expensive organic co-solvents during the critical reaction phase, thereby streamlining the process flow. The data indicates that this aqueous-dominant strategy dramatically accelerates the consumption rate of the haloacetic acid reactant, leading to much higher conversion efficiencies within shorter timeframes. Consequently, the isolation of the final alpha-amino-gamma-butyrolactone salt is achieved with yields reaching up to 75.5%, a substantial improvement over the legacy 58% benchmark. This method not only enhances the economic viability of the synthesis but also aligns with green chemistry principles by reducing the load of volatile organic compounds (VOCs) released during production.
Mechanistic Insights into Methionine Cyclization
The core of this synthesis involves the intramolecular cyclization of a methionine derivative following an initial alkylation with a haloacetic acid, such as chloroacetic acid or bromoacetic acid. The reaction mechanism proceeds through a nucleophilic attack where the sulfur atom of the methionine side chain initially interacts with the haloacetic acid, followed by a cascade of transformations that ultimately close the gamma-lactone ring. The presence of a high concentration of water plays a pivotal role in stabilizing the transition states and facilitating the ionization of the reactants, which is crucial for the reaction kinetics. Unlike organic solvents that might stabilize non-reactive conformers or lead to side reactions, the aqueous environment promotes the necessary polarity for the displacement of the halide leaving group. This mechanistic efficiency is visually represented in the general reaction scheme below, highlighting the transformation from the linear amino acid precursor to the cyclic lactone structure.
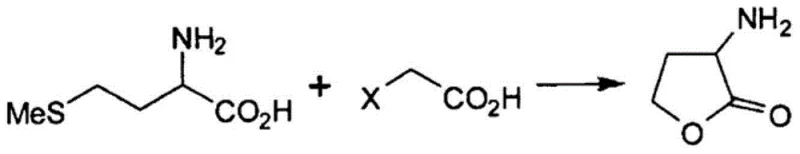
Beyond mere yield improvement, a critical aspect of this mechanism is the preservation of stereochemical integrity, which is paramount for pharmaceutical applications. When using optically active L-methionine as the starting material, there is an inherent risk of racemization under harsh acidic or basic conditions. The patent specifies that maintaining the reaction mixture pH within a weakly acidic range, specifically between pH 3 and 6.5, effectively suppresses epimerization at the alpha-carbon. This precise pH control ensures that the chiral center derived from the natural amino acid remains intact throughout the cyclization process. As a result, the final product exhibits high optical purity, with experimental data confirming enantiomeric excess (ee) values exceeding 99%. This level of stereocontrol is essential for producing intermediates destined for chiral drug molecules, where the presence of the wrong enantiomer could lead to regulatory rejection or reduced therapeutic efficacy.
How to Synthesize Alpha-Amino-Gamma-Butyrolactone Efficiently
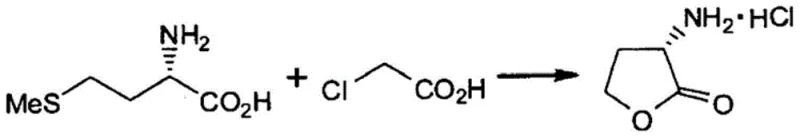
Implementing this high-yield synthesis requires careful attention to solvent ratios and temperature profiles to maximize the benefits of the aqueous system. The process begins with the preparation of a reaction mixture where L-methionine is suspended or dissolved in water, followed by the controlled addition of chloroacetic acid. The reaction is typically conducted at elevated temperatures, ranging from 65°C to 95°C, to ensure complete conversion while avoiding thermal decomposition. Following the reaction period, the workup procedure is streamlined; the mixture is washed with a hydrophobic solvent like ethyl acetate to remove non-polar impurities, leaving the desired product in the aqueous phase. The specific stoichiometry and conditions for a representative batch are illustrated in the reaction diagram below, which serves as a practical guide for scaling this chemistry from the bench to the pilot plant.
- React L-methionine with chloroacetic acid in a solvent containing greater than 60% water by weight at temperatures between 65°C and 95°C.
- Wash the reaction mixture with a hydrophobic solvent such as ethyl acetate to remove impurities, then concentrate the aqueous layer.
- Introduce hydrogen chloride gas or acid to the concentrate, cool the mixture, and induce crystallization using 2-propanol to isolate the high-purity salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this aqueous-based manufacturing route offers compelling strategic advantages that extend beyond simple chemistry. The most immediate impact is seen in the reduction of raw material costs, as water replaces a significant portion of the expensive organic solvents traditionally required for this synthesis. This substitution not only lowers the direct material spend but also mitigates the volatility associated with petrochemical-derived solvent pricing. Furthermore, the simplified downstream processing—characterized by fewer distillation steps and easier crystallization—translates into reduced utility consumption and shorter cycle times. These operational efficiencies allow for a more agile supply chain capable of responding rapidly to fluctuating market demands for pharmaceutical intermediates without the bottleneck of complex solvent recovery infrastructure.
- Cost Reduction in Manufacturing: The elimination of large volumes of acetic acid and 2-propanol from the reaction phase results in a drastic decrease in solvent procurement costs. Additionally, the higher reaction yield means that less starting material (methionine) is wasted per kilogram of final product, directly improving the overall material efficiency and lowering the cost per unit. The simplified workup also reduces the energy burden associated with heating and cooling large volumes of organic solvents, contributing to substantial long-term operational savings.
- Enhanced Supply Chain Reliability: By relying on water as the primary solvent, the process becomes less susceptible to supply disruptions common with specialized organic chemicals. Water is universally available and inexpensive, ensuring that production can continue uninterrupted even during periods of global chemical shortages. The robustness of the reaction conditions also allows for greater flexibility in sourcing raw materials, as the process tolerates standard commercial grades of chloroacetic acid without requiring ultra-high purity specifications.
- Scalability and Environmental Compliance: The reduction in organic solvent usage significantly lowers the facility's VOC emissions, making it easier to comply with increasingly stringent environmental regulations. This green chemistry profile facilitates smoother permitting processes for capacity expansion and reduces the costs associated with hazardous waste disposal. The process is inherently scalable, allowing manufacturers to increase batch sizes from hundreds of kilograms to multi-ton scales without encountering the heat transfer or mixing limitations often seen in viscous organic solvent systems.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of alpha-amino-gamma-butyrolactone using this advanced aqueous methodology. These insights are derived directly from the patent specifications and experimental data to provide clarity on process capabilities and quality standards. Understanding these details is crucial for partners evaluating the feasibility of integrating this intermediate into their own supply chains.
Q: How does the new aqueous solvent system improve yield compared to traditional methods?
A: Traditional methods utilizing mixed organic solvents like 2-propanol and acetic acid often result in yields around 58%. By shifting to a solvent system containing over 60% water, the reaction kinetics are optimized, pushing yields significantly higher, up to 75.5% in demonstrated examples, while simplifying the workup process.
Q: Is optical purity maintained during the cyclization of L-methionine?
A: Yes, maintaining the reaction mixture pH within a weakly acidic range (specifically pH 3 to 6.5) effectively prevents racemization. This ensures that the resulting alpha-amino-gamma-butyrolactone retains high optical purity, typically exceeding 99% ee when starting with optically active methionine.
Q: What are the primary cost drivers eliminated in this manufacturing process?
A: The process drastically reduces reliance on expensive organic solvents like acetic acid and large volumes of 2-propanol during the reaction phase. Furthermore, the simplified crystallization protocol reduces energy consumption associated with solvent recovery and distillation, leading to substantial operational cost savings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Amino-Gamma-Butyrolactone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of new therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering alpha-amino-gamma-butyrolactone with stringent purity specifications, utilizing rigorous QC labs to verify parameters such as optical purity and residual solvent levels. Our state-of-the-art facilities are equipped to handle the specific requirements of aqueous-based synthesis, guaranteeing a consistent and reliable supply for your pharmaceutical manufacturing needs.
We invite you to collaborate with us to optimize your supply chain and reduce your overall production costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your project goals and accelerate your time to market.
